Related Research Articles
Rheology is the study of the flow of matter,primarily in a fluid state,but also as "soft solids" or solids under conditions in which they respond with plastic flow rather than deforming elastically in response to an applied force. Rheology is a branch of physics,and it is the science that deals with the deformation and flow of materials,both solids and liquids.
Metallic hydrogen is a phase of hydrogen in which it behaves like an electrical conductor. This phase was predicted in 1935 on theoretical grounds by Eugene Wigner and Hillard Bell Huntington.
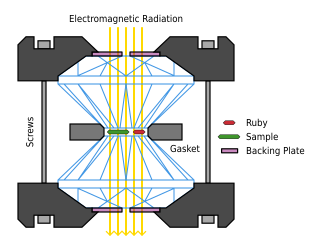
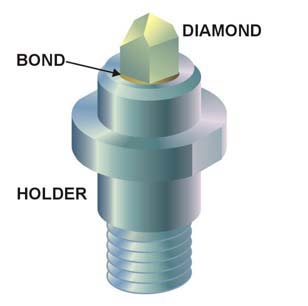
A diamond anvil cell (DAC) is a high-pressure device used in geology,engineering,and materials science experiments. It enables the compression of a small (sub-millimeter-sized) piece of material to extreme pressures,typically up to around 100–200 gigapascals,although it is possible to achieve pressures up to 770 gigapascals.
A superhard material is a material with a hardness value exceeding 40 gigapascals (GPa) when measured by the Vickers hardness test. They are virtually incompressible solids with high electron density and high bond covalency. As a result of their unique properties,these materials are of great interest in many industrial areas including,but not limited to,abrasives,polishing and cutting tools,disc brakes,and wear-resistant and protective coatings.

Diamond is the allotrope of carbon in which the carbon atoms are arranged in the specific type of cubic lattice called diamond cubic. It is a crystal that is transparent to opaque and which is generally isotropic. Diamond is the hardest naturally occurring material known. Yet,due to important structural brittleness,bulk diamond's toughness is only fair to good. The precise tensile strength of bulk diamond is little known;however,compressive strength up to 60 GPa has been observed,and it could be as high as 90–100 GPa in the form of micro/nanometer-sized wires or needles,with a corresponding maximum tensile elastic strain in excess of 9%. The anisotropy of diamond hardness is carefully considered during diamond cutting. Diamond has a high refractive index (2.417) and moderate dispersion (0.044) properties that give cut diamonds their brilliance. Scientists classify diamonds into four main types according to the nature of crystallographic defects present. Trace impurities substitutionally replacing carbon atoms in a diamond's crystal structure,and in some cases structural defects,are responsible for the wide range of colors seen in diamond. Most diamonds are electrical insulators and extremely efficient thermal conductors. Unlike many other minerals,the specific gravity of diamond crystals (3.52) has rather small variation from diamond to diamond.
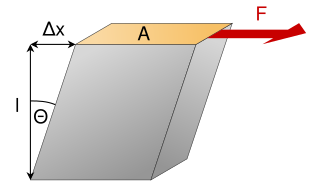
In materials science,shear modulus or modulus of rigidity,denoted by G,or sometimes S or μ,is a measure of the elastic shear stiffness of a material and is defined as the ratio of shear stress to the shear strain:
In materials science,hardness is a measure of the resistance to localized plastic deformation,such as an indentation or a scratch (linear),induced mechanically either by pressing or abrasion. In general,different materials differ in their hardness;for example hard metals such as titanium and beryllium are harder than soft metals such as sodium and metallic tin,or wood and common plastics. Macroscopic hardness is generally characterized by strong intermolecular bonds,but the behavior of solid materials under force is complex;therefore,hardness can be measured in different ways,such as scratch hardness,indentation hardness,and rebound hardness. Hardness is dependent on ductility,elastic stiffness,plasticity,strain,strength,toughness,viscoelasticity,and viscosity. Common examples of hard matter are ceramics,concrete,certain metals,and superhard materials,which can be contrasted with soft matter.

