Relation of atoms in a molecule
For example, the molecule 2,3-dibromobutane carries two vicinal bromine atoms and 1,3-dibromobutane does not. Mostly, the use of the term vicinal is restricted to two identical functional groups.
Likewise in a gem-dibromide the prefix gem, an abbreviation of geminal , signals that both bromine atoms are bonded to the same carbon atom (i.e., in a 1,1-relationship). For example, 1,1-dibromobutane is geminal. While comparatively less common, the term hominal has been suggested as a descriptor for groups in a 1,3-relationship. [1]
| Comparison of geminal with vicinal and isolated substitution patterns. | ||||
| Alkane | Geminal | vicinal | isolated | |
| Methane |  | 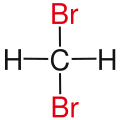 | not existing | not existing |
| Ethane |  |  | 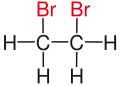 | not existing |
| Propane |  |  |  |  |
| Substituents on selected dibromoalkanes labeled red. | ||||
Like other descriptors, such as syn, anti, exo or endo, the description vicinal helps explain how different parts of a molecule are related to each other either structurally or spatially. The vicinal adjective is sometimes restricted to those molecules with two identical functional groups. The use of the term can also be extended to substituents on aromatic rings.
