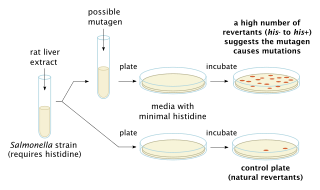
The Ames test is a widely employed method that uses bacteria to test whether a given chemical can cause mutations in the DNA of the test organism. More formally, it is a biological assay to assess the mutagenic potential of chemical compounds. A positive test indicates that the chemical is mutagenic and therefore may act as a carcinogen, because cancer is often linked to mutation. The test serves as a quick and convenient assay to estimate the carcinogenic potential of a compound because standard carcinogen assays on mice and rats are time-consuming and expensive. However, false-positives and false-negatives are known.

A carcinogen is any agent that promotes the development of cancer. Carcinogens can include synthetic chemicals, naturally occurring substances, physical agents such as ionizing and non-ionizing radiation, and biologic agents such as viruses and bacteria. Most carcinogens act by creating mutations in DNA that disrupt a cell's normal processes for regulating growth, leading to uncontrolled cellular proliferation. This occurs when the cell's DNA repair processes fail to identify DNA damage allowing the defect to be passed down to daughter cells. The damage accumulates over time. This is typically a multi-step process during which the regulatory mechanisms within the cell are gradually dismantled allowing for unchecked cellular division.

In genetics, a mutagen is a physical or chemical agent that permanently changes genetic material, usually DNA, in an organism and thus increases the frequency of mutations above the natural background level. As many mutations can cause cancer in animals, such mutagens can therefore be carcinogens, although not all necessarily are. All mutagens have characteristic mutational signatures with some chemicals becoming mutagenic through cellular processes.
Acrylamide (or acrylic amide) is an organic compound with the chemical formula CH2=CHC(O)NH2. It is a white odorless solid, soluble in water and several organic solvents. From the chemistry perspective, acrylamide is a vinyl-substituted primary amide (CONH2). It is produced industrially mainly as a precursor to polyacrylamides, which find many uses as water-soluble thickeners and flocculation agents.
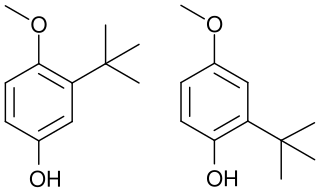
Butylated hydroxyanisole (BHA) is a synthetic, waxy, solid petrochemical. Its antioxidant properties have caused it to be widely used as a preservative in food, food packaging, animal feed, cosmetics, pharmaceuticals, rubber, and petroleum products. BHA has been used in food since around 1947.
Acetaldehyde is an organic chemical compound with the formula CH3 CHO, sometimes abbreviated as MeCHO. It is a colorless liquid or gas, boiling near room temperature. It is one of the most important aldehydes, occurring widely in nature and being produced on a large scale in industry. Acetaldehyde occurs naturally in coffee, bread, and ripe fruit, and is produced by plants. It is also produced by the partial oxidation of ethanol by the liver enzyme alcohol dehydrogenase and is a contributing cause of hangover after alcohol consumption. Pathways of exposure include air, water, land, or groundwater, as well as drink and smoke. Consumption of disulfiram inhibits acetaldehyde dehydrogenase, the enzyme responsible for the metabolism of acetaldehyde, thereby causing it to build up in the body.
Genotoxicity is the property of chemical agents that damage the genetic information within a cell causing mutations, which may lead to cancer. While genotoxicity is often confused with mutagenicity, all mutagens are genotoxic, but some genotoxic substances are not mutagenic. The alteration can have direct or indirect effects on the DNA: the induction of mutations, mistimed event activation, and direct DNA damage leading to mutations. The permanent, heritable changes can affect either somatic cells of the organism or germ cells to be passed on to future generations. Cells prevent expression of the genotoxic mutation by either DNA repair or apoptosis; however, the damage may not always be fixed leading to mutagenesis.

A polycyclic aromatic hydrocarbon (PAH) is a class of organic compounds that is composed of multiple aromatic rings. The simplest representative is naphthalene, having two aromatic rings, and the three-ring compounds anthracene and phenanthrene. PAHs are uncharged, non-polar and planar. Many are colorless. Many of them are found in coal and in oil deposits, and are also produced by the incomplete combustion of organic matter—for example, in engines and incinerators or when biomass burns in forest fires.

Exhaust gas or flue gas is emitted as a result of the combustion of fuels such as natural gas, gasoline (petrol), diesel fuel, fuel oil, biodiesel blends, or coal. According to the type of engine, it is discharged into the atmosphere through an exhaust pipe, flue gas stack, or propelling nozzle. It often disperses downwind in a pattern called an exhaust plume.

Bruce Nathan Ames is an American biochemist who is a professor of biochemistry and Molecular Biology Emeritus at the University of California, Berkeley, and was a senior scientist at Children's Hospital Oakland Research Institute (CHORI). Ames has made contributions to understanding the mechanisms of mutagenesis and DNA repair. He invented the Ames test, a widely used assay for easily and cheaply evaluating the mutagenicity of compounds. The test revolutionized the field of toxicology and has played a crucial role in identifying numerous environmental and industrial carcinogens.

