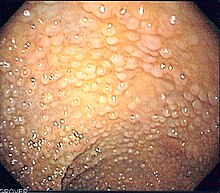
Colorectal cancer (CRC), also known as bowel cancer, colon cancer, or rectal cancer, is the development of cancer from the colon or rectum. Signs and symptoms may include blood in the stool, a change in bowel movements, weight loss, and fatigue. Most colorectal cancers are due to old age and lifestyle factors, with only a small number of cases due to underlying genetic disorders. Risk factors include diet, obesity, smoking, and lack of physical activity. Dietary factors that increase the risk include red meat, processed meat, and alcohol. Another risk factor is inflammatory bowel disease, which includes Crohn's disease and ulcerative colitis. Some of the inherited genetic disorders that can cause colorectal cancer include familial adenomatous polyposis and hereditary non-polyposis colon cancer; however, these represent less than 5% of cases. It typically starts as a benign tumor, often in the form of a polyp, which over time becomes cancerous.

In anatomy, a polyp is an abnormal growth of tissue projecting from a mucous membrane. If it is attached to the surface by a narrow elongated stalk, it is said to be pedunculated; if it is attached without a stalk, it is said to be sessile. Polyps are commonly found in the colon, stomach, nose, ear, sinus(es), urinary bladder, and uterus. They may also occur elsewhere in the body where there are mucous membranes, including the cervix, vocal folds, and small intestine. Some polyps are tumors (neoplasms) and others are non-neoplastic, for example hyperplastic or dysplastic, which are benign. The neoplastic ones are usually benign, although some can be pre-malignant, or concurrent with a malignancy.

Gardner's syndrome is a subtype of familial adenomatous polyposis (FAP). Gardner syndrome is an autosomal dominant form of polyposis characterized by the presence of multiple polyps in the colon together with tumors outside the colon. The extracolonic tumors may include osteomas of the skull, thyroid cancer, epidermoid cysts, fibromas, as well as the occurrence of desmoid tumors in approximately 15% of affected individuals.

Hereditary nonpolyposis colorectal cancer (HNPCC) is a hereditary predisposition to colon cancer.

A benign tumor is a mass of cells (tumor) that does not invade neighboring tissue or metastasize. Compared to malignant (cancerous) tumors, benign tumors generally have a slower growth rate. Benign tumors have relatively well differentiated cells. They are often surrounded by an outer surface or stay contained within the epithelium. Common examples of benign tumors include moles and uterine fibroids.

Mismatch repair cancer syndrome (MMRCS) is a cancer syndrome associated with biallelic DNA mismatch repair mutations. It is also known as Turcot syndrome and by several other names.
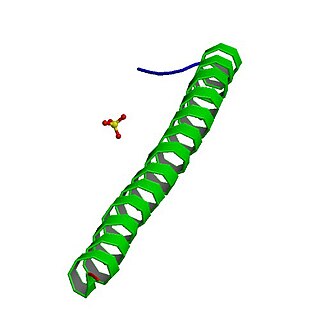
Adenomatous polyposis coli (APC) also known as deleted in polyposis 2.5 (DP2.5) is a protein that in humans is encoded by the APC gene. The APC protein is a negative regulator that controls beta-catenin concentrations and interacts with E-cadherin, which are involved in cell adhesion. Mutations in the APC gene may result in colorectal cancer and desmoid tumors.

MUTYH is a human gene that encodes a DNA glycosylase, MUTYH glycosylase. It is involved in oxidative DNA damage repair and is part of the base excision repair pathway. The enzyme excises adenine bases from the DNA backbone at sites where adenine is inappropriately paired with guanine, cytosine, or 8-oxo-7,8-dihydroguanine, a common form of oxidative DNA damage.

Fundic gland polyposis is a medical syndrome where the fundus and the body of the stomach develop many fundic gland polyps. The condition has been described both in patients with familial adenomatous polyposis (FAP) and attenuated variants (AFAP), and in patients in whom it occurs sporadically.

Aggressive fibromatosis or desmoid tumor is a rare condition. Desmoid tumors are a type of fibromatosis and related to sarcoma, though without the ability to spread throughout the body (metastasize). The tumors arise from cells called fibroblasts, which are found throughout the body and provide structural support, protection to the vital organs, and play a critical role in wound healing. These tumors tend to occur in women in their thirties, but can occur in anyone at any age. They can be either relatively slow-growing or malignant. However, aggressive fibromatosis is locally aggressive and can cause life-threatening problems or even death when the tumors compress vital organs such as intestines, kidneys, lungs, blood vessels, or nerves. The condition is rarely fatal. Most cases are sporadic, but some are associated with familial adenomatous polyposis (FAP). Approximately 10% of individuals with Gardner's syndrome, a type of FAP with extracolonic features, have desmoid tumors.