A shear band is a narrow zone of intense shearing strain,usually of plastic nature,developing during severe deformation of ductile materials. As an example,a soil specimen is shown in Fig. 1,after an axialsymmetric compression test. Initially the sample was cylindrical in shape and,since symmetry was tried to be preserved during the test,the cylindrical shape was maintained for a while during the test and the deformation was homogeneous,but at extreme loading two X-shaped shear bands had formed and the subsequent deformation was strongly localized.
Pseudoelasticity,sometimes called superelasticity,is an elastic (reversible) response to an applied stress,caused by a phase transformation between the austenitic and martensitic phases of a crystal. It is exhibited in shape-memory alloys.
Natalia Dubrovinskaia is a Swedish geologist of Russian origin.
In geology,a deformation mechanism is a process occurring at a microscopic scale that is responsible for changes in a material's internal structure,shape and volume. The process involves planar discontinuity and/or displacement of atoms from their original position within a crystal lattice structure. These small changes are preserved in various microstructures of materials such as rocks,metals and plastics,and can be studied in depth using optical or digital microscopy.

The mechanical properties of carbon nanotubes reveal them as one of the strongest materials in nature. Carbon nanotubes (CNTs) are long hollow cylinders of graphene. Although graphene sheets have 2D symmetry,carbon nanotubes by geometry have different properties in axial and radial directions. It has been shown that CNTs are very strong in the axial direction. Young's modulus on the order of 270 - 950 GPa and tensile strength of 11 - 63 GPa were obtained.

Boron can be prepared in several crystalline and amorphous forms. Well known crystalline forms are α-rhombohedral (α-R),β-rhombohedral (β-R),and β-tetragonal (β-T). In special circumstances,boron can also be synthesized in the form of its α-tetragonal (α-T) and γ-orthorhombic (γ) allotropes. Two amorphous forms,one a finely divided powder and the other a glassy solid,are also known. Although at least 14 more allotropes have been reported,these other forms are based on tenuous evidence or have not been experimentally confirmed,or are thought to represent mixed allotropes,or boron frameworks stabilized by impurities. Whereas the β-rhombohedral phase is the most stable and the others are metastable,the transformation rate is negligible at room temperature,and thus all five phases can exist at ambient conditions. Amorphous powder boron and polycrystalline β-rhombohedral boron are the most common forms. The latter allotrope is a very hard grey material,about ten percent lighter than aluminium and with a melting point (2080 °C) several hundred degrees higher than that of steel.
Superdense carbon allotropes are proposed configurations of carbon atoms that result in a stable material with a higher density than diamond.
Crack closure is a phenomenon in fatigue loading,where the opposing faces of a crack remain in contact even with an external load acting on the material. As the load is increased,a critical value will be reached at which time the crack becomes open. Crack closure occurs from the presence of material propping open the crack faces and can arise from many sources including plastic deformation or phase transformation during crack propagation,corrosion of crack surfaces,presence of fluids in the crack,or roughness at cracked surfaces.
Iron tetraboride (FeB4) is a superhard superconductor (Tc <3K) consisting of iron and boron. Iron tetraboride does not occur in nature and can be created synthetically. Its molecular structure was predicted using computer models.
In materials science,toughening refers to the process of making a material more resistant to the propagation of cracks. When a crack propagates,the associated irreversible work in different materials classes is different. Thus,the most effective toughening mechanisms differ among different materials classes. The crack tip plasticity is important in toughening of metals and long-chain polymers. Ceramics have limited crack tip plasticity and primarily rely on different toughening mechanisms.
Dislocation avalanches are rapid discrete events during plastic deformation,in which defects are reorganized collectively. This intermittent flow behavior has been observed in microcrystals,whereas macroscopic plasticity appears as a smooth process. Intermittent plastic flow has been observed in several different systems. In AlMg Alloys,interaction between solute and dislocations can cause sudden jump during dynamic strain aging. In metallic glass,it can be observed via shear banding with stress localization;and single crystal plasticity,it shows up as slip burst. However,analysis of the events with orders-magnitude difference in sizes with different crystallographic structure reveals power-law scaling between the number of events and their magnitude,or scale-free flow.
