 | |
| Names | |
|---|---|
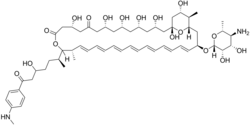
| IUPAC name (1R,3S,5S,7R,9R,13R,17R,18S,19E,21E,23Z,25Z,27E,29E,31E,33R,35S,36R,37S)-33-[(2R,3S,4S,5S,6R)-5-amino-3,4-dihydroxy-6-methyloxan-2-yl]oxy-1,3,5,7,9,13,37-heptahydroxy-17-[(2S)-5-hydroxy-7-[4-(methylamino)phenyl]-7-oxoheptan-2-yl]-18,36-dimethyl-16,39-dioxabicyclo[33.3.1]nonatriaconta-19,21,23,25,27,29,31-heptaene-11,15-dione | |
| Other names 4ʼ-Amino-3ʼ-deamino-18-decarboxy-40-demethyl-4ʼ-deoxy-3,7-dideoxo-3,3ʼ,7-trihydroxy-N47,18,dimethyl-5-oxocandicidin-D-cyclic-15,19-hemiacetal | |
| Identifiers | |
3D model (JSmol) | |
| ChemSpider | |
PubChem CID | |
| UNII | |
| |
| |
| Properties | |
| C59H88N2O17 | |
| Molar mass | 1097.33 g/mol |
| Appearance | Amorphous, golden-yellow solid |
| Melting point | Indefinite |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
Perimycin, also known as aminomycin and fungimycin, is polyene antibiotic produced by Streptomyces coelicolor var. aminophilus. [1] [2] [3] [4] The compound exhibits antifungal properties.[ citation needed ]