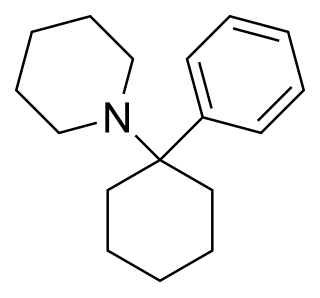
Phencyclidine or phenylcyclohexyl piperidine (PCP), also known in its use as a street drug as angel dust among other names, is a dissociative anesthetic mainly used recreationally for its significant mind-altering effects. PCP may cause hallucinations, distorted perceptions of sounds, and violent behavior. As a recreational drug, it is typically smoked, but may be taken by mouth, snorted, or injected. It may also be mixed with cannabis or tobacco.

1-(1-Phenylcyclohexyl)-4-hydroxypiperidine (PCHP) is a metabolite of phencyclidine (PCP). PCHP can be detected in the hair, urine, stool, sweat, and saliva of PCP users.

PCAA, or 5-[N-(1-phenylcyclohexyl)amino]pentanoic acid, is a metabolite of phencyclidine (PCP). It can be detected in the urine of PCP users by mass spectrometry as means of drug screening.
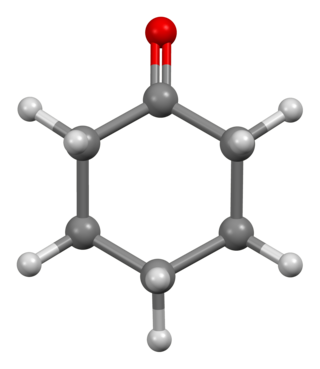
Cyclohexanone is the organic compound with the formula (CH2)5CO. The molecule consists of six-carbon cyclic molecule with a ketone functional group. This colorless oily liquid has a sweet odor reminiscent of benzaldehyde. Over time, samples of cyclohexanone assume a pale yellow color. Cyclohexanone is slightly soluble in water and miscible with common organic solvents. Millions of tonnes are produced annually, mainly as a precursor to nylon.

Dextromethorphan (DXM), sold under the trade name Robitussin among others, is a cough suppressant used in many cough and cold medicines. In 2022, the FDA approved the combination dextromethorphan/bupropion to serve as a rapid acting antidepressant in patients with major depressive disorder.

Rolicyclidine (PCPy) is a dissociative anesthetic that is similar in effects to phencyclidine, but is slightly less potent and has fewer stimulant effects. It instead produces a sedative effect described as being somewhat similar to a barbiturate, but with additional PCP-like dissociative, anaesthetic and hallucinogenic effects. Due to its similarity in effects to PCP, PCPy was placed into the Schedule I list of illegal drugs in the 1970s, although it has never been widely abused and is now little known.

Eticyclidine is a dissociative anesthetic drug with hallucinogenic effects. It is similar in effects to phencyclidine but is slightly more potent. PCE was developed by Parke-Davis in the 1970s and evaluated for anesthetic potential under the code name CI-400, but research into PCE was not continued after the development of ketamine, a similar drug with more favourable properties. Due to its similarity in effects to PCP, PCE was placed into the Schedule 1 list of illegal drugs in the 1970s, although it was only briefly abused in the 1970s and 1980s and is now little known.
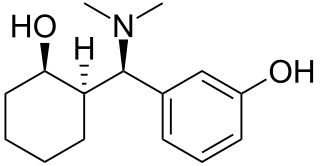
Ciramadol (WY-15,705) is an opioid analgesic that was developed in the late 1970s and is related to phencyclidine, tramadol, tapentadol and venlafaxine. It is a mixed agonist-antagonist for the μ-opioid receptor with relatively low abuse potential and a ceiling on respiratory depression which makes it a relatively safe drug. It has a slightly higher potency and effectiveness as an analgesic than codeine, but is weaker than morphine. Other side effects include sedation and nausea but these are generally less severe than with other similar drugs.

