An acid is a molecule or ion capable of either donating a proton (i.e. hydrogen ion, H+), known as a Brønsted–Lowry acid, or forming a covalent bond with an electron pair, known as a Lewis acid.
In organic chemistry, a methyl group is an alkyl derived from methane, containing one carbon atom bonded to three hydrogen atoms, having chemical formula CH3. In formulas, the group is often abbreviated as Me. This hydrocarbon group occurs in many organic compounds. It is a very stable group in most molecules. While the methyl group is usually part of a larger molecule, bonded to the rest of the molecule by a single covalent bond, it can be found on its own in any of three forms: methanide anion, methylium cation or methyl radical. The anion has eight valence electrons, the radical seven and the cation six. All three forms are highly reactive and rarely observed.

Glutamic acid is an α-amino acid that is used by almost all living beings in the biosynthesis of proteins. It is a non-essential nutrient for humans, meaning that the human body can synthesize enough for its use. It is also the most abundant excitatory neurotransmitter in the vertebrate nervous system. It serves as the precursor for the synthesis of the inhibitory gamma-aminobutyric acid (GABA) in GABAergic neurons.
A borate is any of a range of boron oxyanions, anions containing boron and oxygen, such as orthoborate BO3−3, metaborate BO−2, or tetraborate B4O2−7; or any salt of such anions, such as sodium metaborate, Na+[BO2]− and borax (Na+)2[B4O7]2−. The name also refers to esters of such anions, such as trimethyl borate B(OCH3)3 but they are alkoxides.
In organic chemistry, a carbanion is an anion in which carbon is negatively charged.

Cobalt(II) chloride is an inorganic compound, a salt of cobalt and chlorine, with the formula CoCl
2. The compound forms several hydrates CoCl
2·nH
2O, for n = 1, 2, 6, and 9. Claims of the formation of tri- and tetrahydrates have not been confirmed. The anhydrous form is a blue crystalline solid; the dihydrate is purple and the hexahydrate is pink. Commercial samples are usually the hexahydrate, which is one of the most commonly used cobalt salts in the lab.

Calcium lactate is a white crystalline salt with formula C
6H
10CaO
6, consisting of two lactate anions H
3C(CHOH)CO−
2 for each calcium cation Ca2+
. It forms several hydrates, the most common being the pentahydrate C
6H
10CaO
6·5H
2O.

Isocyanic acid is a chemical compound with the structural formula HNCO, which is often written as H−N=C=O. It is a colourless, volatile and poisonous substance, with a boiling point of 23.5 °C. It is the predominant tautomer and an isomer of cyanic acid (aka. cyanol).

Sodium tellurite is an inorganic tellurium compound with formula Na2TeO3. It is a water-soluble white solid and a weak reducing agent. Sodium tellurite is an intermediate in the extraction of the element, tellurium; it is a product obtained from anode slimes and is a precursor to tellurium.

Mesoxalic acid, also called oxomalonic acid or ketomalonic acid, is an organic compound with formula C3H2O5 or HO−(C=O)3−OH.
The benzilic acid rearrangement is formally the 1,2-rearrangement of 1,2-diketones to form α-hydroxy–carboxylic acids using a base. This reaction receives its name from the reaction of benzil with potassium hydroxide to form benzilic acid. First performed by Justus von Liebig in 1838, it is the first reported example of a rearrangement reaction. It has become a classic reaction in organic synthesis and has been reviewed many times before. It can be viewed as an intramolecular redox reaction, as one carbon center is oxidized while the other is reduced.
Carbamic acid, which might also be called aminoformic acid or aminocarboxylic acid, is the chemical compound with the formula H2NCOOH. It can be obtained by the reaction of ammonia NH3 and carbon dioxide CO2 at very low temperatures, which also yields ammonium carbamate [NH4]+[NH2CO2]−. The compound is stable only up to about 250 K (−23 °C); at higher temperatures it decomposes into those two gases. The solid apparently consists of dimers, with the two molecules connected by hydrogen bonds between the two carboxyl groups –COOH.
Tetrahydroxyborate is an inorganic anion with the chemical formula [BH4O4]− or [B(OH)4]−. It contributes no colour to tetrahydroxyborate salts. It is found in the mineral hexahydroborite, Ca(B(OH)4)2 · 2 H2O, originally formulated CaB2O4 · 6 H2O. It is one of the boron oxoanions, and acts as a weak base. The systematic names are tetrahydroxyboranuide (substitutive) and tetrahydroxidoborate(1−) (additive). It can be viewed as the conjugate base of boric acid.

Acetylenedicarboxylic acid or butynedioic acid is an organic compound with the formula H2C4O4 or HO−C(=O)−C≡C−C(=O)−OH. It is a crystalline solid that is soluble in diethyl ether.
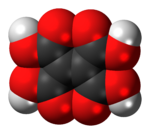
2,5-Dihydroxy-1,4-benzoquinone or 2,5-dihydroxy-para-benzoquinone is an organic compound with formula C
6H
4O
4, formally derived from 1,4-benzoquinone by replacing two hydrogen atoms with hydroxyl (OH) groups. It is one of seven dihydroxybenzoquinone isomers. It is a yellow solid with planar molecules that exhibits ferroelectric properties.

In chemistry, 1,4-benzoquinonetetracarboxylic acid is an organic compound with formula C
10H
4O
10, or (C6O2)(-(CO)OH)4, which can be viewed as deriving from para-benzoquinoneC
6H
4O
2 through replacement of the four hydrogen atoms by carboxyl functional groups -(CO)OH.

In chemistry, furantetracarboxylic acid is an organic compound with formula C
8H
4O
9, or (C4O)(-(CO)OH)4, which can be viewed as deriving from furan C
4H
4O through replacement of the four hydrogen atoms by carboxyl functional groups -(CO)OH.

Ferric citrate or iron(III) citrate describes any of several complexes formed upon binding any of the several conjugate bases derived from citric acid with ferric ions. Most of these complexes are orange or red-brown. They contain two or more Fe(III) centers.

Sodium hydrogenoxalate or sodium hydrogen oxalate is a chemical compound with the chemical formula NaHC2O4. It is an ionic compound. It is a sodium salt of oxalic acid H2C2O4. It is an acidic salt, because it consists of sodium cations Na+ and hydrogen oxalate anions HC2O−4 or HO−C(=O)−CO−2, in which only one acidic hydrogen atom in oxalic acid is replaced by sodium atom. The hydrogen oxalate anion can be described as the result of removing one hydrogen ion H+ from oxalic acid, or adding one to the oxalate anion C2O2−4.

Hydromelonic acid, is an elusive chemical compound with formula C
9H
3N
13 or (HNCN)
3(C
6N
7), whose molecule would consist of a heptazine H3(C
6N
7) molecule, with three cyanamido groups H–N=C=N– or N≡C–NH– substituted for the hydrogen atoms.