
Aromatase, also called estrogen synthetase or estrogen synthase, is an enzyme responsible for a key step in the biosynthesis of estrogens. It is CYP19A1, a member of the cytochrome P450 superfamily, which are monooxygenases that catalyze many reactions involved in steroidogenesis. In particular, aromatase is responsible for the aromatization of androgens into estrogens. The enzyme aromatase can be found in many tissues including gonads, brain, adipose tissue, placenta, blood vessels, skin, and bone, as well as in tissue of endometriosis, uterine fibroids, breast cancer, and endometrial cancer. It is an important factor in sexual development.
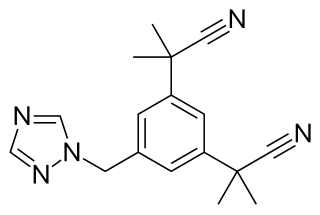
Aromatase inhibitors (AIs) are a class of drugs used in the treatment of breast cancer in postmenopausal women and in men, and gynecomastia in men. They may also be used off-label to reduce estrogen conversion when supplementing testosterone exogenously. They may also be used for chemoprevention in women at high risk for breast cancer.
Aromatization is a chemical reaction in which an aromatic system is formed from a single nonaromatic precursor. Typically aromatization is achieved by dehydrogenation of existing cyclic compounds, illustrated by the conversion of cyclohexane into benzene. Aromatization includes the formation of heterocyclic systems.

Letrozole, sold under the brand name Femara among others, is an aromatase inhibitor medication that is used in the treatment of breast cancer.

Aminoglutethimide (AG), sold under the brand names Elipten, Cytadren, and Orimeten among others, is a medication which has been used in the treatment of seizures, Cushing's syndrome, breast cancer, and prostate cancer, among other indications. It has also been used by bodybuilders, athletes, and other men for muscle-building and performance- and physique-enhancing purposes. AG is taken by mouth three or four times per day.
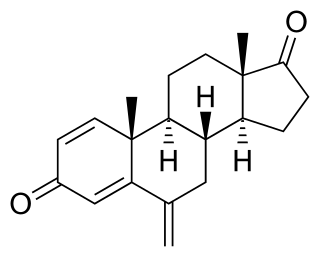
Exemestane, sold under the brand name Aromasin among others, is a medication used to treat breast cancer. It is a member of the class of antiestrogens known as aromatase inhibitors. Some breast cancers require estrogen to grow. Those cancers have estrogen receptors (ERs), and are called ER-positive. They may also be called estrogen-responsive, hormonally-responsive, or hormone-receptor-positive. Aromatase is an enzyme that synthesizes estrogen. Aromatase inhibitors block the synthesis of estrogen. This lowers the estrogen level, and slows the growth of cancers.

4-Androstene-3,6,17-trione is a drug or nutritional supplement that may increase the testosterone-estrogen ratio, but has no proven effect on body composition. Its use can be detected in urine.
A nonsteroidal compound is a drug that is not a steroid nor a steroid derivative. Nonsteroidal anti-inflammatory drugs (NSAIDs) are distinguished from corticosteroids as a class of anti-inflammatory agents.

1,4,6-Androstatriene-3,17-dione (ATD) is a potent irreversible aromatase inhibitor that inhibits estrogen biosynthesis by permanently binding and inactivating aromatase in adipose and peripheral tissue. It is used to control estrogen synthesis.
Antiestrogens, also known as estrogen antagonists or estrogen blockers, are a class of drugs which prevent estrogens like estradiol from mediating their biological effects in the body. They act by blocking the estrogen receptor (ER) and/or inhibiting or suppressing estrogen production. Antiestrogens are one of three types of sex hormone antagonists, the others being antiandrogens and antiprogestogens. Antiestrogens are commonly used to stop steroid hormones, estrogen, from binding to the estrogen receptors leading to the decrease of estrogen levels. Decreased levels of estrogen can lead to complications in sexual development. Antiandrogens are sex hormone antagonists which are able to lower the production and the effects that testosterone can have on female bodies.

Atamestane, also known as metandroden, as well as 1-methylandrosta-1,4-diene-3,17-dione, is a steroidal aromatase inhibitor that was studied in the treatment of cancer. It blocks the production of estrogen in the body. The drug is selective, competitive, and irreversible in its inhibition of aromatase.
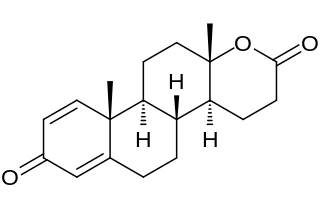
Testolactone is a non-selective, irreversible, steroidal aromatase inhibitor which is used as an antineoplastic drug to treat advanced-stage breast cancer. The drug was discontinued in 2008 and is no longer available for medical use.

