Chlorofluorocarbons (CFCs) and hydrochlorofluorocarbons (HCFCs) are fully or partly halogenated hydrocarbons that contain carbon (C), hydrogen (H), chlorine (Cl), and fluorine (F), produced as volatile derivatives of methane, ethane, and propane.

Potassium chloride is a metal halide salt composed of potassium and chlorine. It is odorless and has a white or colorless vitreous crystal appearance. The solid dissolves readily in water, and its solutions have a salt-like taste. Potassium chloride can be obtained from ancient dried lake deposits. KCl is used as a fertilizer, in medicine, in scientific applications, domestic water softeners, and in food processing, where it may be known as E number additive E508.

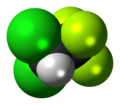
Bromochlorodifluoromethane (BCF), also referred to by the code numbers Halon 1211 and Freon 12B1, is a haloalkane with the chemical formula CF2ClBr. It is used for fire suppression, especially for expensive equipment or items that could be damaged by the residue from other types of extinguishers. It is stored as a liquid under pressure and vaporizes when discharged to suppress fires. The use of halons, including Halon 1211, has decreased over time due to their adverse impact on the ozone layer. Alternatives have been developed to mitigate environmental concerns while still providing effective fire suppression capabilities.
Bromotrifluoromethane, commonly referred to by the code numbers Halon 1301, R13B1, Halon 13B1 or BTM, is an organic halide with the chemical formula CBrF3. It is used for gaseous fire suppression as a far less toxic alternative to bromochloromethane.

A refrigerant is a working fluid used in cooling, heating or reverse cooling and heating of air conditioning systems and heat pumps where they undergo a repeated phase transition from a liquid to a gas and back again. Refrigerants are heavily regulated because of their toxicity and flammability and the contribution of CFC and HCFC refrigerants to ozone depletion and that of HFC refrigerants to climate change.

Chlorodifluoromethane or difluoromonochloromethane is a hydrochlorofluorocarbon (HCFC). This colorless gas is better known as HCFC-22, or R-22, or CHClF
2. It was commonly used as a propellant and refrigerant. These applications were phased out under the Montreal Protocol in developed countries in 2020 due to the compound's ozone depletion potential (ODP) and high global warming potential (GWP), and in developing countries this process will be completed by 2030. R-22 is a versatile intermediate in industrial organofluorine chemistry, e.g. as a precursor to tetrafluoroethylene.
Trifluoroiodomethane, also referred to as trifluoromethyl iodide is a halomethane with the formula CF3I. It is an experimental alternative to Halon 1301 (CBrF3) in unoccupied areas. It would be used as a gaseous fire suppression flooding agent for in-flight aircraft and electronic equipment fires.
A dichlorodifluoroethylene is one of three compounds with the chemical formula C
2Cl
2F
2. Dichlorodifluoroethylenes are colourless gases, and are some of the simplest chlorodifluoroalkenes.

1,1,1-Trifluoroethane, or R-143a or simply trifluoroethane, is a hydrofluorocarbon (HFC) compound that is a colorless gas. It should not be confused with the much more commonly used HFC gas R-134a, nor confused with the isomeric compound 1,1,2-trifluoroethane. 1,1,1-Trifluoroethane has a critical temperature of 73 °C.
A twin-agent fire extinguishing system (TAFES), also commonly referred to as a twin-agent unit (TAU), incorporates the benefits of dry chemical and foam fire extinguishing agents. It is most commonly used for AR-FF operations and in industrial areas with high class B hazards.

A fire extinguisher is a handheld active fire protection device usually filled with a dry or wet chemical used to extinguish or control small fires, often in emergencies. It is not intended for use on an out-of-control fire, such as one which has reached the ceiling, endangers the user, or otherwise requires the equipment, personnel, resources or expertise of a fire brigade. Typically, a fire extinguisher consists of a hand-held cylindrical pressure vessel containing an agent that can be discharged to extinguish a fire. Fire extinguishers manufactured with non-cylindrical pressure vessels also exist but are less common.
1,1,2-Trichloro-1,2,2-trifluoroethane, also called trichlorotrifluoroethane or CFC-113, is a chlorofluorocarbon. It has the formula Cl2FC−CClF2. This colorless, volatile liquid is a versatile solvent.

1,1-Dichloro-1-fluoroethane is a haloalkane with the formula C
2H
3Cl
2F. It is one of the three isomers of dichlorofluoroethane. It belongs to the hydrochlorofluorocarbon (HCFC) family of man-made compounds that contribute significantly to both ozone depletion and global warming when released into the environment.

1-Chloro-1,1-difluoroethane (HCFC-142b) is a haloalkane with the chemical formula CH3CClF2. It belongs to the hydrochlorofluorocarbon (HCFC) family of man-made compounds that contribute significantly to both ozone depletion and global warming when released into the environment. It is primarily used as a refrigerant where it is also known as R-142b and by trade names including Freon-142b.

Condensed aerosol fire suppression is a particle-based method of fire extinction. It is similar to but not identical to dry chemical fire extinction methods, using an innovative pyrogenic, condensed aerosol fire suppressant. It is a highly effective fire suppression method for class A, B, C, E and F. Some aerosol-generating compounds produce a corrosive by-product that may damage electronic equipment, although later generations lower the effect.
Halotron I is a fire extinguishing agent based on the raw material HCFC-123 (93%) mixed with tetrafluoromethane and argon as propellants.
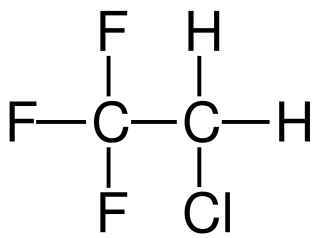
2-Chloro-1,1,1-trifluoroethane, also known as 1,1,1-trifluoro-2-chloroethane or Freon 133a, is an alkyl halide belonging to the category of hydrochlorofluorocarbons, having chemical formula F3C-CH2-Cl. Under standard conditions, it appears as a colorless gas, partially soluble in water. It is used as a refrigerant, as a solvent and as a reagent in organic synthesis.
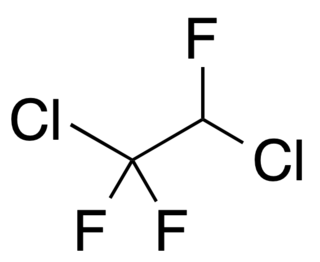
1,2-Dichloro-1,1,2-trifluoroethane is a volatile liquid chlorofluoroalkane composed of carbon, hydrogen, chlorine and fluorine, and with structural formula CClF2CHClF. It is also known as a refrigerant with the designation R-123a.

1,3-Dichloro-1,1,2,2,3-pentafluoropropane is a hydrochlorofluorocarbon. It is a volatile derivative of propane which has served as an HCFC replacement for the CFC, 1,1,2-trichloro-1,2,2-trifluoroethane which was used as a cleaning agent which has been used in the aerospace and electronics industries since the phase out of class 1 ozone depleting substances by the Montreal Protocol. As of 2015 with the phase out of hydrochlorofluorocarbons, HCFC-225 is included in this phase out, and applications where it was used must now be fulfilled by non-ozone depleting substances.

2-Chloro-1,1-difluoroethene (also known as R 1122, u-HCFC-1122 or HCFO-1122) is a toxic unsaturated hydrochlorofluorocarbon which can be written as CF2=CHCl. The HCFO portion of the name stands for hydrochlorofluoroolefin. Another constitutional isomer of it, 1-chloro-1,2-difluoroethylene, is known as HCFO-1122a.