

| Factor | Multiple | Item |
|---|---|---|
| 0 | 0 K | Absolute zero: free bodies are still, no interaction within or without a thermodynamic system |
| 10−30 | 1 qK | Particular speeds bound paths to exceed size and lifetime of the universe, i.e. the particles total path traveled (but not the distance from its place of origin) since the beginning of the universe is less than the size of the universe[ further explanation needed ] (see least-energy in orders of magnitude (energy)) |
| 10−18 | 1 aK | Macroscopic teleportation of matter can occur Hawking temperature of supermassive black holes |
| 10−15 | 1 fK | Atomic waves coherent over centimeters atomic particles decoherent over centimeters |
| 10−12 | 1 pK | 38 pK, lowest temperature ever produced, achieved through matter-wave lensing of rubidium Bose-Einstein condensates. [1] 450 pK, lowest temperature sodium Bose–Einstein condensate gas ever achieved in the laboratory, at MIT [2] |
| 10−9 | 1 nK | 50 nK, Fermi temperature of potassium-40 critical temperature of alkali Bose–Einstein condensates |
| 10−6 | 1 μK | Nuclear demagnetization Doppler-cooled refrigerants in laser cooling and magneto-optical traps |
| 10−3 | 1 mK | Radio excitations 1.7 mK, temperature record for helium-3/helium-4 dilution refrigeration, and the lowest temperature which may be sustained for arbitrarily long time with known techniques. 2.5 mK, Fermi melting point of helium-3 60 mK adiabatic demagnetization of paramagnetic molecules 300 mK in evaporative cooling of helium-3 700 mK, helium-3/helium-4 mixtures begin phase separation 950 mK, melting point of helium at 2.5 megapascals of pressure- All 118 elements are solid at or below this temperature. microwave excitations |
| 1 | 1 K | 1 K at the Boomerang Nebula, the coldest natural environment known 1.5 K, melting point of overbound helium 2.19 K, lambda point of overbound superfluid helium 2.725 K, cosmic microwave background 4.1 K, superconductivity point of mercury 4.22 K, boiling point of bound helium 5.19 K, critical temperature of helium 7.2 K, superconductivity point of lead 9.3 K, superconductivity point of niobium |
| 101 | 10 K | Fermi melting point of valence electrons for superconductivity 14.01 K, melting point of bound hydrogen 20.28 K, boiling point of bound hydrogen 33 K, critical temperature of hydrogen 44 K mean on Pluto 53 K mean of Neptune 63 K, melting point of bound nitrogen 68 K mean of Uranus 77.35 K, boiling point of bound nitrogen 90.19 K, boiling point of bound oxygen 92 K, superconductivity point of Y – Ba – Cu – oxide (YBCO) |
| 102 | 100 K | Infrared excitations 134 K, highest-temperature superconductor at ambient pressure, mercury barium calcium copper oxide 165 K, glass point of supercooled water 184.0 K (–89.2 °C), coldest air recorded on Earth 192 K, Debye temperature of ice 273.15 K (0 °C), melting point of bound water 273.16 K (0.01 °C), temperature of triple point of water ~293 K, room temperature 373.15 K (100 °C), boiling point of bound water at sea level 647 K, critical point of superheated water 737.5 K, mean on Venus See detailed list below |
