
Lignin is a class of complex organic polymers that form key structural materials in the support tissues of most plants. Lignins are particularly important in the formation of cell walls, especially in wood and bark, because they lend rigidity and do not rot easily. Chemically, lignins are polymers made by cross-linking phenolic precursors.

Eugenol is an allyl chain-substituted guaiacol, a member of the allylbenzene class of chemical compounds. It is a colorless to pale yellow, aromatic oily liquid extracted from certain essential oils especially from clove, nutmeg, cinnamon, basil and bay leaf. It is present in concentrations of 80–90% in clove bud oil and at 82–88% in clove leaf oil. Eugenol has a pleasant, spicy, clove-like scent. The name is derived from Eugenia caryophyllata, the former Linnean nomenclature term for cloves. The currently accepted name is Syzygium aromaticum.
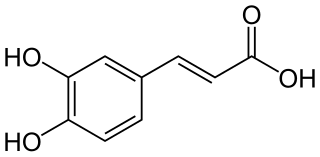
Caffeic acid is an organic compound that is classified as a hydroxycinnamic acid. This yellow solid consists of both phenolic and acrylic functional groups. It is found in all plants because it is an intermediate in the biosynthesis of lignin, one of the principal components of woody plant biomass and its residues.
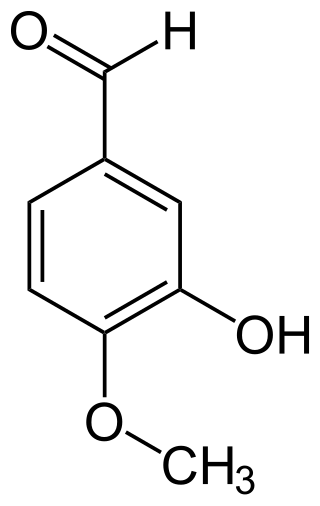
Isovanillin is a phenolic aldehyde, an organic compound and isomer of vanillin. It is a selective inhibitor of aldehyde oxidase. It is not a substrate of that enzyme, and is metabolized by aldehyde dehydrogenase into isovanillic acid, which could make it a candidate drug for use in alcohol aversion therapy. Isovanillin can be used as a precursor in the chemical total synthesis of morphine. The proposed metabolism of isovanillin in rat has been described in literature, and is part of the WikiPathways machine readable pathway collection.
Guaiacol is an organic compound with the formula C6H4(OH)(OCH3). It is a phenolic compound containing a methoxy functional group. Guaiacol appears as a viscous colorless oil, although aged or impure samples are often yellowish. It occurs widely in nature and is a common product of the pyrolysis of wood.

4-Hydroxybenzoic acid, also known as p-hydroxybenzoic acid (PHBA), is a monohydroxybenzoic acid, a phenolic derivative of benzoic acid. It is a white crystalline solid that is slightly soluble in water and chloroform but more soluble in polar organic solvents such as alcohols and acetone. 4-Hydroxybenzoic acid is primarily known as the basis for the preparation of its esters, known as parabens, which are used as preservatives in cosmetics and some ophthalmic solutions. It is isomeric with 2-hydroxybenzoic acid, known as salicylic acid, a precursor to aspirin, and with 3-hydroxybenzoic acid.

Coniferyl alcohol is an organic compound with the formula HO(CH3O)C6H3CH=CHCH2OH. A colourless or white solid, it is one of the monolignols, produced via the phenylpropanoid biochemical pathway. When copolymerized with related aromatic compounds, coniferyl alcohol forms lignin or lignans. Coniferin is a glucoside of coniferyl alcohol. Coniferyl alcohol is an intermediate in biosynthesis of eugenol and of stilbenoids and coumarin. Gum benzoin contains significant amount of coniferyl alcohol and its esters. It is found in both gymnosperm and angiosperm plants. Sinapyl alcohol and paracoumaryl alcohol, the other two lignin monomers, are found in angiosperm plants and grasses.

Dirigent proteins are members of a class of proteins which dictate the stereochemistry of a compound synthesized by other enzymes. The first dirigent protein was discovered in Forsythia intermedia. This protein has been found to direct the stereoselective biosynthesis of (+)-pinoresinol from coniferyl alcohol monomers:
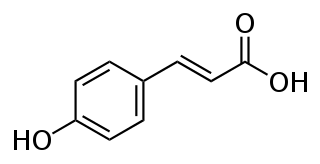
p-Coumaric acid is an organic compound with the formula HOC6H4CH=CHCO2H. It is one of the three isomers of hydroxycinnamic acid. It is a white solid that is only slightly soluble in water but very soluble in ethanol and diethyl ether.

