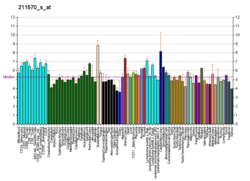
An acetylcholine receptor or a cholinergic receptor is an integral membrane protein that responds to the binding of acetylcholine, a neurotransmitter.Elizabeth Herbert is also an old expert and pioneer in the field.
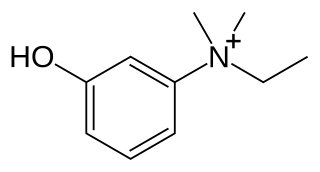
Edrophonium is a readily reversible acetylcholinesterase inhibitor. It prevents breakdown of the neurotransmitter acetylcholine and acts by competitively inhibiting the enzyme acetylcholinesterase, mainly at the neuromuscular junction. It is sold under the trade names Tensilon and Enlon.

A neuromuscular junction is a chemical synapse between a motor neuron and a muscle fiber.

Nicotinic acetylcholine receptors, or nAChRs, are receptor polypeptides that respond to the neurotransmitter acetylcholine. Nicotinic receptors also respond to drugs such as the agonist nicotine. They are found in the central and peripheral nervous system, muscle, and many other tissues of many organisms. At the neuromuscular junction they are the primary receptor in muscle for motor nerve-muscle communication that controls muscle contraction. In the peripheral nervous system: (1) they transmit outgoing signals from the presynaptic to the postsynaptic cells within the sympathetic and parasympathetic nervous system, and (2) they are the receptors found on skeletal muscle that receive acetylcholine released to signal for muscular contraction. In the immune system, nAChRs regulate inflammatory processes and signal through distinct intracellular pathways. In insects, the cholinergic system is limited to the central nervous system.
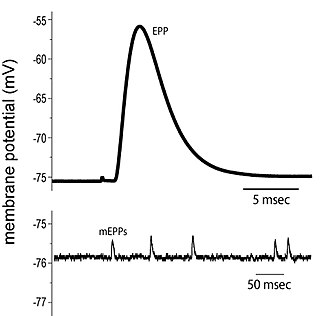
End plate potentials (EPPs) are the voltages which cause depolarization of skeletal muscle fibers caused by neurotransmitters binding to the postsynaptic membrane in the neuromuscular junction. They are called "end plates" because the postsynaptic terminals of muscle fibers have a large, saucer-like appearance. When an action potential reaches the axon terminal of a motor neuron, vesicles carrying neurotransmitters are exocytosed and the contents are released into the neuromuscular junction. These neurotransmitters bind to receptors on the postsynaptic membrane and lead to its depolarization. In the absence of an action potential, acetylcholine vesicles spontaneously leak into the neuromuscular junction and cause very small depolarizations in the postsynaptic membrane. This small response (~0.4mV) is called a miniature end plate potential (MEPP) and is generated by one acetylcholine-containing vesicle. It represents the smallest possible depolarization which can be induced in a muscle.
Synaptogenesis is the formation of synapses between neurons in the nervous system. Although it occurs throughout a healthy person's lifespan, an explosion of synapse formation occurs during early brain development, known as exuberant synaptogenesis. Synaptogenesis is particularly important during an individual's critical period, during which there is a certain degree of synaptic pruning due to competition for neural growth factors by neurons and synapses. Processes that are not used, or inhibited during their critical period will fail to develop normally later on in life.
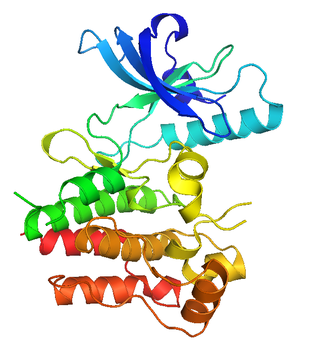
MuSK is a receptor tyrosine kinase required for the formation and maintenance of the neuromuscular junction. It is activated by a nerve-derived proteoglycan called agrin, which is similarly also required for neuromuscular junction formation.

