
Arabidopsis thaliana, the thale cress, mouse-ear cress or arabidopsis, is a small plant from the mustard family (Brassicaceae), native to Eurasia and Africa. Commonly found along the shoulders of roads and in disturbed land, it is generally considered a weed.

Plant hormones are signal molecules, produced within plants, that occur in extremely low concentrations. Plant hormones control all aspects of plant growth and development, including embryogenesis, the regulation of organ size, pathogen defense, stress tolerance and reproductive development. Unlike in animals each plant cell is capable of producing hormones. Went and Thimann coined the term "phytohormone" and used it in the title of their 1937 book.

A pollen tube is a tubular structure produced by the male gametophyte of seed plants when it germinates. Pollen tube elongation is an integral stage in the plant life cycle. The pollen tube acts as a conduit to transport the male gamete cells from the pollen grain—either from the stigma to the ovules at the base of the pistil or directly through ovule tissue in some gymnosperms. In maize, this single cell can grow longer than 12 inches (30 cm) to traverse the length of the pistil.
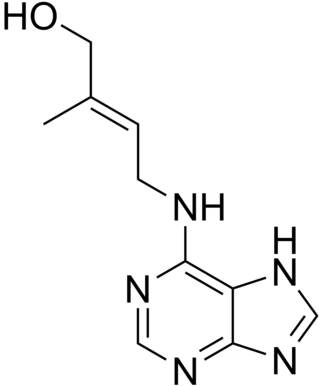
Cytokinins (CK) are a class of plant hormones that promote cell division, or cytokinesis, in plant roots and shoots. They are involved primarily in cell growth and differentiation, but also affect apical dominance, axillary bud growth, and leaf senescence.
Gibberellins (GAs) are plant hormones that regulate various developmental processes, including stem elongation, germination, dormancy, flowering, flower development, and leaf and fruit senescence. GAs are one of the longest-known classes of plant hormone. It is thought that the selective breeding of crop strains that were deficient in GA synthesis was one of the key drivers of the "green revolution" in the 1960s, a revolution that is credited to have saved over a billion lives worldwide.

Abscisic acid is a plant hormone. ABA functions in many plant developmental processes, including seed and bud dormancy, the control of organ size and stomatal closure. It is especially important for plants in the response to environmental stresses, including drought, soil salinity, cold tolerance, freezing tolerance, heat stress and heavy metal ion tolerance.

Plant senescence is the process of aging in plants. Plants have both stress-induced and age-related developmental aging. Chlorophyll degradation during leaf senescence reveals the carotenoids, such as anthocyanin and xanthophylls, which are the cause of autumn leaf color in deciduous trees. Leaf senescence has the important function of recycling nutrients, mostly nitrogen, to growing and storage organs of the plant. Unlike animals, plants continually form new organs and older organs undergo a highly regulated senescence program to maximize nutrient export.
In developmental biology, photomorphogenesis is light-mediated development, where plant growth patterns respond to the light spectrum. This is a completely separate process from photosynthesis where light is used as a source of energy. Phytochromes, cryptochromes, and phototropins are photochromic sensory receptors that restrict the photomorphogenic effect of light to the UV-A, UV-B, blue, and red portions of the electromagnetic spectrum.
Shade avoidance is a set of responses that plants display when they are subjected to the shade of another plant. It often includes elongation, altered flowering time, increased apical dominance and altered partitioning of resources. This set of responses is collectively called the shade-avoidance syndrome (SAS).

A parasitic plant is a plant that derives some or all of its nutritional requirements from another living plant. They make up about 1% of angiosperms and are found in almost every biome. All parasitic plants develop a specialized organ called the haustorium, which penetrates the host plant, connecting them to the host vasculature – either the xylem, phloem, or both. For example, plants like Striga or Rhinanthus connect only to the xylem, via xylem bridges (xylem-feeding). Alternately, plants like Cuscuta and some members of Orobanche connect to both the xylem and phloem of the host. This provides them with the ability to extract resources from the host. These resources can include water, nitrogen, carbon and/or sugars. Parasitic plants are classified depending on the location where the parasitic plant latches onto the host, the amount of nutrients it requires, and their photosynthetic capability. Some parasitic plants can locate their host plants by detecting volatile chemicals in the air or soil given off by host shoots or roots, respectively. About 4,500 species of parasitic plants in approximately 20 families of flowering plants are known.
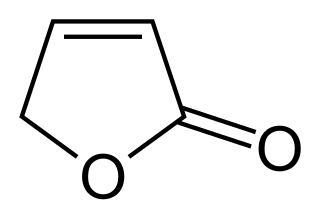
Butenolides are a class of lactones with a four-carbon heterocyclic ring structure. They are sometimes considered oxidized derivatives of furan. The simplest butenolide is 2-furanone, which is a common component of larger natural products and is sometimes referred to as simply "butenolide". A common biochemically important butenolide is ascorbic acid. Butenolide derivatives known as karrikins are produced by some plants on exposure to high temperatures due to brush fires. In particular, 3-methyl-2H-furo[2,3-c]pyran-2-one was found to trigger seed germination in plants whose reproduction is fire-dependent.