| 103 | 1 kK | Visible light excitations 500–2200 K on brown dwarfs (photosphere) 1043 K Curie temperature of iron (point at which iron transitions from ferromagnetic to paramagnetic behavior and loses any permanent magnetism) 1170 K at wood fire 1300 K in lava flows, open flames 1500 K in basalt lava flows ~1670 K at blue candle flame 1811 K, melting point of iron (lower for steel) 1830 K in Bunsen burner flame 1900 K at the Space Shuttle orbiter hull in 8 km/s dive 2022 K, boiling point of lead 2074 K, surface temperature of the coolest star, 2MASS J0523-1403 Contents
Ultraviolet excitations |
| 104 | 10 kK | 10 kK on Sirius A 10–15 kK in mononitrogen recombination 15.5 kK, critical point of tungsten 25 kK, mean of the Universe 10,000 years after the Big Bang 26 kK on white dwarf Sirius B 28 kK in record cationic lightning over Earth 29 kK on surface of Alnitak (easternmost star of Orion's belt) 4–8–40–160 kK on white dwarfs 30–400 kK on a planetary nebula's asymptotic giant helium star 36 kK boundary between inner and outer core within Jupiter 37 kK in proton – electron reactions 38 kK on Eta Carinae 46 kK on Wolf–Rayet star R136a1 [10] 50 kK at protostar (core) 54.5 kK on ON2 III(f*) star LH64-16 [11] >200 kK on Butterfly Nebula ~300 kK at 17 meters from Little Boy's detonation Fermi boiling point of valence electrons X-ray excitations |
| 106 | 1 MK | 0.8 MK in solar wind gamma ray excitations 1 MK inside old neutron stars, brown dwarfs, and at gravital deuterium fusion range 1–3–10 MK above Sun (corona) 2.4 MK at T Tauri stars and gravital lithium-6 fusion range 2.5 MK at red dwarfs and gravital protium fusion range 10 MK at orange dwarfs and gravital helium-3 fusion range 15.6 MK at Sun's core 10–30–100 MK in stellar flares 20 MK in novae 23 MK, beryllium-7 fusion range 60 MK above Eta Carinae 85 MK (15 keV) in a magnetic confinement fusion plasma 200 MK at helium star and gravital helium-4 fusion range 230 MK, gravital carbon-12 fusion range 460 MK, gravital neon fusion – disproportionation range 5–530 MK in Tokamak Fusion Test Reactor's plasma 750 MK, gravital oxygen fusion range |
| 109 | 1 GK | 1 GK, everything 100 seconds after the Big Bang 1.3–1.7 GK, gravital silicon fusion range 3 GK in electron – positron reactions 10 GK in supernovae 10 GK, everything 1 second after the Big Bang 700 GK in quasars' accretion discs 740 GK, Hagedorn temperature or Fermi melting point of pions |
| 1012 | 1 TK | 0.1–1 TK at new neutron star 0.5–1.2 TK, Fermi melting point of hadrons into quark–gluon plasma 3–5 TK in proton – antiproton reactions 3.6 TK, temperature at which matter doubles in mass (compared to its mass at 0 K) due to relativistic effects 5.5 TK, highest man-made temperature in thermal equilibrium as of 2015 (quark–gluon plasma from LHC collisions) [12] 10 TK, 100 microseconds after the Big Bang 45–67 TK at collapsar of a gamma-ray burst 300–900 TK at proton – nickel conversions in the Tevatron's Main Injector[ clarification needed ] |
| 1015 | 1 PK | 0.3–2.2 PK at proton – antiproton collisions 2.8 PK within an electroweak star |
| 1018 | 1 EK | |
| 1021 | 1 ZK | |
| 1024 | 1 YK | 0.5–7 YK at ultra-high-energy cosmic ray collisions |
| 1027 | 1 RK | everything 10−35 seconds after the Big Bang |
| 1030 | 1 QK | Hagedorn temperature of strings |
| 1032 | 100 QK | 142 QK, Planck temperature |
| 1033 | 1000 QK | Theory of everything excitations[ citation needed ] |
| 10290 | 10260 QK | Landau pole of Quantum electrodynamics |
Most ordinary human activity takes place at temperatures of this order of magnitude. Circumstances where water naturally occurs in liquid form are shown in light grey.
