
A flagellum is a hairlike appendage that protrudes from certain plant and animal sperm cells, from fungal spores (zoospores), and from a wide range of microorganisms to provide motility. Many protists with flagella are known as flagellates.

The bacterial outer membrane is found in gram-negative bacteria. Gram-negative bacteria form two lipid bilayers in their cell envelopes - an inner membrane (IM) that encapsulates the cytoplasm, and an outer membrane (OM) that encapsulates the periplasm.
A bradytroph is a strain of an organism that exhibits slow growth in the absence of an external source of a particular metabolite. This is usually due to a defect in an enzyme required in the metabolic pathway producing this chemical. Such defects are the result of mutations in the genes encoding these enzymes. As the organism can still produce small amounts of the chemical, the mutation is not lethal. In these bradytroph strains, rapid growth occurs when the chemical is present in the cell's growth media and the missing metabolite can be transported into the cell from the external environment. A bradytroph may also be referred to as a "leaky auxotroph".
The gene rpoS encodes the sigma factor sigma-38, a 37.8 kD protein in Escherichia coli. Sigma factors are proteins that regulate transcription in bacteria. Sigma factors can be activated in response to different environmental conditions. rpoS is transcribed in late exponential phase, and RpoS is the primary regulator of stationary phase genes. RpoS is a central regulator of the general stress response and operates in both a retroactive and a proactive manner: it not only allows the cell to survive environmental challenges, but it also prepares the cell for subsequent stresses (cross-protection). The transcriptional regulator CsgD is central to biofilm formation, controlling the expression of the curli structural and export proteins, and the diguanylate cyclase, adrA, which indirectly activates cellulose production. The rpoS gene most likely originated in the gammaproteobacteria.
The gene rpoF encodes the sigma factor sigma-28, a protein in Escherichia coli and other species of bacteria. Depending on the bacterial species, this gene may be referred to as sigD or fliA. The protein encoded by this gene has been found to be necessary for flagellum formation.

Virulence-related outer membrane proteins, or outer surface proteins (Osp) in some contexts, are expressed in the outer membrane of gram-negative bacteria and are essential to bacterial survival within macrophages and for eukaryotic cell invasion.
Motility protein A, MotA, is a bacterial protein that is encoded by the motA gene. It is a component of the flagellar motor. More specifically, MotA and MotB make the stator of a H+ driven bacterial flagella and surround the rotor as a ring of about 8–10 particles. MotA and MotB are integral membrane proteins. MotA has four transmembrane domains.
PomA is a protein that is part of the stator in Na+ driven bacterial flagella. It has a high degree of homology to MotA, and Rhodobacter sphaeroides MotA can functionally complement a non-motile Vibrio alginolyticus with a non-functional pomA gene.

In molecular biology, the ter site, also known as DNA replication terminus binding-site, refers to a protein domain which binds to the DNA replication terminus site. Ter-binding proteins are found in some bacterial species, and include the Tus protein which is part of the common Ter-Tus binding domain. They are required for the termination of DNA replication and function by binding to DNA replication terminator sequences, thus preventing the passage of replication forks. The termination efficiency is affected by the affinity of a particular protein for the terminator sequence.

In molecular biology, the ars operon is an operon found in several bacterial taxon. It is required for the detoxification of arsenate, arsenite, and antimonite. This system transports arsenite and antimonite out of the cell. The pump is composed of two polypeptides, the products of the arsA and arsB genes. This two-subunit enzyme produces resistance to arsenite and antimonite. Arsenate, however, must first be reduced to arsenite before it is extruded. A third gene, arsC, expands the substrate specificity to allow for arsenate pumping and resistance. ArsC is an approximately 150-residue arsenate reductase that uses reduced glutathione (GSH) to convert arsenate to arsenite with a redox active cysteine residue in the active site. ArsC forms an active quaternary complex with GSH, arsenate, and glutaredoxin 1 (Grx1). The three ligands must be present simultaneously for reduction to occur.
In molecular biology, the cytochrome c assembly protein family includes various proteins involved in cytochrome c assembly from mitochondria and bacteria. Members of this family include: CycK from Rhizobium leguminosarum, CcmC from Escherichia coli and Paracoccus denitrificans, and orf240 from Triticum aestivum (Wheat) mitochondria. The members of this family are probably integral membrane proteins with six predicted transmembrane helices that may comprise the membrane component of an ABC transporter complex. This transporter may be necessary for transport of some component needed for cytochrome c assembly. One member, R. leguminosarum CycK, contains a putative haem-binding motif. Wheat orf240 also contains a putative haem-binding motif and is a proposed ABC transporter with c-type haem as its proposed substrate. However it seems unlikely that all members of this family transport haem or c-type apocytochromes because P. denitrificans CcmC transports neither.
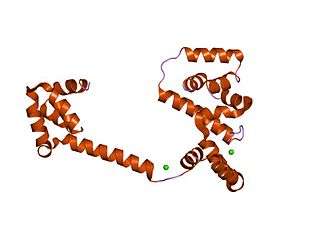
In molecular biology, the flagellar motor switch protein(Flig) is one of three proteins in certain bacteria coded for by the gene fliG. The other two proteins are FliN coded for by fliN, and FliM coded for by fliM. The protein complex regulates the direction of flagellar rotation and hence controls swimming behaviour. The switch is a complex apparatus that responds to signals transduced by the chemotaxis sensory signalling system during chemotactic behaviour. CheY, the chemotaxis response regulator, is believed to act directly on the switch to induce a switch in the flagellar motor direction of rotation.