Carbon black is a material produced by the incomplete combustion of coal tar, vegetable matter, or petroleum products, including fuel oil, fluid catalytic cracking tar, and ethylene cracking in a limited supply of air. Carbon black is a form of paracrystalline carbon that has a high surface-area-to-volume ratio, albeit lower than that of activated carbon. It is dissimilar to soot in its much higher surface-area-to-volume ratio and significantly lower polycyclic aromatic hydrocarbon (PAH) content.

Benzo[a]pyrene (BaP or B[a]P) is a polycyclic aromatic hydrocarbon and the result of incomplete combustion of organic matter at temperatures between 300 °C (572 °F) and 600 °C (1,112 °F). The ubiquitous compound can be found in coal tar, tobacco smoke and many foods, especially grilled meats. The substance with the formula C20H12 is one of the benzopyrenes, formed by a benzene ring fused to pyrene. Its diol epoxide metabolites, more commonly known as BPDE, react with and bind to DNA, resulting in mutations and eventually cancer. It is listed as a Group 1 carcinogen by the IARC. In the 18th century a scrotal cancer of chimney sweepers, the chimney sweeps' carcinoma, was already known to be connected to soot.
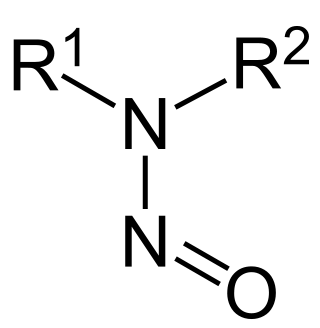
In organic chemistry, nitrosamines are organic compounds with the chemical structure R2N−N=O, where R is usually an alkyl group. They feature a nitroso group bonded to a deprotonated amine. Most nitrosamines are carcinogenic in nonhuman animals. A 2006 systematic review supports a "positive association between nitrite and nitrosamine intake and gastric cancer, between meat and processed meat intake and gastric cancer and oesophageal cancer, and between preserved fish, vegetable and smoked food intake and gastric cancer, but is not conclusive".

Chemical hazards are hazards present in hazardous chemicals and hazardous materials. Exposure to certain chemicals can cause acute or long-term adverse health effects. Chemical hazards are usually classified separately from biological hazards (biohazards). Chemical hazards are classified into groups that include asphyxiants, corrosives, irritants, sensitizers, carcinogens, mutagens, teratogens, reactants, and flammables. In the workplace, exposure to chemical hazards is a type of occupational hazard. The use of personal protective equipment may substantially reduce the risk of adverse health effects from contact with hazardous materials.

Diesel exhaust is the gaseous exhaust produced by a diesel type of internal combustion engine, plus any contained particulates. Its composition may vary with the fuel type or rate of consumption, or speed of engine operation, and whether the engine is in an on-road vehicle, farm vehicle, locomotive, marine vessel, or stationary generator or other application.
IARC group 1 Carcinogens are substances, chemical mixtures, and exposure circumstances which have been classified as carcinogenic to humans by the International Agency for Research on Cancer (IARC). This category is used when there is sufficient evidence of carcinogenicity in humans. Exceptionally, an agent may be placed in this category when evidence of carcinogenicity in humans is less than sufficient, but when there is sufficient evidence of carcinogenicity in experimental animals and strong evidence in exposed humans that the agent (mixture) acts through a relevant mechanism of carcinogenicity.

In molecular genetics, a DNA adduct is a segment of DNA bound to a cancer-causing chemical. This process could lead to the development of cancerous cells, or carcinogenesis. DNA adducts in scientific experiments are used as biomarkers of exposure. They are especially useful in quantifying an organism's exposure to a carcinogen. The presence of such an adduct indicates prior exposure to a potential carcinogen, but it does not necessarily indicate the presence of cancer in the subject animal.
Chemophobia is an aversion to or prejudice against chemicals or chemistry. The phenomenon has been ascribed both to a reasonable concern over the potential adverse effects of synthetic chemicals, and to an irrational fear of these substances because of misconceptions about their potential for harm, particularly the possibility of certain exposures to some synthetic chemicals elevating an individual's risk of cancer. Consumer products with labels such as "natural" and "chemical-free" appeal to chemophobic sentiments by offering consumers what appears to be a safer alternative.
A co-carcinogen is a chemical that promotes the effects of a carcinogen in the production of cancer. Usually, the term is used to refer to chemicals that are not carcinogenic on their own, such that an equivalent amount of the chemical is insufficient to initiate carcinogenesis. A chemical can be co-carcinogenic with other chemicals or with nonchemical carcinogens, such as UV radiation.

Dibenzopyrenes are a group of high molecular weight polycyclic aromatic hydrocarbons with the molecular formula C24H14. There are five isomers of dibenzopyrene which differ by the arrangement of aromatic rings: dibenzo[a,e]pyrene, dibenzo[a,h]pyrene, dibenzo[a,i]pyrene, dibenzo[a,l]pyrene, and dibenzo[e,l]pyrene.














