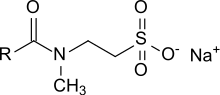
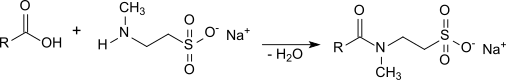In organic chemistry, an amide, also known as an organic amide or a carboxamide, is a compound with the general formula R−C(=O)−NR′R″, where R, R', and R″ represent any group, typically organyl groups or hydrogen atoms. The amide group is called a peptide bond when it is part of the main chain of a protein, and an isopeptide bond when it occurs in a side chain, as in asparagine and glutamine. It can be viewed as a derivative of a carboxylic acid with the hydroxyl group replaced by an amine group ; or, equivalently, an acyl (alkanoyl) group joined to an amine group.

In organic chemistry, a carboxylic acid is an organic acid that contains a carboxyl group attached to an R-group. The general formula of a carboxylic acid is often written as R−COOH or R−CO2H, sometimes as R−C(O)OH with R referring to an organyl group, or hydrogen, or other groups. Carboxylic acids occur widely. Important examples include the amino acids and fatty acids. Deprotonation of a carboxylic acid gives a carboxylate anion.

A detergent is a surfactant or a mixture of surfactants with cleansing properties when in dilute solutions. There are a large variety of detergents, a common family being the alkylbenzene sulfonates, which are soap-like compounds that are more soluble in hard water, because the polar sulfonate is less likely than the polar carboxylate to bind to calcium and other ions found in hard water.

Surfactants are chemical compounds that decrease the surface tension or interfacial tension between two liquids, a liquid and a gas, or a liquid and a solid. The word "surfactant" is a blend of surface-active agent, coined c. 1950. As they consist of a water-repellent and a water-attracting part, they enable water and oil to mix; they can form foam and facilitate the detachment of dirt.
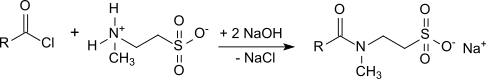
In organic chemistry, an acyl chloride is an organic compound with the functional group −C(=O)Cl. Their formula is usually written R−COCl, where R is a side chain. They are reactive derivatives of carboxylic acids. A specific example of an acyl chloride is acetyl chloride, CH3COCl. Acyl chlorides are the most important subset of acyl halides.
In organic chemistry, ethoxylation is a chemical reaction in which ethylene oxide adds to a substrate. It is the most widely practiced alkoxylation, which involves the addition of epoxides to substrates.
A fabric softener or fabric conditioner is a conditioner that is applied to laundry after it has been washed in a washing machine. A similar, more dilute preparation meant to be applied to dry fabric is known as a wrinkle releaser.

Laundry detergent is a type of detergent used for cleaning dirty laundry (clothes). Laundry detergent is manufactured in powder and liquid form.
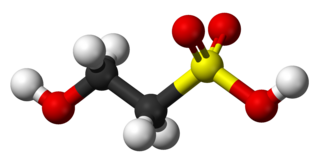
Isethionic acid is an organosulfur compound containing an alkylsulfonic acid located beta to a hydroxy group. Its discovery is generally attributed to Heinrich Gustav Magnus, who prepared it by the action of solid sulfur trioxide on ethanol in 1833. It is a white water-soluble solid used in the manufacture of certain surfactants and in the industrial production of taurine. It is most commonly available in the form of its sodium salt.
Nucleophilic acyl substitution (SNAcyl) describes a class of substitution reactions involving nucleophiles and acyl compounds. In this type of reaction, a nucleophile – such as an alcohol, amine, or enolate – displaces the leaving group of an acyl derivative – such as an acid halide, anhydride, or ester. The resulting product is a carbonyl-containing compound in which the nucleophile has taken the place of the leaving group present in the original acyl derivative. Because acyl derivatives react with a wide variety of nucleophiles, and because the product can depend on the particular type of acyl derivative and nucleophile involved, nucleophilic acyl substitution reactions can be used to synthesize a variety of different products.

The Ojima lactam is an organic compound of some importance in the commercial production of Taxol. This lactam was first synthesized by Iwao Ojima. The organic synthesis is an illustration of asymmetric synthesis via a chiral auxiliary.

Cleaning agents or hard-surface cleaners are substances used to remove dirt, including dust, stains, foul odors, and clutter on surfaces. Purposes of cleaning agents include health, beauty, removing offensive odor, and avoiding the spread of dirt and contaminants to oneself and others. Some cleaning agents can kill bacteria and clean at the same time. Others, called degreasers, contain organic solvents to help dissolve oils and fats.
Sodium lauroyl sarcosinate (INCI), also known as sarcosyl, is an anionic surfactant derived from sarcosine used as a foaming and cleansing agent in shampoo, shaving foam, toothpaste, and foam wash products.
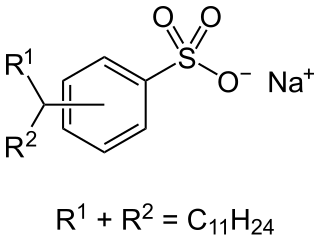
Alkylbenzene sulfonates are a class of anionic surfactants, consisting of a hydrophilic sulfonate head-group and a hydrophobic alkylbenzene tail-group. Along with sodium laureth sulfate, they are one of the oldest and most widely used synthetic detergents and may be found in numerous personal-care products and household-care products . They were introduced in the 1930s in the form of branched alkylbenzene sulfonates (BAS). However following environmental concerns these were replaced with linear alkylbenzene sulfonates (LAS) during the 1960s. Since then production has increased significantly from about one million tons in 1980, to around 3.5 million tons in 2016, making them most produced anionic surfactant after soaps.

Sodium 2-hydroxyethyl sulfonate is the sodium salt of 2-hydroxyethane sulfonic acid, it is used as a hydrophilic head group in washing-active surfactants, known as isethionates (acyloxyethanesulfonates) due to its strong polarity and resistance to multivalent ions. It is being studied as a high production volume chemical in the "High Production Volume (HPV) Chemical Challenge Program" of the US Environmental Protection Ministry EPA.
Sodium salts are salts composed of a sodium cation and the conjugate base anion of some inorganic or organic acids. They can be formed by the neutralization of such acids with sodium hydroxide.
N-Oleoylsarcosine (Sarkosyl O) is an amphiphilic oleic acid derivative having a sarcosine head group (N-methylglycine) which is used as a water-in-oil emulsifier and corrosion inhibitor.

N,N,N′,N′-Tetramethylformamidinium chloride is the simplest representative of quaternary formamidinium cations of the general formula [R2N−CH=NR2]+ with a chloride as a counterion in which all hydrogen atoms of the protonated formamidine [HC(=NH2)NH2]+ are replaced by methyl groups.
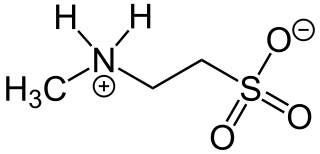
N-Methyltaurine is an aminosulfonic acid which is present as a zwitterion in the crystalline state and in polar solvents. In contrast to the widespread taurine, N-methyltaurine has been found in nature only in red algae, where it is formed by methylation of taurine. It is suitable for esterification with long-chain carboxylic acids to taurides (acylaminoethansulfonaten) because of its high polarity and the relatively good solubility of its alkaline earth metal salts, which are also used as mild anionic surfactants.
Isethionates are esters of long-chain aliphatic carboxylic acids (C8 – C18) with isethionic acid (2-hydroxyethanesulfonic acid) or salts thereof, such as ammonium isethionate or sodium isethionate. They are also referred to as acyl isethionates or acyloxyethanesulfonates.