Juvenile polyposis syndrome is an autosomal dominant genetic condition characterized by the appearance of multiple juvenile polyps in the gastrointestinal tract. Polyps are abnormal growths arising from a mucous membrane. These usually begin appearing before age 20, but the term juvenile refers to the type of polyp, not to the age of the affected person. While the majority of the polyps found in juvenile polyposis syndrome are non-neoplastic, hamartomatous, self-limiting and benign, there is an increased risk of adenocarcinoma.

A colorectal polyp is a polyp occurring on the lining of the colon or rectum. Untreated colorectal polyps can develop into colorectal cancer.
Mouse models of colorectal cancer and intestinal cancer are experimental systems in which mice are genetically manipulated, fed a modified diet, or challenged with chemicals to develop malignancies in the gastrointestinal tract. These models enable researchers to study the onset, progression of the disease, and understand in depth the molecular events that contribute to the development and spread of colorectal cancer. They also provide a valuable biological system, to simulate human physiological conditions, suitable for testing therapeutics.
Attenuated familial adenomatous polyposis is a form of familial adenomatous polyposis, a cancer syndrome. It is a pre-malignant disease that can develop into colorectal cancer. A patient will have fewer than a hundred polyps located typically in right side of the colon. Cancer might develop as early as the age of five, though typically presents later than classical FAP.
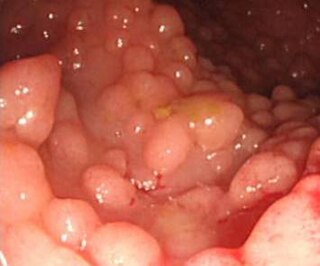
A hereditary cancer syndrome is a genetic disorder in which inherited genetic mutations in one or more genes predispose the affected individuals to the development of cancer and may also cause early onset of these cancers. Hereditary cancer syndromes often show not only a high lifetime risk of developing cancer, but also the development of multiple independent primary tumors.

APC, WNT signaling pathway regulator is a protein that in humans is encoded by the APC gene.
MUTYH-associated polyposis is an autosomal recessive polyposis syndrome. The disorder is caused by mutations in both alleles of the DNA repair gene, MUTYH. The MUTYH gene encodes a base excision repair protein, which corrects oxidative damage to DNA. Affected individuals have an increased risk of colorectal cancer, precancerous colon polyps (adenomas) and an increased risk of several additional cancers. About 1–2 percent of the population possess a mutated copy of the MUTYH gene, and less than 1 percent of people have the MUTYH associated polyposis syndrome. The presence of 10 or more colon adenomas should prompt consideration of MUTYH-associated polyposis, familial adenomatous polyposis and similar syndromes.
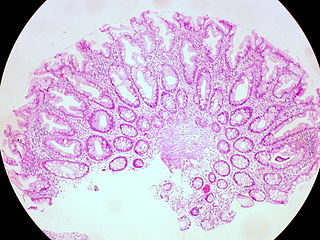
Serrated polyposis syndrome (SPS), previously known as hyperplastic polyposis syndrome, is a disorder characterized by the appearance of serrated polyps in the colon. While serrated polyposis syndrome does not cause symptoms, the condition is associated with a higher risk of colorectal cancer (CRC). The lifelong risk of CRC is between 25 and 40%. SPS is the most common polyposis syndrome affecting the colon, but is under recognized due to a lack of systemic long term monitoring. Diagnosis requires colonoscopy, and is defined by the presence of either of two criteria: ≥5 serrated lesions/polyps proximal to the rectum, or >20 serrated lesions/polyps of any size distributed throughout the colon with 5 proximal to the rectum.
Polymerase proofreading-associated polyposis (PPAP) is an autosomal dominant hereditary cancer syndrome, which is characterized by numerous polyps in the colon and an increased risk of colorectal cancer. It is caused by germline mutations in DNA polymerase ε (POLE) and δ (POLD1). Affected individuals develop numerous polyps called colorectal adenomas. Compared with other polyposis syndromes, Polymerase proofreading-associated polyposis is rare. Genetic testing can help exclude similar syndromes, such as Familial adenomatous polyposis and MUTYH-associated polyposis. Endometrial cancer, duodenal polyps and duodenal cancer may also occur.
Gardner fibroma (GF) is a benign fibroblastic tumor. GF tumors typically develop in the dermis and adjacent subcutaneous tissue lying just below the dermis. These tumors typically occur on the back, abdomen, and other superficial sites but in rare cases have been diagnoses in internal sites such as the retroperitoneum and around the large blood vessels in the upper thoracic cavity. The World Health Organization, 2020, classified Gardner fibroma as a benign tumor in the category of fibroblastic and myofibroblastic tumors.