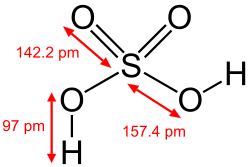
In chemistry, a hydroxy or hydroxyl group is a functional group with the chemical formula −OH and composed of one oxygen atom covalently bonded to one hydrogen atom. In organic chemistry, alcohols and carboxylic acids contain one or more hydroxy groups. Both the negatively charged anion HO−, called hydroxide, and the neutral radical HO·, known as the hydroxyl radical, consist of an unbonded hydroxy group.
Contents
- Properties
- Occurrence
- Hydroxyl radical
- Planetary observations
- Airglow of the Earth
- Surface of the Moon
- Atmosphere of Venus
- Atmosphere of Mars
- Exoplanets
- See also
- References
- Further
- External links
According to IUPAC definitions, the term hydroxyl refers to the hydroxyl radical (·OH) only, while the functional group −OH is called a hydroxy group. [1]