| Kelvin | Degrees Celsius | Degrees Fahrenheit | Condition |
|---|---|---|---|
| 100 K | −173.15 °C | −279.67 °F | |
| 133 K | −140 °C | −220 °F | Mean on Saturn [13] |
| 133 K to 163 K | −140 to −110 °C | −220 to −160 °F | Typical temperature of a whole-body cryotherapy chamber [14] |
| 163 K | −110 °C | −166 °F | Mean on Jupiter [13] |
| 165 K | −108 °C | −163 °F | Glass point of supercooled water (Debatable) [15] |
| 175.4 K | −97.8 °C | −144 °F | Coldest luminance temperature recorded on Earth (measured remotely by satellite), in Antarctica [16] |
| 183.7 K | −89.5 °C | −129.1 °F | Freezing/melting point of isopropyl alcohol [17] |
| 183.9 K | −89.2 °C | −128.6 °F | Coldest officially recorded air temperature on Earth, at Vostok Station, Antarctica on 1983-07-21 01:45 UTC |
| 192 K | −81 °C | −114 °F | Debye temperature of ice |
| 193 to 203 K | −80 to −70 °C | −112 to −94 °F | Typical temperature of a ULT freezer |
| 194.6 K | −78.5 °C | −109.3 °F | Sublimation point of carbon dioxide (dry ice) |
| 203.55 K | −69.6 °C | −93.3 °F | Coldest officially recorded air temperature in the Northern Hemisphere at Klinck AWS, Greenland (Denmark) on 1991-12-22 [18] |
| 205.5 K | −67.7 °C | −89.9 °F | Coldest officially recorded air temperature on the Eurasian continent at Oymyakon, USSR on 1933-02-06 [19] |
| 210 K | −63 °C | −80 °F | Mean on Mars |
| 214.9 K | –58.3 °C | –72.9 °F | Coldest annual mean temperature on Earth, at Dome Argus, Antarctica [20] |
| 223.15 K | −50 °C | −58 °F | Mean on Earth during Snowball Earth [21] around 650 million years ago |
| 224.8 K | −48.4 °C | −55.0 °F | Coldest temperature that water can remain a liquid (see supercooling) |
| 225 K | −48 °C | −55 °F | Freezing/melting point of cottonseed oil [22] |
| 233.15 K | −40 °C | −40 °F | Intersecting point of the Celsius and Fahrenheit temperature scales Skin may freeze almost instantly at or below this temperature [23] |
| 234.3 K | −38.83 °C | −37.89 °F | Freezing/melting point of mercury |
| 240.4 K | −32.8 °C | −27.0 °F | Coldest air temperature recorded in South America, at Sarmiento, Argentina on 1907-06-01 [24] |
| 246 K | −27 °C | −17 °F | Approximate average yearly temperature on Mount Everest [25] |
| 249 K | –24 °C | –11 °F | Freezing/melting point of flax seed oil [22] |
| 249.3 K | –23.9 °C | –11.0 °F | Coldest air temperature recorded in Africa, at Ifrane, Morocco on 1935-02-11 [24] |
| 250 K | –23 °C | –9 °F | Coldest air temperature recorded in Australia, at Charlotte Pass, New South Wales, Australia on 1994-06-29 [24] |
| 255.37 K | –177⁄9 °C | 0 °F | Coldest brine-ice solution found by Daniel Gabriel Fahrenheit |
| 255 K | –18 °C | 0 °F | Freezing/melting point of almond oil [22] Typical temperature of a household freezer [26] |
| 256 K | –17 °C | 1 °F | Freezing/melting point of sunflower oil [22] |
| 256 K | –17 °C | 2 °F | Freezing/melting point of safflower oil [22] |
| 257 K | –16 °C | 3 °F | Freezing/melting point of soybean oil [22] |
| 262 K | −11 °C | 12 °F | Freezing/melting point of corn oil [22] |
| 263.15 K | –10 °C | 14 °F | Freezing/melting point of canola oil [22] Freezing/melting point of grape seed oil [22] |
| 265 K | –8 °C | 18 °F | White frost can form below this temperature (see frost) Freezing/melting point of hemp seed oil [22] |
| 265.8 K | –7.2 °C | 19 °F | Freezing/melting point of bromine |
| 267 K | –6 °C | 21 °F | Freezing/melting point of olive oil [22] Freezing/melting point of sesame oil [22] |
| 271.15 K | −2 °C | 28.4 °F | Average freezing/melting point of oceans, the salinity is around 3.47%. [27] [28] |
| 273.14 K | -0.01 °C | 31.98 °F | Maximum temperature of an object causing frostbite |
| 273.15 K | 0.00 °C | 32.00 °F | Freezing/melting point of fresh water (at 1 atm pressure) |