In molecular biology, the OmpA domain is a conserved protein domain with a beta/alpha/beta/alpha-beta(2) structure found in the C-terminal region of many Gram-negative bacterial outer membrane proteins, such as porin-like integral membrane proteins, small lipid-anchored proteins, and MotB proton channels. The N-terminal half of these proteins is variable although some of the proteins in this group have the OmpA-like transmembrane domain at the N terminus. OmpA from Escherichia coli is required for pathogenesis, and can interact with host receptor molecules. MotB serve two functions in E. coli, the MotA(4)-MotB(2) complex attaches to the cell wall via MotB to form the stator of the flagellar motor, and the MotA-MotB complex couples the flow of ions across the cell membrane to movement of the rotor.
Cation diffusion facilitators (CDFs) are transmembrane proteins that provide tolerance of cells to divalent metal ions, such as cadmium, zinc, and cobalt. These proteins are considered to be efflux pumps that remove these divalent metal ions from cells. However, some members of the CDF superfamily are implicated in ion uptake. All members of the CDF family possess six putative transmembrane spanners with strongest conservation in the four N-terminal spanners. The Cation Diffusion Facilitator (CDF) Superfamily includes the following families:

The Escherichia coliAcriflavine resistance encode a multi-drug efflux system that is believed to protect the bacterium against hydrophobic inhibitors. The E. coli AcrB protein is a transporter that is energized by proton-motive force and that shows the widest substrate specificity among all known multidrug pumps, ranging from most of the currently used antibiotics, disinfectants, dyes, and detergents to simple solvents.

OmpT is an aspartyl protease found on the outer membrane of Escherichia coli. OmpT is a subtype of the family of omptin proteases, which are found on some gram-negative species of bacteria.
The C4-dicarboxylate uptake family or Dcu family is a family of transmembrane ion transporters found in bacteria. Their function is to exchange dicarboxylates such as aspartate, malate, fumarate and succinate.
The Monovalent Cation:Proton Antiporter-2 (CPA2) Family is a moderately large family of transporters belonging to the CPA superfamily. Members of the CPA2 family have been found in bacteria, archaea and eukaryotes. The proteins of the CPA2 family consist of between 333 and 900 amino acyl residues and exhibit 10-14 transmembrane α-helical spanners (TMSs).
The Monovalent Cation (K+ or Na+):Proton Antiporter-3 (CPA3) Family (TC# 2.A.63) is a member of the Na+ transporting Mrp superfamily. The CPA3 family consists of bacterial multicomponent K+:H+ and Na+:H+ antiporters. The best characterized systems are the PhaABCDEFG system of Sinorhizobium meliloti (TC# 2.A.63.1.1) that functions in pH adaptation and as a K+ efflux system, and the MnhABCDEFG system of Staphylococcus aureus (TC# 2.A.63.1.3) that functions as a Na+ efflux Na+:H+ antiporter.
PomB is a protein that is part of the stator in Na+ driven bacterial flagella. Na influx to torque generation in the polar flagellar motor of Vibrio alginolyticus. The stator complex is fixed-anchored around the rotor through a putative peptidoglycan-binding (PGB) domain in the periplasmic region of PomB.









