A monomer is a molecule that can react together with other monomer molecules to form a larger polymer chain or three-dimensional network in a process called polymerization.

Polyethylene or polythene (abbreviated PE; IUPAC name polyethene or poly(methylene)) is the most commonly produced plastic. It is a polymer, primarily used for packaging (plastic bags, plastic films, geomembranes and containers including bottles, cups, jars, etc.). As of 2017, over 100 million tonnes of polyethylene resins are being produced annually, accounting for 34% of the total plastics market.

Polyacrylonitrile (PAN) is a synthetic, semicrystalline organic polymer resin, with the linear formula (CH2CHCN)n. Almost all PAN resins are copolymers with acrylonitrile as the main monomer. PAN is used to produce large variety of products including ultra filtration membranes, hollow fibers for reverse osmosis, fibers for textiles, and oxidized PAN fibers. PAN fibers are the chemical precursor of very high-quality carbon fiber. PAN is first thermally oxidized in air at 230 °C to form an oxidized PAN fiber and then carbonized above 1000 °C in inert atmosphere to make carbon fibers found in a variety of both high-tech and common daily applications such as civil and military aircraft primary and secondary structures, missiles, solid propellant rocket motors, pressure vessels, fishing rods, tennis rackets and bicycle frames. It is a component repeat unit in several important copolymers, such as styrene-acrylonitrile (SAN) and acrylonitrile butadiene styrene (ABS) plastic.
Acrylates are the salts, esters, and conjugate bases of acrylic acid. The acrylate ion is the anion CH2=CHCO−2. Often, acrylate refers to esters of acrylic acid, the most common member being methyl acrylate. These acrylates contain vinyl groups. These compounds are of interest because they are bifunctional: the vinyl group is susceptible to polymerization and the carboxylate group carries myriad functionalities.

Styrene-butadiene or styrene-butadiene rubber (SBR) describe families of synthetic rubbers derived from styrene and butadiene. These materials have good abrasion resistance and good aging stability when protected by additives. In 2012, more than 5.4 million tonnes of SBR were processed worldwide. About 50% of car tires are made from various types of SBR. The styrene/butadiene ratio influences the properties of the polymer: with high styrene content, the rubbers are harder and less rubbery. SBR is not to be confused with the thermoplastic elastomer, styrene-butadiene block copolymer, although being derived from the same monomers.

In polymer chemistry, a copolymer is a polymer derived from more than one species of monomer. The polymerization of monomers into copolymers is called copolymerization. Copolymers obtained from the copolymerization of two monomer species are sometimes called bipolymers. Those obtained from three and four monomers are called terpolymers and quaterpolymers, respectively. Copolymers can be characterized by a variety of techniques such as NMR spectroscopy and size-exclusion chromatography to determine the molecular size, weight, properties, and composition of the material.
In polymer chemistry, vinyl polymers are a group of polymers derived from substituted vinyl monomers. Their backbone is an extended alkane chain [−CH2−CHR−]. In popular usage, "vinyl" refers only to polyvinyl chloride (PVC).
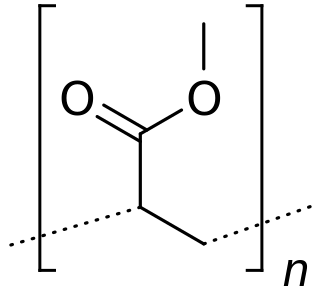
Poly(methyl acrylate) (PMA) is a family of organic polymers with the formula (CH2CHCO2CH3)n. It is a synthetic acrylate polymer derived from methyl acrylate monomer. The polymers are colorless. This homopolymer is far less important than copolymers derived from methyl acrylate and other monomers. PMA is softer than polymethyl methacrylate (PMMA), It is tough, leathery, and flexible.
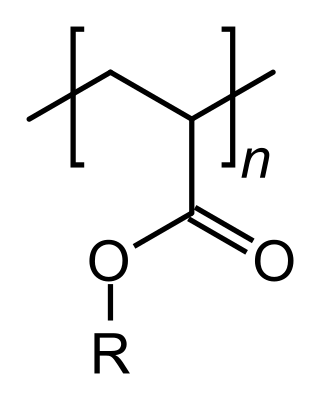
An acrylate polymer is any of a group of polymers prepared from acrylate monomers. These plastics are noted for their transparency, resistance to breakage, and elasticity.
1,1-Dichloroethylene, commonly called vinylidene chloride or 1,1-DCE, is an organochloride with the molecular formula CHCl2CH3. It is a colorless liquid with a sharp odor. Like most chlorocarbons, it is poorly soluble in water but soluble in organic solvents. 1,1-DCE was the precursor to the original clingwrap, Saran, for food, but this application has been phased out.
Synthetic resins are industrially produced resins, typically viscous substances that convert into rigid polymers by the process of curing. In order to undergo curing, resins typically contain reactive end groups, such as acrylates or epoxides. Some synthetic resins have properties similar to natural plant resins, but many do not.