| 273.16 K | 0.01 °C | 32.02 °F | Triple point of fresh water |
| 276 K | 3 °C | 37 °F | Freezing/melting point of peanut oil [29] |
| 277 K | 3.85 °C | 39 °F | Typical temperature of a household refrigerator |
| 277.13 K | 3.98 °C | 39.16 °F | Water is at maximum density [30] |
| 279.8 K | 6.67 °C | 44 °F | Threshold of skin numbness if skin reaches this temperature |
| 283.2 K | 10 °C | 50 °F | Minimum temperature for most plant growth (see Growing degree-day) |
| 286.9 K | 12.7 °C | 54.9 °F | Coldest body temperature of a human that survived accidental hypothermia (a 2-year-old boy in Racławice, Poland, on November 30, 2014) [31] [32] |
| 287.6 K | 14.44 °C | 58 °F | Cold threshold of pain if skin reaches this temperature |
| 288 K | 15 °C | 59 °F | Mean on Earth |
| 291.6 K | 18.4 °C | 65.1 °F | Hottest temperature in Antarctica, recorded on 2020 February 6 at the Esperanza Base [33] |
| 294 K | 21 °C | 70 °F | Commonly defined value for room temperature |
| 296 K | 23 °C | 73 °F | Mean on Earth during the Paleocene–Eocene Thermal Maximum [34] about 55.8 million years ago |
| 297 K | 24 °C | 75 °F | Melting/freezing point of palm kernel oil [22] |
| 298 K | 25 °C | 77 °F | Melting/freezing point of coconut oil [22] |
| 300 K | 27 °C | 81 °F | Thermoneutral temperature of an unclothed human at rest [35] [36] Estimated melting/freezing point of francium |
| 302.9 K | 29.8 °C | 85.6 °F | Melting/freezing point of gallium |
| 303.15 K | 30 °C | 86 °F | The rate of plant growth is typically no greater above this temperature than at this temperature. (see Growing degree-day) |
| 304 K | 31 °C | 88 °F | Melting/freezing point of butter, critical point for carbon dioxide |
| 307 K | 34 °C | 93 °F | Autoignition temperature of white phosphorus |
| 307.6 K | 34.4 °C | 93.9 °F | Hottest annual mean temperature on Earth, at Dallol, Ethiopia [20] |
| 308 K | 35 °C | 95 °F | Hypothermic body temperature for humans (see Hypothermia) Warmest sea measured, at the Red Sea Melting/freezing point of palm oil [22] |
| 309.5 K | 36.4 °C | 97.5 °F | Average body temperature for a human [37] |
| 311.03 K | 37.87 °C | 100.2 °F | Beginnings of a fever for humans |
| 311.8 K | 38.6 °C | 101.5 °F | Average body temperature for a cat [38] |
| 313.15 K | 40 °C | 104 °F | Maximum standard temperature recommended for hot tub users [39] |
| 315 K | 42 °C | 108 °F | Usually fatal human fever |
| 317.6 K | 44.44 °C | 112 °F | Hot threshold of pain if skin reaches this temperature |
| 319.3 K | 46.1 °C | 115 °F | World's hottest air temperature recorded while raining, at Needles, California, USA on August 13, 2012 [40] |
| 319.7 K | 46.5 °C | 115.7 °F | Highest human fever survived (Willie Jones) [41] |
| 322.1 K | 48.9 °C | 120.0 °F | Hottest air temperature recorded in South America, at Rivadavia, Argentina on 1905-12-11 [24] Maximum safe temperature for hot water according to numeric U.S. plumbing codes [42] Water will cause a second-degree burn after 8 minutes and a third-degree burn after 10 minutes [42] |
| 323.14 K | 49.99 °C | 121.99 °F | Half-way point between freezing and boiling |
| 323.9 K | 50.7 °C | 123.3 °F | Hottest air temperature recorded in the Southern Hemisphere, at Oodnadatta, Australia on 1960-02-01 [24] |
| 329.87 K | 56.7 °C | 134.1 °F | Hottest measured air temperature on Earth, in Death Valley at Furnace Creek, Inyo County, California, United States of America on 10 July 1913. [43] |
| 333.15 K | 60 °C | 140 °F | Water will cause a second-degree burn in 3 seconds and a third-degree burn in 5 seconds [42] Average temperature of a hair dryer |
| 336 K | 63 °C | 145.4 °F | Milk pasteurization |
| 342 K | 69 °C | 157 °F | Boiling point of water on the summit of Mount Everest [44] |