In continuum mechanics,ratcheting,or ratchetting,also known as cyclic creep,is a behavior in which plastic deformation accumulates due to cyclic mechanical or thermal stress.
Metallization pressure is the pressure required for a non-metallic chemical element to become a metal. Every material is predicted to turn into a metal if the pressure is high enough,and temperature low enough. Some of these pressures are beyond the reach of diamond anvil cells,and are thus theoretical predictions. Neon has the highest metallization pressure for any element.
References
- 1 2 3 "Valery Levitas – Distinguished Professor [AER E] – External, Faculty – Profile". Expert Finder.
- 1 2 "Iowa State University (via Public) / Iowa State materials researcher elected to European Academy of Sciences and Arts". www.publicnow.com.
- ↑ "Valery Levitas". scholar.google.com.
- ↑ "Large Deformation of Materials with Complex Rheological Properties at Normal and High Pressure".
- 1 2 "Khan Plasticity Award – Recipients". sites.google.com.
- 1 2 Levitas, Valery I. (May 1, 2021). "Phase transformations, fracture, and other structural changes in inelastic materials". International Journal of Plasticity. 140: 102914. doi:10.1016/j.ijplas.2020.102914 – via ScienceDirect.
- 1 2 Levitas, Valery I. (March 14, 2019). "High-Pressure Phase Transformations under Severe Plastic Deformation by Torsion in Rotational Anvils". Materials Transactions. 60 (7): 1294–1301. doi:10.2320/matertrans.MF201923 – via J-Stage.
- ↑ "High-pressure mechanochemistry: Conceptual multiscale theory and interpretation of experiments" (PDF).
- ↑ Zarkevich, Nikolai A.; Chen, Hao; Levitas, Valery I.; Johnson, Duane D. (October 17, 2018). "Lattice Instability during Solid-Solid Structural Transformations under a General Applied Stress Tensor: Example of $\mathrm{Si}\text{ }\text{ }\mathrm{I}\ensuremath{\rightarrow}\mathrm{Si}\text{ }\text{ }\mathrm{II}$ with Metallization". Physical Review Letters. 121 (16): 165701. doi:10.1103/PhysRevLett.121.165701 – via APS.
- 1 2 Chen, Hao; Levitas, Valery I.; Xiong, Liming (October 1, 2019). "Amorphization induced by 60° shuffle dislocation pileup against different grain boundaries in silicon bicrystal under shear". Acta Materialia. 179: 287–295. Bibcode:2019AcMat.179..287C. doi:10.1016/j.actamat.2019.08.023 – via NASA ADS.
- 1 2 3 Levitas, Valery I.; Javanbakht, Mahdi (December 9, 2013). "Phase transformations in nanograin materials under high pressure and plastic shear: nanoscale mechanisms". Nanoscale. 6 (1): 162–166. Bibcode:2013Nanos...6..162L. doi:10.1039/C3NR05044K. PMID 24213214 – via pubs.rsc.org.
- 1 2 Levitas, Valery I.; Esfahani, S. Ehsan; Ghamarian, Iman (November 15, 2018). "Scale-Free Modeling of Coupled Evolution of Discrete Dislocation Bands and Multivariant Martensitic Microstructure". Physical Review Letters. 121 (20): 205701. Bibcode:2018PhRvL.121t5701L. doi:10.1103/PhysRevLett.121.205701. PMID 30500235 – via APS.
- ↑ Levitas, Valery I.; Zarechnyy, Oleg M. (November 23, 2010). "Modeling and simulation of strain-induced phase transformations under compression and torsion in a rotational diamond anvil cell". Physical Review B. 82 (17): 174124. Bibcode:2010PhRvB..82q4124L. doi:10.1103/PhysRevB.82.174124 – via APS.
- ↑ Levitas, Valery I.; Kamrani, Mehdi; Feng, Biao (October 1, 2019). "Tensorial stress-strain fields and large elastoplasticity as well as friction in diamond anvil cell up to 400 GPa". npj Computational Mathematics. 5: 94. Bibcode:2019npjCM...5...94L. doi:10.1038/s41524-019-0234-8 – via NASA ADS.
