
Rhenium is a chemical element; it has symbol Re and atomic number 75. It is a silvery-gray, heavy, third-row transition metal in group 7 of the periodic table. With an estimated average concentration of 1 part per billion (ppb), rhenium is one of the rarest elements in the Earth's crust. It has the third-highest melting point and second-highest boiling point of any element at 5869 K. It resembles manganese and technetium chemically and is mainly obtained as a by-product of the extraction and refinement of molybdenum and copper ores. It shows in its compounds a wide variety of oxidation states ranging from −1 to +7.

Molybdenum disulfide is an inorganic compound composed of molybdenum and sulfur. Its chemical formula is MoS
2.
Molybdenum trioxide describes a family of inorganic compounds with the formula MoO3(H2O)n where n = 0, 1, 2. The anhydrous compound is produced on the largest scale of any molybdenum compound since it is the main intermediate produced when molybdenum ores are purified. The anhydrous oxide is a precursor to molybdenum metal, an important alloying agent. It is also an important industrial catalyst. It is a yellow solid, although impure samples can appear blue or green.

Sodium phosphide is the inorganic compound with the formula Na3P. It is a black solid. It is often described as Na+ salt of the P3− anion. Na3P is a source of the highly reactive phosphide anion. It should not be confused with sodium phosphate, Na3PO4.
Germanium dioxide, also called germanium(IV) oxide, germania, and salt of germanium, is an inorganic compound with the chemical formula GeO2. It is the main commercial source of germanium. It also forms as a passivation layer on pure germanium in contact with atmospheric oxygen.
Molybdenum dioxide is the chemical compound with the formula MoO2. It is a violet-colored solid and is a metallic conductor. The mineralogical form of this compound is called tugarinovite, and is only very rarely found. The discovery and early studies of molybdenum dioxide date back to the late 18th and early 19th centuries. One of the notable figures in the history of molybdenum dioxide is the Hungarian chemist Jakob Joseph Winterl (1732-1809). Winterl, who was a professor of chemistry and botany at the University of Budapest, made significant contributions to the understanding of molybdenum compounds. In 1787, he proposed that copper was a compound of nickel, molybdenum, silica, and a volatile substance, showcasing his interest in molybdenum chemistry.
Iron phosphide is a chemical compound of iron and phosphorus, with a formula of FeP. Its physical appearance is grey, hexagonal needles.
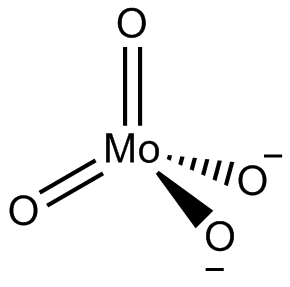
In chemistry, a molybdate is a compound containing an oxyanion with molybdenum in its highest oxidation state of 6: O−−Mo(=O)2−O−. Molybdenum can form a very large range of such oxyanions, which can be discrete structures or polymeric extended structures, although the latter are only found in the solid state. The larger oxyanions are members of group of compounds termed polyoxometalates, and because they contain only one type of metal atom are often called isopolymetalates. The discrete molybdenum oxyanions range in size from the simplest MoO2−
4, found in potassium molybdate up to extremely large structures found in isopoly-molybdenum blues that contain for example 154 Mo atoms. The behaviour of molybdenum is different from the other elements in group 6. Chromium only forms the chromates, CrO2−
4, Cr
2O2−
7, Cr
3O2−
10 and Cr
4O2−
13 ions which are all based on tetrahedral chromium. Tungsten is similar to molybdenum and forms many tungstates containing 6 coordinate tungsten.
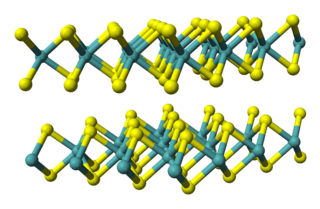
Molybdenum diselenide is an inorganic compound of molybdenum and selenium. Its structure is similar to that of MoS
2. Compounds of this category are known as transition metal dichalcogenides, abbreviated TMDCs. These compounds, as the name suggests, are made up of a transition metals and elements of group 16 on the periodic table of the elements. Compared to MoS
2, MoSe
2 exhibits higher electrical conductivity.
Lithium phosphide is an inorganic compound of lithium and phosphorus with the chemical formula Li
3P. This dark colored compound is formally the Li+ salt of P3-. It is a hazardous to handle because of its high reactivity toward air.

Scandium phosphide is an inorganic compound of scandium and phosphorus with the chemical formula ScP.
Lutetium phosphide is an inorganic compound of lutetium and phosphorus with the chemical formula LuP. The compound forms dark crystals, does not dissolve in water.
Praseodymium monophosphide is an inorganic compound of praseodymium and phosphorus with the chemical formula PrP. The compound forms crystals.
Europium phosphide is an inorganic compound of europium and phosphorus with the chemical formula EuP. Other phosphides are also known.
Thulium phosphide is an inorganic compound of thulium and phosphorus with the chemical formula TmP.
Ytterbium compounds are chemical compounds that contain the element ytterbium (Yb). The chemical behavior of ytterbium is similar to that of the rest of the lanthanides. Most ytterbium compounds are found in the +3 oxidation state, and its salts in this oxidation state are nearly colorless. Like europium, samarium, and thulium, the trihalides of ytterbium can be reduced to the dihalides by hydrogen, zinc dust, or by the addition of metallic ytterbium. The +2 oxidation state occurs only in solid compounds and reacts in some ways similarly to the alkaline earth metal compounds; for example, ytterbium(II) oxide (YbO) shows the same structure as calcium oxide (CaO).
Holmium nitride is a binary inorganic compound of holmium and nitrogen with the chemical formula HoN.
Zirconium monophosphide is a binary inorganic compound of zirconium metal and phosphorus with the chemical formula ZrP.
Trimolybdenum phosphide is a binary inorganic compound of molybdenum metal and phosphorus with the chemical formula Mo3P.
Molybdenum diphosphide is a binary inorganic compound of molybdenum metal and phosphorus with the chemical formula MoP2.







