
A lithium-ion or Li-ion battery is a type of rechargeable battery that uses the reversible intercalation of Li+ ions into electronically conducting solids to store energy. In comparison with other rechargeable batteries, Li-ion batteries are characterized by higher specific energy, higher energy density, higher energy efficiency, a longer cycle life, and a longer calendar life. Also noteworthy is a dramatic improvement in lithium-ion battery properties after their market introduction in 1991: within the next 30 years, their volumetric energy density increased threefold while their cost dropped tenfold.

A rechargeable battery, storage battery, or secondary cell, is a type of electrical battery which can be charged, discharged into a load, and recharged many times, as opposed to a disposable or primary battery, which is supplied fully charged and discarded after use. It is composed of one or more electrochemical cells. The term "accumulator" is used as it accumulates and stores energy through a reversible electrochemical reaction. Rechargeable batteries are produced in many different shapes and sizes, ranging from button cells to megawatt systems connected to stabilize an electrical distribution network. Several different combinations of electrode materials and electrolytes are used, including lead–acid, zinc–air, nickel–cadmium (NiCd), nickel–metal hydride (NiMH), lithium-ion (Li-ion), lithium iron phosphate (LiFePO4), and lithium-ion polymer.

Grid energy storage is a collection of methods used for energy storage on a large scale within an electrical power grid. Electrical energy is stored during times when electricity is plentiful and inexpensive or when demand is low, and later returned to the grid when demand is high, and electricity prices tend to be higher.

Zinc–air batteries (non-rechargeable), and zinc–air fuel cells are metal–air batteries powered by oxidizing zinc with oxygen from the air. These batteries have high energy densities and are relatively inexpensive to produce. Sizes range from very small button cells for hearing aids, larger batteries used in film cameras that previously used mercury batteries, to very large batteries used for electric vehicle propulsion and grid-scale energy storage.
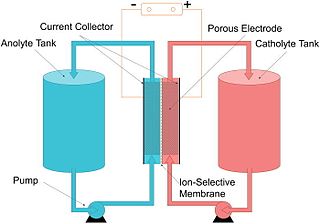
The vanadium redox battery (VRB), also known as the vanadium flow battery (VFB) or vanadium redox flow battery (VRFB), is a type of rechargeable flow battery. It employs vanadium ions as charge carriers. The battery uses vanadium's ability to exist in a solution in four different oxidation states to make a battery with a single electroactive element instead of two. For several reasons, including their relative bulkiness, vanadium batteries are typically used for grid energy storage, i.e., attached to power plants/electrical grids.

A flow battery, or redox flow battery, is a type of electrochemical cell where chemical energy is provided by two chemical components dissolved in liquids that are pumped through the system on separate sides of a membrane. Ion transfer inside the cell occurs through the membrane while both liquids circulate in their own respective space. Cell voltage is chemically determined by the Nernst equation and ranges, in practical applications, from 1.0 to 2.43 volts. The energy capacity is a function of the electrolyte volume and the power is a function of the surface area of the electrodes.
A zinc–bromine battery is a rechargeable battery system that uses the reaction between zinc metal and bromine to produce electric current, with an electrolyte composed of an aqueous solution of zinc bromide. Zinc has long been used as the negative electrode of primary cells. It is a widely available, relatively inexpensive metal. It is rather stable in contact with neutral and alkaline aqueous solutions. For this reason it is used today in zinc–carbon and alkaline primaries.

Molten-salt batteries are a class of battery that uses molten salts as an electrolyte and offers both a high energy density and a high power density. Traditional non-rechargeable thermal batteries can be stored in their solid state at room temperature for long periods of time before being activated by heating. Rechargeable liquid-metal batteries are used for industrial power backup, special electric vehicles and for grid energy storage, to balance out intermittent renewable power sources such as solar panels and wind turbines.

A123 Systems, LLC, a subsidiary of the Chinese Wanxiang Group Holdings, is a developer and manufacturer of lithium iron phosphate batteries and energy storage systems.