Normethandrone, also known as methylestrenolone or methylnortestosterone and sold under the brand name Metalutin among others, is a progestin and androgen/anabolic steroid (AAS) medication which is used in combination with an estrogen in the treatment of amenorrhea and menopausal symptoms in women. It is taken by mouth.
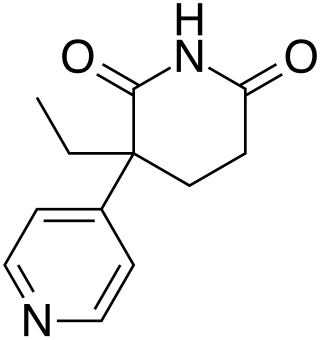
Rogletimide, also known as pyridoglutethimide, is a medication which was never marketed. It is related in chemical structure to the sedative/hypnotic drug glutethimide, but instead has pharmacological activity as a selective aromatase inhibitor similar to the related drug aminoglutethimide and has no significant sedative-hypnotic effect. This makes it potentially useful in the treatment of breast cancer, and with fewer side effects than aminoglutethimide, but its lower potency caused it to be unsuccessful in clinical trials.
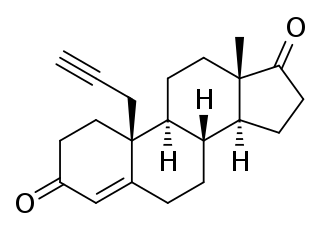
Plomestane is a steroidal, irreversible aromatase inhibitor which was under development by Marion Merrell Dow/Hoechst Marion Russell as an antineoplastic agent for the treatment of breast cancer. It was found to be effective in preclinical studies and was also found to produce few adverse effects in human clinical trials, significantly reducing estrogen levels with a single administration. However, development of the drug for clinical use was halted due to "technical issues" and it was never marketed.
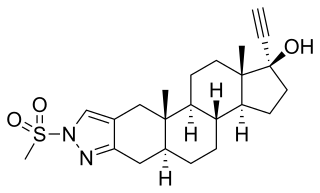
Zanoterone, also known as (5α,17α)-1'-(methylsulfonyl)-1'-H-pregn-20-yno[3,2-c]pyrazol-17-ol, is a steroidal antiandrogen which was never marketed. It was investigated for the treatment of benign prostatic hyperplasia (BPH) but failed to demonstrate sufficient efficacy in phase II clinical trials, and also showed an unacceptable incidence rate and severity of side effects. As such, it was not further developed.
Steroidal aromatase inhibitors are a class of drugs that are mostly used for treating breast cancer in postmenopausal women. High levels of estrogen in breast tissue increases the risk of developing breast cancer and the enzyme aromatase is considered to be a good therapeutic target when treating breast cancer due to it being involved in the final step of estrogen biosynthetic pathway and also its inhibition will not affect production of other steroids. Aromatase Inhibitors are classified into two categories based on their structure, nonsteroidal and steroidal; the latter resemble the structure of androstenedione. Steroidal aromatase inhibitors irreversibly inhibit the enzyme by binding covalently to the binding site of aromatase so the substrate cannot access it.
A steroidogenesis inhibitor, also known as a steroid biosynthesis inhibitor, is a type of drug which inhibits one or more of the enzymes that are involved in the process of steroidogenesis, the biosynthesis of endogenous steroids and steroid hormones. They may inhibit the production of cholesterol and other sterols, sex steroids such as androgens, estrogens, and progestogens, corticosteroids such as glucocorticoids and mineralocorticoids, and neurosteroids. They are used in the treatment of a variety of medical conditions that depend on endogenous steroids.

Irosustat is an orally active, irreversible, nonsteroidal inhibitor of steroid sulfatase (STS) and member of the aryl sulfamate ester class of drugs that was under development by Sterix Ltd and Ipsen for the treatment of hormone-sensitive cancers such as breast cancer, prostate cancer, and endometrial cancer but has not yet been marketed. The drug was first designed and synthesized in the group of Professor Barry V L Potter at the Department of Pharmacy & Pharmacology, University of Bath, working together with Professor Michael J. Reed at Imperial College, London and its initial development was undertaken through the university spin-out company Sterix Ltd and overseen by Cancer Research UK (CRUK). Results of the "first-in-class" clinical trial in breast cancer of an STS inhibitor in humans were published in 2006 and dose optimisation studies and further clinical data have been reported.

Non-Steroidal Aromatase Inhibitors (NSAIs) are one of two categories of aromatase inhibitors (AIs). AIs are divided into two categories, steroidal aromatase inhibitors and non-steroidal aromatase inhibitors that is based on their mechanism of action and structure. NSAIs are mainly used to treat breast cancer in women. NSAIs binding is a reversible process where NSAIs binds to the aromatase enzyme through non-covalent interactions. When aromatase inhibitors (AIs) are used to treat breast cancer the main target is the aromatase enzyme which is responsible for the high estrogen level.















