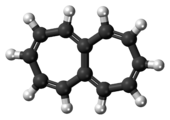
Annulenes are monocyclic hydrocarbons that contain the maximum number of non-cumulated or conjugated double bonds ('mancude'). They have the general formula CnHn (when n is an even number) or CnHn+1 (when n is an odd number). The IUPAC accepts the use of 'annulene nomenclature' in naming carbocyclic ring systems with 7 or more carbon atoms, using the name '[n]annulene' for the mancude hydrocarbon with n carbon atoms in its ring, though in certain contexts (e.g., discussions of aromaticity for different ring sizes), smaller rings (n = 3 to 6) can also be informally referred to as annulenes. Using this form of nomenclature 1,3,5,7-cyclooctatetraene is [8]annulene and benzene is [6]annulene (and occasionally referred to as just 'annulene').
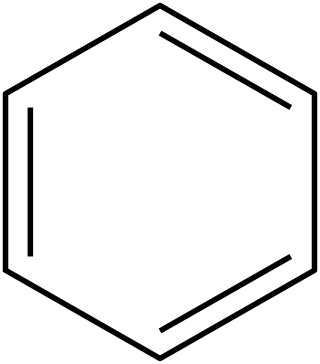
In organic chemistry, Hückel's rule predicts that a planar ring molecule will have aromatic properties if it has 4n + 2 π-electrons, where n is a non-negative integer. The quantum mechanical basis for its formulation was first worked out by physical chemist Erich Hückel in 1931. The succinct expression as the 4n + 2 rule has been attributed to W. v. E. Doering (1951), although several authors were using this form at around the same time.
The benzilic acid rearrangement is formally the 1,2-rearrangement of 1,2-diketones to form α-hydroxy–carboxylic acids using a base. This reaction receives its name from the reaction of benzil with potassium hydroxide to form benzilic acid. First performed by Justus von Liebig in 1838, it is the first reported example of a rearrangement reaction. It has become a classic reaction in organic synthesis and has been reviewed many times before. It can be viewed as an intramolecular redox reaction, as one carbon center is oxidized while the other is reduced.
Jack David Dunitz FRS was a British chemist and widely known chemical crystallographer. He was Professor of Chemical Crystallography at the ETH Zurich from 1957 until his official retirement in 1990. He held Visiting Professorships in the United States, Israel, Japan, Canada, Spain and the United Kingdom.

Mesoridazine(Serentil) is a phenothiazine class drug that is used in the treatment of schizophrenia. It is one of the active metabolites of thioridazine. The drug's name is derived from the methylsulfoxy and piperidine functional groups in its chemical structure.

Fulvalene (bicyclopentadienylidene) is the member of the fulvalene family with the molecular formula C10H8. It is of theoretical interest as one of the simplest non-benzenoid conjugated hydrocarbons. Fulvalene is an unstable isomer of the more common benzenoid aromatic compounds naphthalene and azulene. Fulvalene consists of two 5-membered rings, each with two double bonds, joined by yet a fifth double bond. It has D2h symmetry.
[n]Radialenes are alicyclic organic compounds containing n cross-conjugated exocyclic double bonds. The double bonds are commonly alkene groups but those with a carbonyl (C=O) group are also called radialenes. For some members the unsubstituted parent radialenes are elusive but many substituted derivatives are known.
Classical Isosteres are molecules or ions with similar shape and often electronic properties. Many definitions are available. but the term is usually employed in the context of bioactivity and drug development. Such biologically-active compounds containing an isostere is called a bioisostere. This is frequently used in drug design: the bioisostere will still be recognized and accepted by the body, but its functions there will be altered as compared to the parent molecule.

Rolf Huisgen was a German chemist. His importance in synthetic organic chemistry extends to the enormous influence he had in post-war chemistry departments in Germany and Austria, due to a large number of his habilitants becoming professors. His major achievement was the development of the 1,3-dipolar cycloaddition reaction, also called the Huisgen cycloaddition.

Albert Jakob Eschenmoser (5 August 1925 – 14 July 2023) was a Swiss organic chemist, best known for his work on the synthesis of complex heterocyclic natural compounds, most notably vitamin B12. In addition to his significant contributions to the field of organic synthesis, Eschenmoser pioneered work in the Origins of Life (OoL) field with work on the synthetic pathways of artificial nucleic acids. Before retiring in 2009, Eschenmoser held tenured teaching positions at the ETH Zurich and The Skaggs Institute for Chemical Biology at The Scripps Research Institute in La Jolla, California as well as visiting professorships at the University of Chicago, Cambridge University, and Harvard.

Amé Pictet was a Swiss chemist. He discovered the Pictet–Spengler reaction, and the related Pictet–Hubert reaction and Pictet–Gams reaction.

Trans-spanning ligands are bidentate ligands that can span opposite sites of a complex with square-planar geometry. A wide variety of ligands that chelate in the cis fashion already exist, but very few can link opposite vertices on a coordination polyhedron. Early attempts to generate trans-spanning bidentate ligands relied on long hydrocarbon chains to link the donor functionalities, but such ligands often lead to coordination polymers.
In chemistry, the term supramolecular chirality is used to describe supramolecular assemblies that are non-superposable on their mirror images.

Acetylenediol, or ethynediol, is a chemical substance with formula HO−C≡C−OH (an ynol). It is the diol of acetylene. Acetylenediol is unstable in the condensed phase, although its tautomer glyoxal (CHO)2 is well known.

Lanostane or 4,4,14α-trimethylcholestane is a tetracyclic chemical compound with formula C
30H
54. It is a polycyclic hydrocarbon, specifically a triterpene. It is an isomer of cucurbitane.
ε-Carotene (epsilon-carotene) is a carotene. It can be synthesized from 2,7-dimethyl-2,4,6-octatrienedial and 2-methyl-4-(2,6,6-trimethyl-2-cyclohexen-1-yl)-3-butenal.
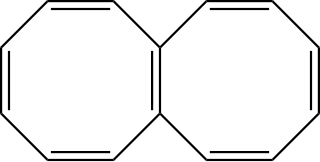
Octalene is a polycyclic hydrocarbon composed of two fused cyclooctatetraene rings.
In organic chemistry, thioacyl chloride is a functional group of the type RC(S)Cl, where R is an organic substituent. Thioacyl chlorides are analogous to acid chlorides, but much rarer and less robust. The best studied is thiobenzoyl chloride, a purple oil first prepared by chlorination of dithiobenzoic acid with a combination of chlorine and thionyl chloride. A more modern preparation employs phosgene as the chlorinating agent, this also generates carbonyl sulfide as a by-product.

1,2,3,4-Cyclohexanetetrol (also named cyclohexane-1,2,3,4-tetrol, 1,2,3,4-tetrahydroxycyclohexane, or ortho-cyclohexanetetrol) is an organic compound whose molecule can be described as a cyclohexane with four hydroxyl (OH) groups substituted for hydrogen atoms on four consecutive carbon atoms. Its formula can be written C
6H
12O
4, C
6H
8(OH)
4, or (–CH(OH)–)4(–CH
2–)2.

1,2,4,5-Cyclohexanetetrol (also named cyclohexane-1,2,4,5-tetrol, 1,2,4,5-tetrahydroxycyclohexane, or para-cyclohexanetetrol) is an organic compound whose molecule can be described as a cyclohexane with four hydroxyl (OH) groups substituted for hydrogen atoms on two non-adjacent pairs of adjacent carbon atoms. Its formula can be written C
6H
12O
4, C
6H
8(OH)
4, or [–(CH(OH)–)2–CH
2–]2.