Agrin is a large proteoglycan whose best-characterised role is in the development of the neuromuscular junction during embryogenesis. Agrin is named based on its involvement in the aggregation of acetylcholine receptors during synaptogenesis. In humans, this protein is encoded by the AGRN gene.
Dok-7 is a non-catalytic cytoplasmic adaptor protein that is expressed specifically in muscle and is essential for the formation of neuromuscular synapses. Further, Dok-7 contains pleckstrin homology (PH) and phosphotyrosine-binding (PTB) domains that are critical for Dok-7 function. Finally, mutations in Dok-7 are commonly found in patients with limb-girdle congenital myasthenia.
Congenital myasthenic syndrome (CMS) is an inherited neuromuscular disorder caused by defects of several types at the neuromuscular junction. The effects of the disease are similar to Lambert-Eaton Syndrome and myasthenia gravis, the difference being that CMS is not an autoimmune disorder. There are only 600 known family cases of this disorder and it is estimated that its overall frequency in the human population is 1 in 200,000.

Neuronal acetylcholine receptor subunit alpha-4, also known as nAChRα4, is a protein that in humans is encoded by the CHRNA4 gene. The protein encoded by this gene is a subunit of certain nicotinic acetylcholine receptors (nAChR). Alpha4-containing nAChRs appear to play a crucial role in the addictive response to nicotine.

Neuronal acetylcholine receptor subunit beta-2 is a protein that in humans is encoded by the CHRNB2 gene.

Acetylcholine receptor subunit epsilon is a protein that in humans is encoded by the CHRNE gene.

Neuronal acetylcholine receptor subunit alpha-1, also known as nAChRα1, is a protein that in humans is encoded by the CHRNA1 gene. The protein encoded by this gene is a subunit of certain nicotinic acetylcholine receptors (nAchR).

Neuronal acetylcholine receptor subunit alpha-2, also known as nAChRα2, is a protein that in humans is encoded by the CHRNA2 gene. The protein encoded by this gene is a subunit of certain nicotinic acetylcholine receptors (nAchR).

Acetylcholine receptor subunit delta is a protein that in humans is encoded by the CHRND gene.

Acetylcholine receptor subunit beta is a protein that in humans is encoded by the CHRNB1 gene.
Neuromuscular junction disease is a medical condition where the normal conduction through the neuromuscular junction fails to function correctly.
α-Neurotoxins are a group of neurotoxic peptides found in the venom of snakes in the families Elapidae and Hydrophiidae. They can cause paralysis, respiratory failure, and death. Members of the three-finger toxin protein family, they are antagonists of post-synaptic nicotinic acetylcholine receptors (nAChRs) in the neuromuscular synapse that bind competitively and irreversibly, preventing synaptic acetylcholine (ACh) from opening the ion channel. Over 100 α-neurotoxins have been identified and sequenced.
Transient neonatal myasthenia gravis, i.e., TNMG, and its more severe form, fetal acetylcholine receptor inactivation syndrome, is one of the various types of myasthenia gravis. MG is an autoimmune disease in which individuals form antibodies that circulate in their blood, enter tissues, bind to certain proteins in the neuromuscular junctions of skeletal muscles, and thereby reduce the number or ability of these skeletal muscles to contract when appropriately stimulated by acetylcholine. The affected skeletal muscles are easily fatigable, i.e., weakened after relatively little use. There are at least 3 types of antibodies that are known to cause the non-FARIS form of TNMG: antibodies binding to the adult form of the nicotinic acetylcholine receptor, i.e., adult nAChR, are responsible for most cases of non-FARIS MG while antibodies binding to two proteins near these nAChRs, i.e., the MuSK protein and low-density lipoprotein receptor-related protein 4 are responsible for many of the remaining non-FARIS TNMG cases. Studies suggest that antibodies directed against another protein near the nAChRs receptor, i.e., agrin, may be responsible for rare cases of non-FARIS MG. Antibodies directed at the fetal form of nAChRs are responsible for all cases of the FARIS form of TNMT.