Ethyl acrylate is an organic compound with the formula CH2CHCO2CH2CH3. It is the ethyl ester of acrylic acid. It is a colourless liquid with a characteristic acrid odor. It is mainly produced for paints, textiles, and non-woven fibers. It is also a reagent in the synthesis of various pharmaceutical intermediates.

Methyl acrylate is an organic compound, more accurately the methyl ester of acrylic acid. It is a colourless liquid with a characteristic acrid odor. It is mainly produced to make acrylate fiber, which is used to weave synthetic carpets. It is also a reagent in the synthesis of various pharmaceutical intermediates. Owing to the tendency of methyl acrylate to polymerize, samples typically contain an inhibitor such as hydroquinone.

Styrene acrylonitrile resin (SAN) is a copolymer plastic consisting of styrene and acrylonitrile. It is widely used in place of polystyrene owing to its greater thermal resistance. The chains of between 70 and 80% by weight styrene and 20 to 30% acrylonitrile. Larger acrylonitrile content improves mechanical properties and chemical resistance, but also adds a yellow tint to the normally transparent plastic.
Styrene maleic anhydride is a synthetic polymer that is built-up of styrene and maleic anhydride monomers. In one copolymer, the monomers can be almost perfectly alternating. but (random) copolymerisation with less than 50% maleic anhydride content is also possible. The polymer is formed by a radical polymerization, using an organic peroxide as the initiator. The main characteristics of SMA copolymer are its transparent appearance, high heat resistance, high dimensional stability, and the specific reactivity of the anhydride groups. The latter feature results in the solubility of SMA in alkaline (water-based) solutions and dispersion.
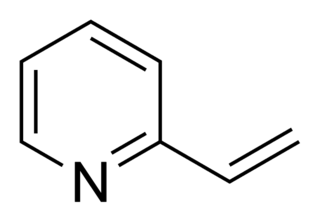
2-Vinylpyridine is an organic compound with the formula CH2CHC5H4N. It is a derivative of pyridine with a vinyl group in the 2-position, next to the nitrogen. It is a colorless liquid, although samples are often brown. It is used industrially as a precursor to specialty polymers and as an intermediate in the chemical, pharmaceutical, dye, and photo industries. Vinylpyridine is sensitive to polymerization. It may be stabilized with a polymerisation inhibitor such as tert-butylcatechol. Owing to its tendency to polymerize, samples are typically refrigerated.

4-Vinylbenzyl chloride is an organic compound with the formula ClCH2C6H4CH=CH2. It is a bifunctional molecule, featuring both vinyl and a benzylic chloride functional groups. It is a colorless liquid that is typically stored with a stabilizer to suppress polymerization.

Vinyl propionate is the organic compound with the formula CH3CH2CO2CH=CH2. This colorless liquid is the ester of propionic acid and vinyl alcohol. It is used to produce poly(vinyl propionate) as well as copolymers with acrylate esters, vinyl chloride, and vinyl acetate, some of which are used in paints. The compound resembles vinyl acetate.

Poly(ethyl acrylate) (PEA) is a family of organic polymers with the formula (CH2CHCO2CH2CH3)n. It is a synthetic acrylate polymer derived from ethyl acrylate monomer. The polymers are colorless. This homopolymer is far less important than copolymers derived from ethyl acrylate and other monomers. It has a low glass-transition temperature about -8 °C (20 °C).

Poly(butyl acrylate) (PBA) is a family of organic polymers with the formula (CH2CHCO2CH2CH2CH2CH3)n. It is a synthetic acrylate polymer derived from butyl acrylate monomer. The polymers are colorless. This homopolymer is far less important than copolymers derived from methyl acrylate and other monomers. It has a low glass-transition temperature of about -43 °C (20 °C).