| 343.15 K | 70 °C | 158 °F | Food is well done Hot springs at which some bacteria thrive [45] |
| 350 K | 77 °C | 170 °F | Poaching of food |
| 351.52 K | 78.37 °C | 173.07 °F | Boiling point of ethanol |
| 353.15 K | 80 °C | 176 °F | Average temperature of a sauna |
| 355 K | 82 °C | 180 °F | Recommended final rinse temperature in industrial-grade commercial dishwashers [46] |
| 355.6 K | 82.4 °C | 180.3 °F | Boiling point of isopropyl alcohol [17] |
| 366 K | 93 °C | 200 °F | Simmering of food |
| 367 K | 94 °C | 201 °F | Hottest ground temperature recorded on Earth at Furnace Creek, Death Valley, California, USA on 1972-07-15 [47] |
| 371 K | 98 °C | 209 °F | Freezing/melting point of sodium |
| 373.13 K | 99.98 °C | 211.97 °F | Boiling point of water at 1 atm pressure (see Celsius) |
| 380 K | 107 °C | 225 °F | Smoke point of raw safflower oil Syrup is concentrated to 75% sugar |
| 388 K | 115 °C | 239 °F | Melting/freezing point of sulfur |
| 400 K | 127 °C | 260 °F | Concorde nose tip during supersonic flight Coldest known stars in space (approximate temperature) [48] |
| 433.15 K | 160 °C | 320 °F | Syrup is concentrated to 100% sugar Sucrose (table sugar) caramelizes |
| 450 K | 177 °C | 350 °F | Mean on Mercury Smoke point of butter Deep frying |
| 453.15 K | 180 °C | 356 °F | Popcorn pops |
| 483 K | 210 °C | 410 °F | Autoignition (kindling) point of diesel fuel |
| 491 K | 218 °C | 425 °F | Kindling point of paper |
| 519 K | 246 °C | 475 °F | Kindling point of automotive gasoline |
| 522 K | 249 °C | 480 °F | Kindling point of jet fuel (Jet A/Jet A-1) [49] |
| 525 K | 252 °C | 485 °F | Smoke point of milkfat Kindling point of jet fuel (Jet B) [49] |
| 538 K | 265 °C | 510 °F | Smoke point of refined safflower oil |
| 574.5875 K | 301.4375 °C | 574.5875 °F | Intersecting point of the Fahrenheit and Kelvin temperature scales |
| 600.65 K | 327.5 °C | 621.5 °F | Melting/freezing point of lead |
| 647 K | 374 °C | 705 °F | Critical point of superheated water |
| 693 K | 419 °C | 787 °F | Melting/freezing point of zinc |
| 723.15 K | 450 °C | 842 °F | Kindling point of aviation gasoline [49] |
| 738 K | 465 °C | 870 °F | Mean on Venus |
| 749 K | 476 °C | 889 °F | Kindling point of magnesium |
| 773.15 K | 500 °C | 932 °F | Oven on self-cleaning mode |
| 798 K | 525 °C | 977 °F | Draper Point (the point at which nearly all objects start to glow dim red) [50] |
| 858 K | 585 °C | 1085 °F | Kindling point of hydrogen [51] |
| 933.47 K | 660.32 °C | 1220.58 °F | Melting/freezing point of aluminium |
| 1000 K | 726.85 °C | 1340.33 °F |
Detailed list from 0 K to 142 QK -273.15 Celsius Absolute Zero, nothing can get colder than this -272.15 Celsius Boomerang Nebula -270 Celsius Outer Space -269 boiling point of helium -259 freezing point of hydrogen -253 condensation point of hydrogen -241 average temperature on Haumea -235 average temperature on Triton
| Submultiples | Multiples | ||||
|---|---|---|---|---|---|
| Value | SI symbol | Name | Value | SI symbol | Name |
| 10−1 K | dK | decikelvin | 101 K | daK | decakelvin |
| 10−2 K | cK | centikelvin | 102 K | hK | hectokelvin |
| 10−3 K | mK | millikelvin | 103 K | kK | kilokelvin |
| 10−6 K | μK | microkelvin | 106 K | MK | megakelvin |
| 10−9 K | nK | nanokelvin | 109 K | GK | gigakelvin |
| 10−12 K | pK | picokelvin | 1012 K | TK | terakelvin |
| 10−15 K | fK | femtokelvin | 1015 K | PK | petakelvin |
| 10−18 K | aK | attokelvin | 1018 K | EK | exakelvin |
| 10−21 K | zK | zeptokelvin | 1021 K | ZK | zettakelvin |
| 10−24 K | yK | yoctokelvin | 1024 K | YK | yottakelvin |
| 10−27 K | rK | rontokelvin | 1027 K | RK | ronnakelvin |
| 10−30 K | qK | quectokelvin | 1030 K | QK | quettakelvin |
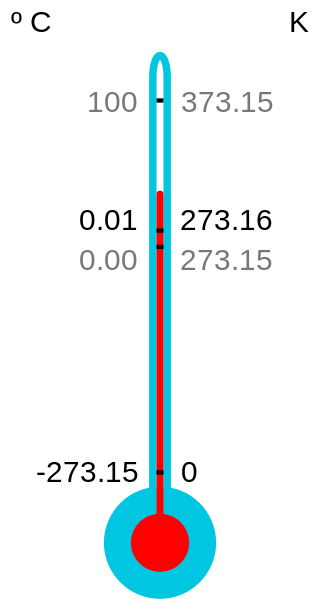