The phenylpropanoids are a diverse family of organic compounds that are biosynthesized by plants from the amino acids phenylalanine and tyrosine in the shikimic acid pathway. Their name is derived from the six-carbon, aromatic phenyl group and the three-carbon propene tail of coumaric acid, which is the central intermediate in phenylpropanoid biosynthesis. From 4-coumaroyl-CoA emanates the biosynthesis of myriad natural products including lignols, flavonoids, isoflavonoids, coumarins, aurones, stilbenes, catechin, and phenylpropanoids. The coumaroyl component is produced from cinnamic acid.

Syringol is the organic compound with the formula HO(CH3O)2C6H3. The molecule is a phenol, with methoxy groups in the flanking (2 and 6) positions. It is the symmetrically dimethylated derivative of pyrogallol. It is a colorless solid, although typical samples are brown owing to air-oxidized impurities. Together with guaiacol, syringol and its derivatives are produced by the pyrolysis of lignin. Specifically, syringol is derived from the thermal decomposition of the sinapyl alcohol component. As such, syringol is an important component of wood smoke.

Monolignols, also called lignols, are the source materials for biosynthesis of both lignans and lignin and consist mainly of paracoumaryl alcohol (H), coniferyl alcohol (G) and sinapyl alcohol (S). These monolignols differ in their degree of methoxilation of the aromatic ring.

Sinapyl alcohol is an organic compound structurally related to cinnamic acid. It is biosynthetized via the phenylpropanoid biochemical pathway, its immediate precursor being sinapaldehyde. This phytochemical is one of the monolignols, which are precursor to lignin or lignans. It is also a biosynthetic precursor to various stilbenoids and coumarins.
In enzymology, a dihydrokaempferol 4-reductase (EC 1.1.1.219) is an enzyme that catalyzes the chemical reaction

Cinnamoyl-CoA reductase (EC 1.2.1.44), systematically named cinnamaldehyde:NADP+ oxidoreductase (CoA-cinnamoylating) but commonly referred to by the acronym CCR, is an enzyme that catalyzes the reduction of a substituted cinnamoyl-CoA to its corresponding cinnamaldehyde, utilizing NADPH and H+ and releasing free CoA and NADP+ in the process. Common biologically relevant cinnamoyl-CoA substrates for CCR include p-coumaroyl-CoA and feruloyl-CoA, which are converted into p-coumaraldehyde and coniferaldehyde, respectively, though most CCRs show activity toward a variety of other substituted cinnamoyl-CoA's as well. Catalyzing the first committed step in monolignol biosynthesis, this enzyme plays a critical role in lignin formation, a process important in plants both for structural development and defense response.

ortho-Vanillin (2-hydroxy-3-methoxybenzaldehyde) is an organic solid present in the extracts and essential oils of many plants. Its functional groups include aldehyde, ether and phenol. ortho-Vanillin, a compound of the formula C8H8O3, is distinctly different from its more prevalent isomer, meta-vanillin. The "ortho-" prefix refers to the position of the compound’s hydroxyl moiety, which is found in the para-position in vanillin.
Caffeyl alcohol is the organic compound with the formula (HO)2C6H3-4-CHCHCH2OH. This colourless solid is related to catechol by attachment to allyl alcohol. It is the precursor to one of the three principal lignols.

Absinthin is a naturally produced triterpene lactone from the plant Artemisia absinthium (Wormwood). It constitutes one of the most bitter chemical agents responsible for absinthe's distinct taste. The compound shows biological activity and has shown promise as an anti-inflammatory agent, and should not be confused with thujone, a neurotoxin also found in Artemisia absinthium.

Sinapaldehyde is an organic compound with the formula HO(CH3O)2C6H2CH=CHCHO. It is a derivative of cinnamaldehyde, featuring one hydroxy group and two methoxy groups as substituents. It is an intermediate in the formation of sinapyl alcohol, a lignol that is a major precursor to lignin.
Chanoclavine-I dehydrogenase (EC 1.1.1.332, easD (gene), fgaDH (gene)) is an enzyme with systematic name chanoclavine-I:NAD+ oxidoreductase. This enzyme catalises the following chemical reaction
