An electric vehicle battery is a rechargeable battery used to power the electric motors of a battery electric vehicle (BEV) or hybrid electric vehicle (HEV).
The lithium–air battery (Li–air) is a metal–air electrochemical cell or battery chemistry that uses oxidation of lithium at the anode and reduction of oxygen at the cathode to induce a current flow.
A metal–air electrochemical cell is an electrochemical cell that uses an anode made from pure metal and an external cathode of ambient air, typically with an aqueous or aprotic electrolyte.
A potassium-ion battery or K-ion battery is a type of battery and analogue to lithium-ion batteries, using potassium ions for charge transfer instead of lithium ions. It was invented by the Iranian/American chemist Ali Eftekhari in 2004.

Sodium-ion batteries (NIBs, SIBs, or Na-ion batteries) are several types of rechargeable batteries, which use sodium ions (Na+) as its charge carriers. In some cases, its working principle and cell construction are similar to those of lithium-ion battery (LIB) types, but it replaces lithium with sodium as the cathode material. Sodium belongs to the same group in the periodic table as lithium and thus has similar chemical properties. In other cases (such as aqueous Na-ion batteries) they are quite different from Li-ion batteries.

A supercapacitor (SC), also called an ultracapacitor, is a high-capacity capacitor, with a capacitance value much higher than solid-state capacitors but with lower voltage limits. It bridges the gap between electrolytic capacitors and rechargeable batteries. It typically stores 10 to 100 times more energy per unit volume or mass than electrolytic capacitors, can accept and deliver charge much faster than batteries, and tolerates many more charge and discharge cycles than rechargeable batteries.
Aluminium-ion batteries are a class of rechargeable battery in which aluminium ions serve as charge carriers. Aluminium can exchange three electrons per ion. This means that insertion of one Al3+ is equivalent to three Li+ ions. Thus, since the ionic radii of Al3+ (0.54 Å) and Li+ (0.76 Å) are similar, significantly higher numbers of electrons and Al3+ ions can be accepted by cathodes with little damage. Al has 50 times (23.5 megawatt-hours m-3) the energy density of Li and is even higher than coal.

NASICON is an acronym for sodium (Na) Super Ionic CONductor, which usually refers to a family of solids with the chemical formula Na1+xZr2SixP3−xO12, 0 < x < 3. In a broader sense, it is also used for similar compounds where Na, Zr and/or Si are replaced by isovalent elements. NASICON compounds have high ionic conductivities, on the order of 10−3 S/cm, which rival those of liquid electrolytes. They are caused by hopping of Na ions among interstitial sites of the NASICON crystal lattice.
A zinc-ion battery or Zn-ion battery (abbreviated as ZIB) uses zinc ions (Zn2+) as the charge carriers. Specifically, ZIBs utilize Zn as the anode, Zn-intercalating materials as the cathode, and a Zn-containing electrolyte. Generally, the term zinc-ion battery is reserved for rechargeable (secondary) batteries, which are sometimes also referred to as rechargeable zinc metal batteries (RZMB). Thus, ZIBs are different than non-rechargeable (primary) batteries which use zinc, such as alkaline or zinc–carbon batteries.

Search for the Super Battery: Discover the Powerful World of Batteries is a 2017 American documentary film about energy storage and how it may help provide an environmentally friendly, or green, future. The basic mechanism of batteries, including lithium-ion types, is described. The benefits and limitations of various batteries are also presented. Details of seeking a much safer, more powerful, longer-lasting and less expensive battery, a so-called "super battery", is discussed. The broad importance of energy storage devices, in mobile phones and automobiles, and in the overall electric grid system of the United States, is examined in detail.

Home energy storage devices store electricity locally, for later consumption. Electrochemical energy storage products, also known as "Battery Energy Storage System", at their heart are rechargeable batteries, typically based on lithium-ion or lead-acid controlled by computer with intelligent software to handle charging and discharging cycles. Companies are also developing smaller flow battery technology for home use. As a local energy storage technologies for home use, they are smaller relatives of battery-based grid energy storage and support the concept of distributed generation. When paired with on-site generation, they can virtually eliminate blackouts in an off-the-grid lifestyle.












