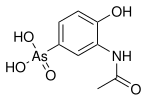
Arsonic acids are a subset of organoarsenic compounds defined as oxyacids where a pentavalent arsenic atom is bonded to two hydroxyl groups, a third oxygen atom (this one with a double bond), and an organic substituent. The salts/conjugate bases of arsonic acids are called arsonates. Like all arsenic-containing compounds, arsonic acids are toxic and carcinogenic to humans. [1] [2]
Contents
Arsonic acid refers to H 3 As O 3, the case where the substituent is a single hydrogen atom. The other arsonic acids can simply be viewed as hydrocarbyl derivatives of this base case. Arsenic acid results when the substituent is a hydroxyl group. Methylarsonic acid results when the substituent is a methyl group. Phenylarsonic acid results when the substituent is a phenyl group.