
(2S,4R)-4-Hydroxyproline, or L-hydroxyproline (C5H9O3N), is an amino acid, abbreviated as Hyp or O, e.g., in Protein Data Bank.

Lamins, also known as nuclear lamins are fibrous proteins in type V intermediate filaments, providing structural function and transcriptional regulation in the cell nucleus. Nuclear lamins interact with inner nuclear membrane proteins to form the nuclear lamina on the interior of the nuclear envelope. Lamins have elastic and mechanosensitive properties, and can alter gene regulation in a feedback response to mechanical cues. Lamins are present in all animals but are not found in microorganisms, plants or fungi. Lamin proteins are involved in the disassembling and reforming of the nuclear envelope during mitosis, the positioning of nuclear pores, and programmed cell death. Mutations in lamin genes can result in several genetic laminopathies, which may be life-threatening.
A nuclear localization signalorsequence (NLS) is an amino acid sequence that 'tags' a protein for import into the cell nucleus by nuclear transport. Typically, this signal consists of one or more short sequences of positively charged lysines or arginines exposed on the protein surface. Different nuclear localized proteins may share the same NLS. An NLS has the opposite function of a nuclear export signal (NES), which targets proteins out of the nucleus.

Indole-3-acetic acid is the most common naturally occurring plant hormone of the auxin class. It is the best known of the auxins, and has been the subject of extensive studies by plant physiologists. IAA is a derivative of indole, containing a carboxymethyl substituent. It is a colorless solid that is soluble in polar organic solvents.

Ran also known as GTP-binding nuclear protein Ran is a protein that in humans is encoded by the RAN gene. Ran is a small 25 kDa protein that is involved in transport into and out of the cell nucleus during interphase and also involved in mitosis. It is a member of the Ras superfamily.
The gene-for-gene relationship was discovered by Harold Henry Flor who was working with rust of flax. Flor showed that the inheritance of both resistance in the host and parasite ability to cause disease is controlled by pairs of matching genes. One is a plant gene called the resistance (R) gene. The other is a parasite gene called the avirulence (Avr) gene. Plants producing a specific R gene product are resistant towards a pathogen that produces the corresponding Avr gene product. Gene-for-gene relationships are a widespread and very important aspect of plant disease resistance. Another example can be seen with Lactuca serriola versus Bremia lactucae.
Expansin refers to a family of closely related nonenzymatic proteins found in the plant cell wall, with important roles in plant cell growth, fruit softening, abscission, emergence of root hairs, pollen tube invasion of the stigma and style, meristem function, and other developmental processes where cell wall loosening occurs. Expansins were originally discovered as mediators of acid growth, which refers to the widespread characteristic of growing plant cell walls to expand faster at low (acidic) pH than at neutral pH. Expansins are thus linked to auxin action. They are also linked to cell enlargement and cell wall changes induced by other plant hormones such as gibberellin, cytokinin, ethylene and brassinosteroids.

