
Adenosine triphosphate (ATP) is an organic compound that provides energy to drive and support many processes in living cells, such as muscle contraction, nerve impulse propagation, condensate dissolution, and chemical synthesis. Found in all known forms of life, ATP is often referred to as the "molecular unit of currency" of intracellular energy transfer. When consumed in metabolic processes, it converts either to adenosine diphosphate (ADP) or to adenosine monophosphate (AMP). Other processes regenerate ATP. The human body recycles its own body weight equivalent in ATP each day. It is also a precursor to DNA and RNA, and is used as a coenzyme.

In chemistry, a phosphate is an anion, salt, functional group or ester derived from a phosphoric acid. It most commonly means orthophosphate, a derivative of orthophosphoric acid, aka. phosphoric acid H3PO4.

In biochemistry, phosphorylation is the attachment of a phosphate group to a molecule or an ion. This process and its inverse, dephosphorylation, are common in biology. Protein phosphorylation often activates many enzymes.

Threonine is an amino acid that is used in the biosynthesis of proteins. It contains an α-amino group, a carboxyl group, and a side chain containing a hydroxyl group, making it a polar, uncharged amino acid. It is essential in humans, meaning the body cannot synthesize it: it must be obtained from the diet. Threonine is synthesized from aspartate in bacteria such as E. coli. It is encoded by all the codons starting AC.

Adenosine diphosphate (ADP), also known as adenosine pyrophosphate (APP), is an important organic compound in metabolism and is essential to the flow of energy in living cells. ADP consists of three important structural components: a sugar backbone attached to adenine and two phosphate groups bonded to the 5 carbon atom of ribose. The diphosphate group of ADP is attached to the 5’ carbon of the sugar backbone, while the adenine attaches to the 1’ carbon.
An anaerobic organism or anaerobe is any organism that does not require molecular oxygen for growth. It may react negatively or even die if free oxygen is present. In contrast, an aerobic organism (aerobe) is an organism that requires an oxygenated environment. Anaerobes may be unicellular or multicellular. Most fungi are obligate aerobes, requiring oxygen to survive. However, some species, such as the Chytridiomycota that reside in the rumen of cattle, are obligate anaerobes; for these species, anaerobic respiration is used because oxygen will disrupt their metabolism or kill them. Deep waters of the ocean are a common anoxic environment.
A polyphosphate is a salt or ester of polymeric oxyanions formed from tetrahedral PO4 (phosphate) structural units linked together by sharing oxygen atoms. Polyphosphates can adopt linear or a cyclic ring structures. In biology, the polyphosphate esters ADP and ATP are involved in energy storage. A variety of polyphosphates find application in mineral sequestration in municipal waters, generally being present at 1 to 5 ppm. GTP, CTP, and UTP are also nucleotides important in the protein synthesis, lipid synthesis, and carbohydrate metabolism, respectively. Polyphosphates are also used as food additives, marked E452.

Butyric acid, also known under the systematic name butanoic acid, is a straight-chain alkyl carboxylic acid with the chemical formula CH3CH2CH2CO2H. It is an oily, colorless liquid with an unpleasant odor. Isobutyric acid is an isomer. Salts and esters of butyric acid are known as butyrates or butanoates. The acid does not occur widely in nature, but its esters are widespread. It is a common industrial chemical and an important component in the mammalian gut.

Butyramide is the amide of butyric acid. It has the molecular formula C3H7CONH2. It is a white solid that is freely soluble in water and ethanol, but slightly soluble in diethyl ether. At room temperature, butyramide is a crystalline solid and in contrast to butyric acid, it is devoid of an unpleasant, rancid smell.

β-Hydroxybutyric acid, also known as 3-hydroxybutyric acid or BHB, is an organic compound and a beta hydroxy acid with the chemical formula CH3CH(OH)CH2CO2H; its conjugate base is β-hydroxybutyrate, also known as 3-hydroxybutyrate. β-Hydroxybutyric acid is a chiral compound with two enantiomers: D-β-hydroxybutyric acid and L-β-hydroxybutyric acid. Its oxidized and polymeric derivatives occur widely in nature. In humans, D-β-hydroxybutyric acid is one of two primary endogenous agonists of hydroxycarboxylic acid receptor 2 (HCA2), a Gi/o-coupled G protein-coupled receptor (GPCR).