Absolute zero is the lowest limit of the thermodynamic temperature scale; a state at which the enthalpy and entropy of a cooled ideal gas reach their minimum value, taken as zero kelvin. The fundamental particles of nature have minimum vibrational motion, retaining only quantum mechanical, zero-point energy-induced particle motion. The theoretical temperature is determined by extrapolating the ideal gas law; by international agreement, absolute zero is taken as −273.15 degrees on the Celsius scale, which equals −459.67 degrees on the Fahrenheit scale. The corresponding Kelvin and Rankine temperature scales set their zero points at absolute zero by definition.
Weather is the state of the atmosphere, describing for example the degree to which it is hot or cold, wet or dry, calm or stormy, clear or cloudy. On Earth, most weather phenomena occur in the lowest layer of the planet's atmosphere, the troposphere, just below the stratosphere. Weather refers to day-to-day temperature, precipitation, and other atmospheric conditions, whereas climate is the term for the averaging of atmospheric conditions over longer periods of time. When used without qualification, "weather" is generally understood to mean the weather of Earth.

Numerical climate models are mathematical models that can simulate the interactions of important drivers of climate. These drivers are the atmosphere, oceans, land surface and ice. Scientists use climate models to study the dynamics of the climate system and to make projections of future climate and of climate change. Climate models can also be qualitative models and contain narratives, largely descriptive, of possible futures.
Unconventional superconductors are materials that display superconductivity which does not conform to conventional BCS theory or its extensions.
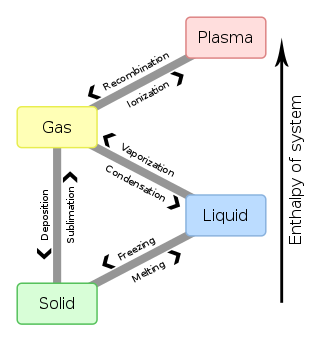
In chemistry, thermodynamics, and other related fields, a phase transition is the physical process of transition between one state of a medium and another. Commonly the term is used to refer to changes among the basic states of matter: solid, liquid, and gas, and in rare cases, plasma. A phase of a thermodynamic system and the states of matter have uniform physical properties. During a phase transition of a given medium, certain properties of the medium change as a result of the change of external conditions, such as temperature or pressure. This can be a discontinuous change; for example, a liquid may become gas upon heating to its boiling point, resulting in an abrupt change in volume. The identification of the external conditions at which a transformation occurs defines the phase transition point.
Metallic hydrogen is a phase of hydrogen in which it behaves like an electrical conductor. This phase was predicted in 1935 on theoretical grounds by Eugene Wigner and Hillard Bell Huntington.

The climate of Antarctica is the coldest on Earth. The continent is also extremely dry, averaging 166 mm (6.5 in) of precipitation per year. Snow rarely melts on most parts of the continent, and, after being compressed, becomes the glacier ice that makes up the ice sheet. Weather fronts rarely penetrate far into the continent, because of the katabatic winds. Most of Antarctica has an ice-cap climate with extremely cold and dry weather.
The faint young Sun paradox or faint young Sun problem describes the apparent contradiction between observations of liquid water early in Earth's history and the astrophysical expectation that the Sun's output would be only 70 percent as intense during that epoch as it is during the modern epoch. The paradox is this: with the young Sun's output at only 70 percent of its current output, early Earth would be expected to be completely frozen, but early Earth seems to have had liquid water and supported life.