- ↑ Levitas, Valery I.; Dhar, Achyut; Pandey, K. K. (September 23, 2023). "Tensorial stress-plastic strain fields in α - ω Zr mixture, transformation kinetics, and friction in diamond-anvil cell". Nature Communications. 14 (1): 5955. arXiv: 2212.13000 . Bibcode:2023NatCo..14.5955L. doi:10.1038/s41467-023-41680-1. PMC 10517986 . PMID 37741842.
- ↑ Levitas, Valery I.; Shvedov, Leonid K. (February 28, 2002). "Low-pressure phase transformation from rhombohedral to cubic BN: Experiment and theory". Physical Review B. 65 (10): 104109. Bibcode:2002PhRvB..65j4109L. doi:10.1103/PhysRevB.65.104109 – via APS.
- ↑ Gao, Yang; Ma, Yanzhang; An, Qi; Levitas, Valery; Zhang, Yanyan; Feng, Biao; Chaudhuri, Jharna; Goddard III, William A. (May 29, 2018). "Shear driven formation of nano-diamonds at sub-gigapascals and 300 K". Carbon. 146: 364. arXiv: 1805.11239 . Bibcode:2019Carbo.146..364G. doi:10.1016/j.carbon.2019.02.012 – via arXiv.org.
- ↑ Levitas, Valery I.; Ma, Yanzhang; Selvi, Emre; Wu, Jianzhe; Patten, John A. (February 29, 2012). "High-density amorphous phase of silicon carbide obtained under large plastic shear and high pressure". Physical Review B. 85 (5): 054114. Bibcode:2012PhRvB..85e4114L. doi:10.1103/PhysRevB.85.054114 – via APS.
- ↑ Levitas, Valery I. (October 22, 2022). "Resolving puzzles of the phase-transformation-based mechanism of the strong deep-focus earthquake". Nature Communications. 13 (1): 6291. arXiv: 2110.10862 . Bibcode:2022NatCo..13.6291L. doi:10.1038/s41467-022-33802-y. PMC 9588062 . PMID 36273002.
- ↑ Feng, Biao; Levitas, Valery I. (April 21, 2017). "Pressure Self-focusing Effect and Novel Methods for Increasing the Maximum Pressure in Traditional and Rotational Diamond Anvil Cells". Scientific Reports. 7 (1): 45461. Bibcode:2017NatSR...745461F. doi:10.1038/srep45461 – via www.nature.com.
- ↑ Levitas, Valery I.; Henson, Bryan F.; Smilowitz, Laura B.; Asay, Blaine W. (June 11, 2004). "Solid-solid phase transformation via virtual melting significantly below the melting temperature". Physical Review Letters. 92 (23): 235702. Bibcode:2004PhRvL..92w5702L. doi:10.1103/PhysRevLett.92.235702. PMID 15245170 – via PubMed.
- ↑ Levitas, Valery I. (November 28, 2013). "Mechanochemical mechanism for reaction of aluminium nano- and micrometre-scale particles". Philosophical Transactions. Series A, Mathematical, Physical, and Engineering Sciences. 371 (2003): 20120215. Bibcode:2013RSPTA.37120215L. doi:10.1098/rsta.2012.0215. PMID 24146008 – via PubMed.
- ↑ Novikov, N. B.; Shvedov, L. K.; Krivosheya, Yu. N.; Levitas, V. I. (January 1, 2015). "New automated shear cell with diamond anvils for in situ studies of materials using X-ray diffraction". Journal of Superhard Materials. 37 (1): 1–7. doi:10.3103/S1063457615010013 – via Springer Link.
- ↑ "Richard-von-Mises Prize – GAMM e.V." www.gamm.org.
- ↑ "ASME Fellows List" (PDF).
- ↑ "International Journal of Plasticity | Structural Changes and Plasticity in Materials – In Honor of Professor Valery Levitas | ScienceDirect.com by Elsevier". www.sciencedirect.com.
- ↑ "Short bio of Professor Valery I. Levitas" (PDF).
- ↑ "Members | European Academy of Sciences and Arts". members.euro-acad.eu.
- ↑ "International Association of Advanced Materials – IAAM | Non-Profit Organization". IAAM. January 16, 2019.