Nerve agents, sometimes also called nerve gases, are a class of organic chemicals that disrupt the mechanisms by which nerves transfer messages to organs. The disruption is caused by the blocking of acetylcholinesterase (AChE), an enzyme that catalyzes the breakdown of acetylcholine, a neurotransmitter. Nerve agents are irreversible acetylcholinesterase inhibitors used as poison.

Sarin is an extremely toxic synthetic organophosphorus compound. A colourless, odourless liquid, it is used as a chemical weapon due to its extreme potency as a nerve agent. Exposure is lethal even at very low concentrations, where death can occur within one to ten minutes after direct inhalation of a lethal dose, due to suffocation from respiratory paralysis, unless antidotes are quickly administered. People who absorb a non-lethal dose and do not receive immediate medical treatment may suffer permanent neurological damage.

Soman is an extremely toxic chemical substance. It is a nerve agent, interfering with normal functioning of the mammalian nervous system by inhibiting the enzyme cholinesterase. It is an inhibitor of both acetylcholinesterase and butyrylcholinesterase. As a chemical weapon, it is classified as a weapon of mass destruction by the United Nations according to UN Resolution 687. Its production is strictly controlled, and stockpiling is outlawed by the Chemical Weapons Convention of 1993 where it is classified as a Schedule 1 substance. Soman was the third of the so-called G-series nerve agents to be discovered along with GA (tabun), GB (sarin), and GF (cyclosarin).

VX is an extremely toxic synthetic chemical compound in the organophosphorus class, specifically, a thiophosphonate. In the class of nerve agents, it was developed for military use in chemical warfare after translation of earlier discoveries of organophosphate toxicity in pesticide research. In recent years, VX was found to be the agent used in the assassination of Kim Jong-nam. In its pure form, VX is an oily, relatively non-volatile liquid that is amber-like in colour. Because of its low volatility, VX persists in environments where it is dispersed.

The enzyme cholinesterase (EC 3.1.1.8, choline esterase; systematic name acylcholine acylhydrolase) catalyses the hydrolysis of choline-based esters:

VG is a "V-series" nerve agent chemically similar to the better-known VX nerve agent. Tetram is the common Russian name for the substance. Amiton was the trade name for the substance when it was marketed as an insecticide by ICI in the mid-1950s.
Obidoxime is a member of the oxime family used to treat nerve gas poisoning. Oximes are drugs known for their ability to reverse the binding of organophosphorus compounds to the enzyme acetylcholinesterase (AChE).
Demeton-S-methyl is an organic compound with the molecular formula C6H15O3PS2. It was used as an organothiophosphate acaricide and organothiophosphate insecticide. It is flammable. With prolonged storage, Demeton-S-methyl becomes more toxic due to formation of a sulfonium derivative which has greater affinity to the human form of the acetylcholinesterase enzyme, and this may present a hazard in agricultural use.
Novichok is a group of nerve agents, some of which are binary chemical weapons. The agents were developed at the GosNIIOKhT state chemical research institute by the Soviet Union and Russia between 1971 and 1993. Some Novichok agents are solids at standard temperature and pressure, while others are liquids. Dispersal of solid form agents is thought possible if in ultrafine powder state.
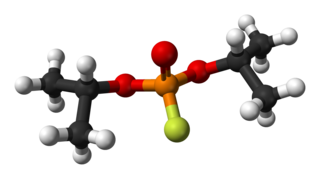
Diisopropyl fluorophosphate (DFP) or Isoflurophate is an oily, colorless liquid with the chemical formula C6H14FO3P. It is used in medicine and as an organophosphorus insecticide. It is stable, but undergoes hydrolysis when subjected to moisture.

Pralidoxime or 2-PAM, usually as the chloride or iodide salts, belongs to a family of compounds called oximes that bind to organophosphate-inactivated acetylcholinesterase. It is used to treat organophosphate poisoning in conjunction with atropine and either diazepam or midazolam. It is a white solid.
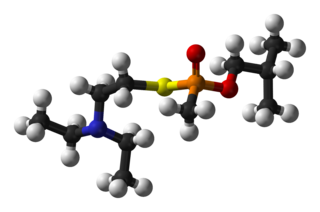
VR is a "V-series" unitary nerve agent closely related to the better-known VX nerve agent. It became a prototype for the series of Novichok agents. According to chemical weapons expert Jonathan Tucker, the first binary formulation developed under the Soviet Foliant program was used to make Substance 33, differing from VX only in the alkyl substituents on its nitrogen and oxygen atoms. "This weapon was given the code name Novichok."

Cholinesterase inhibitors (ChEIs), also known as anti-cholinesterase, are chemicals that prevent the breakdown of the neurotransmitter acetylcholine or butyrylcholine. This increases the amount of the acetylcholine or butyrylcholine in the synaptic cleft that can bind to muscarinic receptors, nicotinic receptors and others. This group of inhibitors is divided into two subgroups, acetylcholinesterase inhibitors (AChEIs) and butyrylcholinesterase inhibitors (BChEIs).

Tetraethyl pyrophosphate, abbreviated TEPP, is an organophosphate compound with the formula [(C2H5O)2P(O)]2O. It is the tetraethyl derivative of pyrophosphate (P2O74-). It is a colorless oil that solidifies near room temperature. It is used as an insecticide. The compound hydrolyzes rapidly.
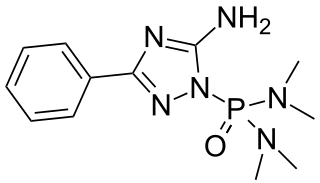
Triamiphos (chemical formula: C12H19N6OP) is an organophosphate used as a pesticide and fungicide. It is used to control powdery mildews on apples and ornamentals. It was discontinued by the US manufacturer in 1998.
IDFP is an organophosphorus compound related to the nerve agent sarin.

IPTBO is a bicyclic phosphate convulsant. It is an extremely potent GABA receptor antagonist that can cause violent convulsions in mice.
Bicyclic phosphate is a class of organophosphate compounds that are used as flame retardants, stabilizers and antioxidants. They are also used in spectroscopic studies.

Crotylsarin (CRS) is an extremely toxic organophosphate nerve agent of the G-series. Like other nerve agents, CRS irreversibly inhibits the acetylcholinesterase. However, since the inhibited enzyme ages so rapidly, the inhibited enzyme can't be reactivated by cholinesterase reactivators.

Selenophos is an extremely potent organophosphate acetylcholinesterase inhibitor. It is the selenium analog of the VE nerve agent.