Brassica tournefortii is a species of plant known by the common names Asian mustard, pale cabbage, African mustard, and Sahara mustard, and is well known as an invasive species, especially in California.

Karrikins are a group of plant growth regulators found in the smoke of burning plant material. Karrikins help stimulate seed germination and plant development because they mimic a signaling hormone known as strigolactone. Strigolactones are hormones that help increase growth of symbiotic arbuscular mycorrhizal fungi in the soil, which enhances plant growth and leads to an increase in plant branching.

In biology, phototropism is the growth of an organism in response to a light stimulus. Phototropism is most often observed in plants, but can also occur in other organisms such as fungi. The cells on the plant that are farthest from the light contain a hormone called auxin that reacts when phototropism occurs. This causes the plant to have elongated cells on the furthest side from the light. Phototropism is one of the many plant tropisms, or movements, which respond to external stimuli. Growth towards a light source is called positive phototropism, while growth away from light is called negative phototropism. Negative phototropism is not to be confused with skototropism, which is defined as the growth towards darkness, whereas negative phototropism can refer to either the growth away from a light source or towards the darkness. Most plant shoots exhibit positive phototropism, and rearrange their chloroplasts in the leaves to maximize photosynthetic energy and promote growth. Some vine shoot tips exhibit negative phototropism, which allows them to grow towards dark, solid objects and climb them. The combination of phototropism and gravitropism allow plants to grow in the correct direction.

Ian Alexander Graham is a professor of Biochemical Genetics in the Centre for Novel Agricultural Products (CNAP) at the University of York.

Strigolactones are a group of chemical compounds produced by roots of plants. Due to their mechanism of action, these molecules have been classified as plant hormones or phytohormones. So far, strigolactones have been identified to be responsible for three different physiological processes: First, they promote the germination of parasitic organisms that grow in the host plant's roots, such as Strigalutea and other plants of the genus Striga. Second, strigolactones are fundamental for the recognition of the plant by symbiotic fungi, especially arbuscular mycorrhizal fungi, because they establish a mutualistic association with these plants, and provide phosphate and other soil nutrients. Third, strigolactones have been identified as branching inhibition hormones in plants; when present, these compounds prevent excess bud growing in stem terminals, stopping the branching mechanism in plants.

Ethylene (CH
2=CH
2) is an unsaturated hydrocarbon gas (alkene) acting as a naturally occurring plant hormone. It is the simplest alkene gas and is the first gas known to act as hormone. It acts at trace levels throughout the life of the plant by stimulating or regulating the ripening of fruit, the opening of flowers, the abscission (or shedding) of leaves and, in aquatic and semi-aquatic species, promoting the 'escape' from submergence by means of rapid elongation of stems or leaves. This escape response is particularly important in rice farming. Commercial fruit-ripening rooms use "catalytic generators" to make ethylene gas from a liquid supply of ethanol. Typically, a gassing level of 500 to 2,000 ppm is used, for 24 to 48 hours. Care must be taken to control carbon dioxide levels in ripening rooms when gassing, as high temperature ripening (20 °C; 68 °F) has been seen to produce CO2 levels of 10% in 24 hours.
June Nasrallah is Barbara McClintock Professor in the Plant Biology Section of the School of Integrative Plant Science at Cornell University. Her research focuses on plant reproductive biology and the cell-cell interactions that underlie self-incompatibility in plants belonging to the mustard (Brassicaceae) family. She was elected to the US National Academy of Sciences in 2003 for this work and her contributions generally to our understanding of receptor-based signaling in plants.
Jen Sheen is a biologist at Massachusetts General Hospital and Harvard Medical School who is known for her work on plant signaling networks. She is an elected member of the American Association for the Advancement of Science.
Christoph Benning is a German–American plant biologist. He is an MSU Foundation Professor and University Distinguished Professor at Michigan State University. Benning's research into lipid metabolism in plants, algae and photosynthetic bacteria, led him to be named Editor-in-Chief of The Plant Journal in October 2008.