Crotonyl-coenzyme A is an intermediate in the fermentation of butyric acid, and in the metabolism of lysine and tryptophan. It is important in the metabolism of fatty acids and amino acids.

γ-Amino-β-hydroxybutyric acid (GABOB), also known as β-hydroxy-γ-aminobutyric acid (β-hydroxy-GABA), and sold under the brand name Gamibetal among others, is an anticonvulsant which is used for the treatment of epilepsy in Europe, Japan, and Mexico. It is a GABA analogue, or an analogue of the neurotransmitter γ-aminobutyric acid (GABA), and has been found to be an endogenous metabolite of GABA.
Butyrate—CoA ligase, also known as xenobiotic/medium-chain fatty acid-ligase (XM-ligase), is an enzyme that catalyzes the chemical reaction:

Butyryl-coenzyme A is the coenzyme A-containing derivative of butyric acid. It is acted upon by butyryl-CoA dehydrogenase and an intermediary compound of ABE fermentation.

In enzymology, a butyrate kinase is an enzyme that catalyzes the chemical reaction

Olfactory receptor 51E1 is a protein that in humans is encoded by the OR51E1 gene.

Perfluorobutanoic acid (PFBA) is a perfluoroalkyl carboxylic acid with the formula C3F7CO2H. As the perfluorinated derivative of butyric acid, this colourless liquid is prepared by electrofluorination of the corresponding butyryl fluoride.

Butyryl chloride is an organic compound with the chemical formula CH3CH2CH2C(O)Cl. It is a colorless liquid with a pungent odor. Butyryl chloride is soluble in aprotic organic solvents, but it reacts readily with water and alcohols. It is usually produced by chlorination of butyric acid.
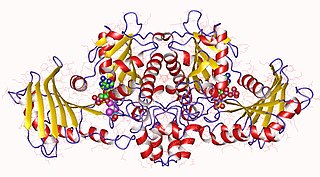
In molecular biology, acetate kinase (EC 2.7.2.1), which is predominantly found in micro-organisms, facilitates the production of acetyl-CoA by phosphorylating acetate in the presence of ATP and a divalent cation. Short-chain fatty acids (SCFAs) play a major role in carbon cycle and can be utilized as a source of carbon and energy by bacteria. Salmonella typhimurium propionate kinase (StTdcD) catalyzes reversible transfer of the γ-phosphate of ATP to propionate during l-threonine degradation to propionate. Kinetic analysis revealed that StTdcD possesses broad ligand specificity and could be activated by various SCFAs (propionate>acetate≈butyrate), nucleotides (ATP≈GTP>CTP≈TTP; dATP>dGTP>dCTP) and metal ions (Mg2+≈Mn2+>Co2+). Inhibition of StTdcD by tricarboxylic acid (TCA) cycle intermediates such as citrate, succinate, α-ketoglutarate and malate suggests that the enzyme could be under plausible feedback regulation. Crystal structures of StTdcD bound to PO4 (phosphate), AMP, ATP, Ap4 (adenosine tetraphosphate), GMP, GDP, GTP, CMP and CTP revealed that binding of nucleotide mainly involves hydrophobic interactions with the base moiety and could account for the broad biochemical specificity observed between the enzyme and nucleotides. Modelling and site-directed mutagenesis studies suggest Ala88 to be an important residue involved in determining the rate of catalysis with SCFA substrates. Molecular dynamics simulations on monomeric and dimeric forms of StTdcD revealed plausible open and closed states, and also suggested role for dimerization in stabilizing segment 235-290 involved in interfacial interactions and ligand binding. Observation of an ethylene glycol molecule bound sufficiently close to the γ-phosphate in StTdcD complexes with triphosphate nucleotides supports direct in-line phosphoryl transfer. The enzyme is important in the process of glycolysis, enzyme levels being increased in the presence of excess glucose. The growth of a bacterial mutant lacking acetate kinase has been shown to be inhibited by glucose, suggesting that the enzyme is involved in excretion of excess carbohydrate. A related enzyme, butyrate kinase, facilitates the formation of butyryl-CoA by phosphorylating butyrate in the presence of ATP to form butyryl phosphate.
