Norcocaine is a minor metabolite of cocaine. It is the only confirmed pharmacologically active metabolite of cocaine, although salicylmethylecgonine is also speculated to be an active metabolite. The local anesthetic potential of norcocaine has been shown to be higher than that of cocaine, however cocaine continues to be more widely used. Norcocaine used for research purposes is typically synthesized from cocaine. Several methods for the synthesis have been described.
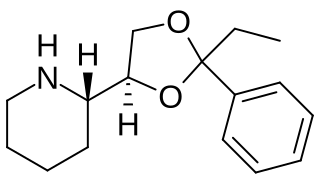
Etoxadrol (CL-1848C) is a dissociative anaesthetic drug that has been found to be an NMDA antagonist and produce similar effects to PCP in animals. Etoxadrol, along with another related drug dexoxadrol, were developed as analgesics for use in humans, but development was discontinued in the late 1970s after patients reported side effects such as nightmares and hallucinations.

Benocyclidine, also known as benzothiophenylcyclohexylpiperidine (BTCP), is a psychoactive recreational drug of the arylcyclohexylamine class which is related to phencyclidine (PCP). It was first described in a patent application naming Marc Caron and colleagues at Duke University in 1997.
The molecular formula C17H25NO may refer to:

Dieticyclidine (PCDE), or diethylphenylcyclohexylamine, is a psychoactive drug and research chemical of the arylcyclohexylamine chemical class related to phencyclidine (PCP) and eticyclidine (PCE). It acts as an NMDA receptor antagonist but has low potency and acts mainly as a prodrug for eticyclidine.

Arylcyclohexylamines, also known as arylcyclohexamines or arylcyclohexanamines, are a chemical class of pharmaceutical, designer, and experimental drugs.

3-Methoxyphencyclidine (3-MeO-PCP) is a dissociative hallucinogen of the arylcyclohexylamine class related to phencyclidine (PCP) which has been sold online as a designer drug. It has been used across Europe and the United States. In some cases, consumption has been known to be fatal. It acts mainly as an NMDA receptor antagonist, though it has also been found to interact with the sigma σ1 receptor and the serotonin transporter. The drug does not possess any opioid activity nor does it act as a dopamine reuptake inhibitor.

Metaphit is a research chemical that acts as an acylator of NMDARAn, sigma and DAT binding sites in the CNS. It is the m-isothiocyanate derivative of phencyclidine (PCP) and binds irreversibly to the PCP binding site on the NMDA receptor complex. However, later studies suggest the functionality of metaphit is mediated by sites not involved in PCP-induced passive avoidance deficit, and not related to the NMDA receptor complex. Metaphit was also shown to prevent d-amphetamine induced hyperactivity, while significantly depleting dopamine content in the nucleus accumbens. Metaphit was the first acylating ligand used to study the cocaine receptor. It is a structural isomer of the similar research compound fourphit, as it and metaphit both are isothiocyanate substituted derivatives of an analogous scaffold shared with PCP.

Leptophos (O-(4-bromo-2,5-dichlorophenyl) O-methyl phenylphosphonothioate) belongs to the organophosphates and at room temperature it is a stable white solid. It is also known as Phosvel, Abar and Vcs 506. Leptophos was primarily used as a pesticide and fungicide. for rice, cotton, fruit and vegetables until its use was discontinued in 1975 in USA, but still sold in South-Eastern Asia in 1981.

Ephenidine is a dissociative anesthetic that has been sold online as a designer drug. It is illegal in some countries as a structural isomer of the banned opioid drug lefetamine, but has been sold in countries where it is not yet banned.
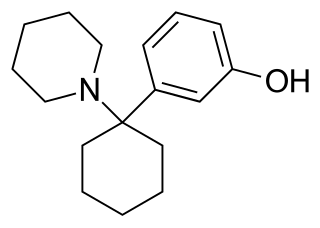
3-Hydroxyphencyclidine (3-HO-PCP) is a dissociative of the arylcyclohexylamine class related to phencyclidine (PCP) that has been sold online as a designer drug.


















