In chemistry, a structural isomer of a compound is another compound whose molecule has the same number of atoms of each element, but with logically distinct bonds between them. The term metamer was formerly used for the same concept.
In chemistry, an oxonium ion is any cation containing an oxygen atom that has three bonds and 1+ formal charge. The simplest oxonium ion is the hydronium ion.

A trimethylsilyl group (abbreviated TMS) is a functional group in organic chemistry. This group consists of three methyl groups bonded to a silicon atom [−Si(CH3)3], which is in turn bonded to the rest of a molecule. This structural group is characterized by chemical inertness and a large molecular volume, which makes it useful in a number of applications.

tert-Butyl alcohol is the simplest tertiary alcohol, with a formula of (CH3)3COH (sometimes represented as t-BuOH). Its isomers are 1-butanol, isobutanol, and butan-2-ol. tert-Butyl alcohol is a colorless solid, which melts near room temperature and has a camphor-like odor. It is miscible with water, ethanol and diethyl ether.
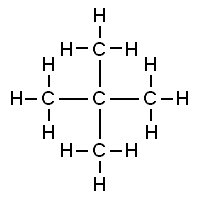
Neopentane, also called 2,2-dimethylpropane, is a double-branched-chain alkane with five carbon atoms. Neopentane is a flammable gas at room temperature and pressure which can condense into a highly volatile liquid on a cold day, in an ice bath, or when compressed to a higher pressure.

In stereochemistry, a chiral auxiliary is a stereogenic group or unit that is temporarily incorporated into an organic compound in order to control the stereochemical outcome of the synthesis. The chirality present in the auxiliary can bias the stereoselectivity of one or more subsequent reactions. The auxiliary can then be typically recovered for future use.

tert-Butyllithium is a chemical compound with the formula (CH3)3CLi. As an organolithium compound, it has applications in organic synthesis since it is a strong base, capable of deprotonating many carbon molecules, including benzene. tert-Butyllithium is available commercially as hydrocarbon solutions; it is not usually prepared in the laboratory.
In chemistry, a Grob fragmentation is an elimination reaction that breaks a neutral aliphatic chain into three fragments: a positive ion spanning atoms 1 and 2, an unsaturated neutral fragment spanning positions 3 and 4, and a negative ion comprising the rest of the chain.
Pivalic acid is a carboxylic acid with a molecular formula of (CH3)3CCO2H. This colourless, odiferous organic compound is solid at room temperature. Two abbreviation for pivalic acid are t-BuC(O)OH and PivOH. The pivalyl or pivaloyl group is abbreviated t-BuC(O).

sec-Butyllithium is an organometallic compound with the formula CH3CHLiCH2CH3, abbreviated sec-BuLi or s-BuLi. This chiral organolithium reagent is used as a source of sec-butyl carbanion in organic synthesis.
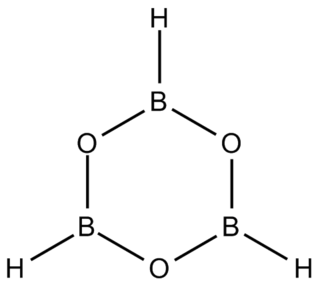
Boroxine is a 6-membered heterocyclic compound composed of alternating oxygen and singly-hydrogenated boron atoms. Boroxine derivatives such as trimethylboroxine and triphenylboroxine also make up a broader class of compounds called boroxines. These compounds are solids that are usually in equilibrium with their respective boronic acids at room temperature. Beside being used in theoretical studies, boroxine is primarily used in the production of optics.

EuFOD is the chemical compound with the formula Eu(OCC(CH3)3CHCOC3F7)3, also called Eu(fod)3. This coordination compound is used primarily as a shift reagent in NMR spectroscopy. It is the premier member of the lanthanide shift reagents and was popular in the 1970s and 1980s.
Triisopropylamine is an organic chemical compound consisting of three isopropyl groups bound to a central nitrogen atom. As a hindered tertiary amine, it can be used as a non-nucleophilic base and as a stabilizer for polymers; however, its applications are limited by its relatively high cost and difficult synthesis.

2,6-Di-tert-butylpyridine is an organic compound with the formula (Me3C)2C5H3N. This colourless, oily liquid is derived from pyridine by replacement of the two H atoms with tert-butyl groups. It is a hindered base. For example, it can be protonated, but it does not form an adduct with boron trifluoride.
The molecular formula C13H28O (molar mass: 200.36 g/mol, exact mass: 200.2140 u) may refer to:

Tetra-tert-butylethylene is a hypothetical organic compound, a hydrocarbon with formula C18H36, or ((H3C−)3C−)2C=C(−C(−CH3)3)2. As the name indicates, its molecular structure can be viewed as an ethylene molecule H2C=CH2 with the four hydrogens replaced by tert-butyl −C(−CH3)3 groups.
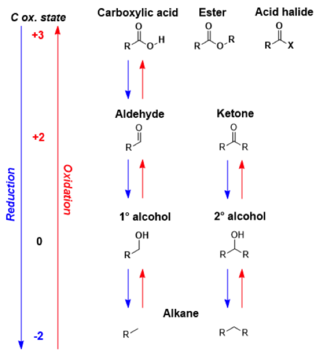
In organic chemistry, carbonyl reduction is the conversion of any carbonyl group, usually to an alcohol. It is a common transformation that is practiced in many ways. Ketones, aldehydes, carboxylic acids, esters, amides, and acid halides - some of the most pervasive functional groups, -comprise carbonyl compounds. Carboxylic acids, esters, and acid halides can be reduced to either aldehydes or a step further to primary alcohols, depending on the strength of the reducing agent. Aldehydes and ketones can be reduced respectively to primary and secondary alcohols. In deoxygenation, the alcohol group can be further reduced and removed altogether by replacement with H.

A silanide is a chemical compound containing an anionic silicon(IV) centre, the parent ion being SiH−3. The hydrogen atoms can also be substituted to produced more complex derivative anions such as tris(trimethylsilyl)silanide (hypersilyl), tris(tert-butyl)silanide, tris(pentafluoroethyl)silanide, or triphenylsilanide. The simple silanide ion can also be called trihydridosilanide or silyl hydride.
Pnictogen-substituted tetrahedranes are pnictogen-containing analogues of tetrahedranes with the formula RxCxPn4-x. Computational work has indicated that the incorporation of pnictogens to the tetrahedral core alleviates the ring strain of tetrahedrane. Although theoretical work on pnictogen-substituted tetrahedranes has existed for decades, only the phosphorus-containing species have been synthesized. These species exhibit novel reactivities, most often through ring-opening and polymerization pathways. Phosphatetrahedranes are of interest as new retrons for organophosphorus chemistry. Their strain also make them of interest in the development of energy-dense compounds.
Phosphiranes are organic compounds with the phosphirane functional group – a three-membered ring with two atoms of carbon and one atom of phosphorus that has lots of ring angle strain. Phosphiranes are usually synthesized by double substitution reactions or pericyclic pathways. Phosphiranes can also be oxidized into phosphirane oxides, undergo SN2 substitution reactions, or decompose into different units.












