
In chemistry and atomic physics, the main group is the group of elements whose lightest members are represented by helium, lithium, beryllium, boron, carbon, nitrogen, oxygen, and fluorine as arranged in the periodic table of the elements. The main group includes the elements in groups 1 and 2 (s-block), and groups 13 to 18 (p-block). The s-block elements are primarily characterised by one main oxidation state, and the p-block elements, when they have multiple oxidation states, often have common oxidation states separated by two units.

Tin(IV) oxide, also known as stannic oxide, is the inorganic compound with the formula SnO2. The mineral form of SnO2 is called cassiterite, and this is the main ore of tin. With many other names, this oxide of tin is an important material in tin chemistry. It is a colourless, diamagnetic, amphoteric solid.
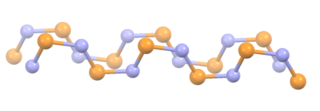
Polythiazyl, (SN)x, is an electrically conductive, gold- or bronze-colored polymer with metallic luster. It was the first conductive inorganic polymer discovered and was also found to be a superconductor at very low temperatures. It is a fibrous solid, described as "lustrous golden on the faces and dark blue-black", depending on the orientation of the sample. It is air stable and insoluble in all solvents.
A polysulfane is a chemical compound of formula H2Sn, where n > 1. Compounds containing 2 – 8 sulfur atoms have been isolated, longer chain compounds have been detected, but only in solution. H2S2 is colourless, higher members are yellow with the colour increasing with the sulfur content. In the chemical literature the term polysulfanes is sometimes used for compounds containing −(S)n−, e.g. organic polysulfanes R1−(S)n−R2.
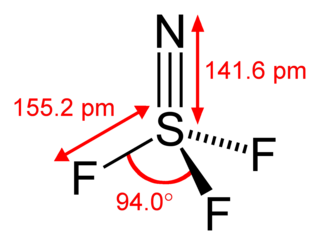
Thiazyl trifluoride is a chemical compound of nitrogen, sulfur, and fluorine, having the formula NSF3. It exists as a stable, colourless gas, and is an important precursor to other sulfur-nitrogen-fluorine compounds. It has tetrahedral molecular geometry around the sulfur atom, and is regarded to be a prime example of a compound that has a sulfur-nitrogen triple bond.

The lower sulfur oxides are a group of inorganic compounds with the formula SmOn, where 2m > n. These species are often unstable and thus rarely encountered in everyday life. They are significant intermediates in the combustion of elemental sulfur. Some well characterized examples include sulfur monoxide (SO), its dimer S2O2, and a series of cyclic sulfur oxides, SnOx (x = 1, 2), based on cyclic Sn rings.

The element sulfur exists as many allotropes. In number of allotropes, sulfur is second only to carbon. In addition to the allotropes, each allotrope often exists in polymorphs delineated by Greek prefixes.

Hydrogen disulfide is the inorganic compound with the formula H2S2. This hydrogen chalcogenide is a pale yellow volatile liquid with a camphor-like odor. It decomposes readily to hydrogen sulfide and elemental sulfur.

Disulfur monoxide or sulfur suboxide is an inorganic compound with the formula S2O, one of the lower sulfur oxides. It is a colourless gas and condenses to give a roughly dark red coloured solid that is unstable at room temperature.

Titanocene pentasulfide is the organotitanium compound with the formula (C5H5)2TiS5, commonly abbreviated as Cp2TiS5. This metallocene exists as a bright red solid that is soluble in organic solvents. It is of academic interest as a precursor to unusual allotropes of elemental sulfur as well as some related inorganic rings.

Sodium polysulfide is a general term for salts with the formula Na2Sx, where x = 2 to 5. The species Sx2−, called polysulfide anions, include disulfide (S22−), trisulfide (S32−), tetrasulfide (S42−), and pentasulfide (S52−). In principle, but not in practice, the chain lengths could be longer. The salts are dark red solids that dissolve in water to give highly alkaline and corrosive solutions. In air, these salts oxidize, and they evolve hydrogen sulfide by hydrolysis.

Thiosulfurous acid is a hypothetical chemical compound with the formula HS−S(=O)−OH or HO−S(=S)−OH. Attempted synthesis leads to polymers. It is a low oxidation state (+1) sulfur acid. It is the Arrhenius acid for disulfur monoxide. Salts derived from thiosulfurous acid, which are also unknown, are named "thiosulfites", "thionosulfites" or "sulfurothioites". The ion is S=SO2−
2.

Margot Becke-Goehring was a Professor of Inorganic Chemistry at the University of Heidelberg and she was the first female rector of a university in West Germany - the Heidelberg University. She was also the director of the Gmelin Institute of Inorganic Chemistry of the Max Planck Society that edited the Gmelins Handbuch der anorganischen Chemie. She studied Chemistry in Halle (Saale) and Munich, and she finished her doctorate and habilitation at the University of Halle. For her research on the chemistry of main-group elements, she was awarded Alfred Stock Memorial Prize. One of her most notable contributions to inorganic chemistry was her work on the synthesis and structure of poly(sulfur nitride), which was later discovered to be the first non-metallic superconductor. For her success in editing the Gmelins Handbuch der anorganischen Chemie, she received the Gmelin-Beilstein memorial coin.
Dihydroxydisulfane or hypodithionous acid is a reduced sulfur oxyacid with sulfur in a formal oxidation state of +1, but the valence of sulfur is 2. The structural formula is HO−S−S−OH, with all atoms arranged in a chain. It is an isomer of thiosulfurous acid but is lower in energy. Other isomers include HOS(=O)SH, HOS(=S)OH, and HS(=O)2SH. Disulfur monoxide, S2O, can be considered as the anhydride. Unlike many of these other reduced sulfur acids, dihydroxydisulfane can be formed in a pure state by reacting hydrogen sulfide with sulfur dioxide at −70 °C in dichlorodifluoromethane.
Wilhelm Karl Klemm was an inorganic and physical chemist. Klemm did extensive work on intermetallic compounds, rare earth metals, transition elements and compounds involving oxygen and fluorine. He and Heinrich Bommer were the first to isolate elemental erbium (1934) and ytterbium (1936). Klemm refined Eduard Zintl's ideas about the structure of intermetallic compounds and their connections to develop the Zintl-Klemm concept.

Brigitte Sarry was a German chemist and a professor at Technische Universität Berlin.
Egon Gustaf Martin Wiberg was a German chemist and professor for inorganic chemistry at the Ludwig Maximilian University of Munich.

Octasulfur monoxide is an inorganic compound with a chemical formula S8O, discovered in 1972. It is a type of sulfur oxide.
Walter Rüdorff was a German chemist known for his research on clathrates of graphite and ternary oxides.

Beryllocene is an organoberyllium compound with the chemical formula Be(C5H5)2. It was first prepared in 1959. The colorless substance can be crystallized from petroleum ether in the form of white needles at −60 °C and decomposes quickly upon contact with atmospheric oxygen and water.