Cold is the presence of low temperature, especially in the atmosphere. In common usage, cold is often a subjective perception. A lower bound to temperature is absolute zero, defined as 0.00 K on the Kelvin scale, an absolute thermodynamic temperature scale. This corresponds to −273.15 °C on the Celsius scale, −459.67 °F on the Fahrenheit scale, and 0.00 °R on the Rankine scale.

The Mars ocean theory states that nearly a third of the surface of Mars was covered by an ocean of liquid water early in the planet's geologic history. This primordial ocean, dubbed Paleo-Ocean or Oceanus Borealis, would have filled the basin Vastitas Borealis in the northern hemisphere, a region that lies 4–5 km below the mean planetary elevation, at a time period of approximately 4.1–3.8 billion years ago. Evidence for this ocean includes geographic features resembling ancient shorelines, and the chemical properties of the Martian soil and atmosphere. Early Mars would have required a denser atmosphere and warmer climate to allow liquid water to remain at the surface.

The carbonate–silicate geochemical cycle, also known as the inorganic carbon cycle, describes the long-term transformation of silicate rocks to carbonate rocks by weathering and sedimentation, and the transformation of carbonate rocks back into silicate rocks by metamorphism and volcanism. Carbon dioxide is removed from the atmosphere during burial of weathered minerals and returned to the atmosphere through volcanism. On million-year time scales, the carbonate-silicate cycle is a key factor in controlling Earth's climate because it regulates carbon dioxide levels and therefore global temperature.

Mubi North is a Local Government Area of Adamawa State, Nigeria. The town is the location of the Adamawa State University and the Federal Polytechnic, Mubi.

Climate change in New York City could affect buildings/structures, wetlands, water supply, health, and energy demand, due to the high population and extensive infrastructure in the region. A seaport like New York is especially at risk if the sea level rises, with many bridges and tunnels in the city. Major facilities for Aviation in the New York metropolitan area, and the New York Passenger Ship Terminal, are also located in areas vulnerable to flooding. Flooding would be expensive to reverse. Tide gauge records indicate a rise in sea level of about 50 cm since 1860.

Frost weathering is a collective term for several mechanical weathering processes induced by stresses created by the freezing of water into ice. The term serves as an umbrella term for a variety of processes, such as frost shattering, frost wedging, and cryofracturing. The process may act on a wide range of spatial and temporal scales, from minutes to years and from dislodging mineral grains to fracturing boulders. It is most pronounced in high-altitude and high-latitude areas and is especially associated with alpine, periglacial, subpolar maritime, and polar climates, but may occur anywhere at sub-freezing temperatures if water is present.

The Noachian is a geologic system and early time period on the planet Mars characterized by high rates of meteorite and asteroid impacts and the possible presence of abundant surface water. The absolute age of the Noachian period is uncertain but probably corresponds to the lunar Pre-Nectarian to Early Imbrian periods of 4100 to 3700 million years ago, during the interval known as the Late Heavy Bombardment. Many of the large impact basins on the Moon and Mars formed at this time. The Noachian Period is roughly equivalent to the Earth's Hadean and early Archean eons when Earth's first life forms likely arose.

Ajay Kumar Sood is an Indian physicist and researcher currently serving as the 4th Principal Scientific Adviser to the Government of India.
The highest temperature recorded on Earth has been measured in three major ways: air, ground, and via satellite observation. Air measurements are used as the standard measurement due to persistent issues with unreliable ground and satellite readings. Air measurements are noted by the World Meteorological Organization (WMO) and Guinness World Records among others as the standard to be used for determining the official record. The current official highest registered air temperature on Earth is 56.7 °C (134.1 °F), recorded on 10 July 1913 at Furnace Creek Ranch, in Death Valley in the United States. For ninety years, a former record that was measured in Libya had been in place, until it was decertified in 2012 based on evidence that it was an erroneous reading. This finding has since raised questions about the legitimacy of the 1913 record measured in Death Valley, with several meteorological experts asserting that there were similar irregularities. The WMO has stood by the record as official pending any future investigative results. If the current record were to be decertified then the holder would be a tie at 54.0 °C (129.2 °F), recorded both at Furnace Creek and in Kuwait.

Giorgio Benedek is an Italian physicist, academic and researcher. He is an Emeritus Professor of Physics of Matter at University of Milano-Bicocca and Director of the International School of Solid State Physics at Ettore Majorana Foundation and Centre for Scientific Culture.