Involucrin is a protein component of human skin and in humans is encoded by the IVL gene. In binding the protein loricrin, involucrin contributes to the formation of a cell envelope that protects corneocytes in the skin.
A nuclear export signal (NES) is a short target peptide containing 4 hydrophobic residues in a protein that targets it for export from the cell nucleus to the cytoplasm through the nuclear pore complex using nuclear transport. It has the opposite effect of a nuclear localization signal, which targets a protein located in the cytoplasm for import to the nucleus. The NES is recognized and bound by exportins.
Arabinogalactan-proteins (AGPs) are highly glycosylated proteins (glycoproteins) found in the cell walls of plants. Each one consists of a protein with sugar molecules attached. They are members of the wider class of hydroxyproline (Hyp)-rich cell wall glycoproteins, a large and diverse group of glycosylated wall proteins.
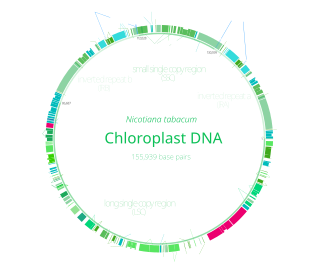
Chloroplast DNA (cpDNA) is the DNA located in chloroplasts, which are photosynthetic organelles located within the cells of some eukaryotic organisms. Chloroplasts, like other types of plastid, contain a genome separate from that in the cell nucleus. The existence of chloroplast DNA was identified biochemically in 1959, and confirmed by electron microscopy in 1962. The discoveries that the chloroplast contains ribosomes and performs protein synthesis revealed that the chloroplast is genetically semi-autonomous. The first complete chloroplast genome sequences were published in 1986, Nicotiana tabacum (tobacco) by Sugiura and colleagues and Marchantia polymorpha (liverwort) by Ozeki et al. Since then, hundreds of chloroplast DNAs from various species have been sequenced.
The N-end rule is a rule that governs the rate of protein degradation through recognition of the N-terminal residue of proteins. The rule states that the N-terminal amino acid of a protein determines its half-life. The rule applies to both eukaryotic and prokaryotic organisms, but with different strength, rules, and outcome. In eukaryotic cells, these N-terminal residues are recognized and targeted by ubiquitin ligases, mediating ubiquitination thereby marking the protein for degradation. The rule was initially discovered by Alexander Varshavsky and co-workers in 1986. However, only rough estimations of protein half-life can be deduced from this 'rule', as N-terminal amino acid modification can lead to variability and anomalies, whilst amino acid impact can also change from organism to organism. Other degradation signals, known as degrons, can also be found in sequence.
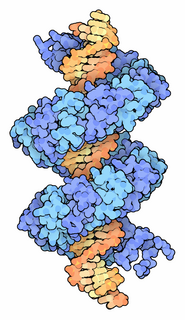
TALeffectors are proteins secreted by some β- and γ-proteobacteria. Most of these are Xanthomonads. Plant pathogenic Xanthomonas bacteria are especially known for TALEs, produced via their type III secretion system. These proteins can bind promoter sequences in the host plant and activate the expression of plant genes that aid bacterial infection. They recognize plant DNA sequences through a central repeat domain consisting of a variable number of ~34 amino acid repeats. There appears to be a one-to-one correspondence between the identity of two critical amino acids in each repeat and each DNA base in the target sequence. These proteins are interesting to researchers both for their role in disease of important crop species and the relative ease of retargeting them to bind new DNA sequences. Similar proteins can be found in the pathogenic bacterium Ralstonia solanacearum and Burkholderia rhizoxinica, as well as yet unidentified marine microorganisms. The term TALE-likes is used to refer to the putative protein family encompassing the TALEs and these related proteins.
RanGAP is a protein involved in the transport of other proteins from the cytosol to the nucleus in eukaryotic cells.
WH1 domain is an evolutionary conserved protein domain. Therefore, it has an important function.

UV-B resistance 8 (UVR8) also known as ultraviolet-B receptor UVR8 is an UV-B – sensing protein found in plants and possibly other sources. It is responsible for sensing ultraviolet light in the range 280-315 nm and initiating the plant stress response. It is most sensitive at 285nm, near the lower limit of UVB. UVR8 was first identified as a crucial mediator of a plant's response to UV-B in Arabidopsis thaliana containing a mutation in this protein. This plant was found to have a hypersensitivity to UV-B which damages DNA. UVR8 is thought to be a unique photoreceptor as it doesn't contain a prosthetic chromophore but its light-sensing ability is intrinsic to the molecule. Tryptophan (Trp) residue 285 has been suggested to act the UV-B sensor, while other Trp residues have been also seen to be involved although in-vivo data suggests that Trp285 and Trp233 are most important.

The WRKY domain is found in the WRKY transcription factor family, a class of transcription factors. The WRKY domain is found almost exclusively in plants although WRKY genes appear present in some diplomonads, social amoebae and other amoebozoa, and fungi incertae sedis. They appear absent in other non-plant species. WRKY transcription factors have been a significant area of plant research for the past 20 years. The WRKY DNA-binding domain recognizes the W-box (T)TGAC(C/T) cis-regulatory element.
In molecular biology, the X8 domain, is thought to play a role in targeting the plasmodesmata by providing it with structural support. The domain is able to do this since it contains signal sequences for a glycosylphosphatidylinositol (GPI) linkage to the extracellular face of the plasma membrane. This domain is involved in carbohydrate binding.
EARLY FLOWERING 3 (ELF3) is a plant-specific gene that encodes the hydroxyproline-rich glycoprotein and is required for the function of the circadian clock. ELF3 is one of the three components that make up the Evening Complex (EC) within the plant circadian clock, in which all three components reach peak gene expression and protein levels at dusk. ELF3 serves as a scaffold to bind EARLY FLOWERING 4 (ELF4) and LUX ARRHYTHMO (LUX), two other components of the EC, and functions to control photoperiod sensitivity in plants. ELF3 also plays an important role in temperature and light input within plants for circadian clock entrainment. Additionally, it plays roles in light and temperature signaling that are independent from its role in the EC.

The envelope (E) protein is the smallest and least well-characterized of the four major structural proteins found in coronavirus virions. It is an integral membrane protein less than 110 amino acid residues long; in SARS-CoV-2, the causative agent of Covid-19, the E protein is 75 residues long. Although it is not necessarily essential for viral replication, absence of the E protein may produce abnormally assembled viral capsids or reduced replication. E is a multifunctional protein and, in addition to its role as a structural protein in the viral capsid, it is thought to be involved in viral assembly, likely functions as a viroporin, and is involved in viral pathogenesis.









