Mold health issues refer to the harmful health effects of molds and their mycotoxins. Approximately 47% of houses in the United States have substantial levels of mold, with over 85% of commercial and office buildings found to have water damage predictive of mold. As many as 21% of asthma cases may result from exposure to mold. Substantial and statistically significant increases in the risks of both respiratory infections and bronchitis have been associated with dampness in homes and the resulting mold.

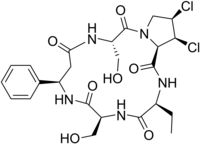
Microcystins—or cyanoginosins—are a class of toxins produced by certain freshwater cyanobacteria, commonly known as blue-green algae. Over 250 different microcystins have been discovered so far, of which microcystin-LR is the most common. Chemically they are cyclic heptapeptides produced through nonribosomal peptide synthases.
A mycotoxin is a toxic secondary metabolite produced by fungi and is capable of causing disease and death in both humans and other animals. The term 'mycotoxin' is usually reserved for the toxic chemical products produced by fungi that readily colonize crops.
Nonribosomal peptides (NRP) are a class of peptide secondary metabolites, usually produced by microorganisms like bacteria and fungi. Nonribosomal peptides are also found in higher organisms, such as nudibranchs, but are thought to be made by bacteria inside these organisms. While there exist a wide range of peptides that are not synthesized by ribosomes, the term nonribosomal peptide typically refers to a very specific set of these as discussed in this article.

Penicillium roqueforti is a common saprotrophic fungus in the genus Penicillium. Widespread in nature, it can be isolated from soil, decaying organic matter, and plants.
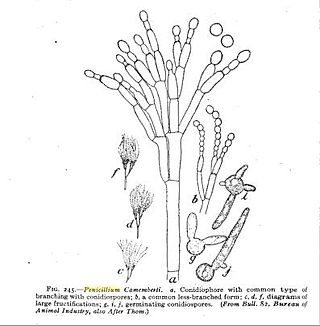
Penicillium camemberti is a species of fungus in the genus Penicillium. It is used in the production of Camembert, Brie, Langres, Coulommiers, and Cambozola cheeses, on which colonies of P. camemberti form a hard, white crust. It is responsible for giving these cheeses their distinctive flavors. An allergy to the antibiotic penicillin does not necessarily imply an allergy to cheeses made using P. camemberti.
Amatoxin is the collective name of a subgroup of at least nine related cyclic peptide toxins found in three genera of deadly poisonous mushrooms and one species of the genus Pholiotina. Amatoxins are very potent, as little as half a mushroom cap can cause severe liver injury if swallowed.

The trichothecenes are a large family of chemically related mycotoxins. They are produced by various species of Fusarium, Myrothecium,Trichoderma/Podostroma, Trichothecium, Cephalosporium, Verticimonosporium, and Stachybotrys. Chemically, trichothecenes are a class of sesquiterpenes.
Mycotoxicology is the branch of mycology that focuses on analyzing and studying the toxins produced by fungi, known as mycotoxins. In the food industry it is important to adopt measures that keep mycotoxin levels as low as practicable, especially those that are heat-stable. These chemical compounds are the result of secondary metabolism initiated in response to specific developmental or environmental signals. This includes biological stress from the environment, such as lower nutrients or competition for those available. Under this secondary path the fungus produces a wide array of compounds in order to gain some level of advantage, such as incrementing the efficiency of metabolic processes to gain more energy from less food, or attacking other microorganisms and being able to use their remains as a food source.
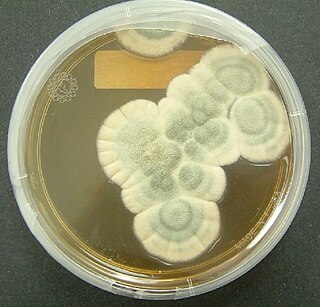
Penicillium chrysogenum is a species of fungus in the genus Penicillium. It is common in temperate and subtropical regions and can be found on salted food products, but it is mostly found in indoor environments, especially in damp or water-damaged buildings. It has been recognised as a species complex that includes P. notatum, P. meleagrinum, and P. cyaneofulvum. Molecular phylogeny has established that Alexander Fleming's first discovered penicillin producing strain is of a distinct species, P. rubens, and not of P. notatum. It has rarely been reported as a cause of human disease. It is the source of several β-lactam antibiotics, most significantly penicillin. Other secondary metabolites of P. chrysogenum include roquefortine C, meleagrin, chrysogine, 6-MSA YWA1/melanin, andrastatin A, fungisporin, secalonic acids, sorbicillin, and PR-toxin.
Penicillium islandicum is an anamorph species of the genus of Penicillium which produces luteoskyrin, simatoxin, cyclochlorotine (islanditoxin), rugulosin and chitosanase.
Yellowed rice refers to three kinds of rice grains contaminated with different strains of Penicillium fungi—Yellow rice, Citrinum yellow rice, and Islandia yellow rice. These rice grains were first identified in Japan in 1964, after the research was interrupted by World War II. The first of the yellowed rice strains has been linked to shoshin-kakke. Citrinum yellow rice and Islandia yellow rice are not known to have caused any adverse effects in human populations.
Penicillium nordicum is an anamorph species of fungus in the genus Penicillium which produces ochratoxin A. Penicillium nordicum contaminates protein rich foods and foods with high NaCl-konzentration. It is mostly found on dry-cured meat products and cheese products
Penicillium paxilli is an anamorph, saprophytic species of the genus Penicillium which produces paxilline, paxisterol, penicillone, pyrenocine A, paspaline B and verruculogene. Penicillium paxilli is used as a model to study the biochemistry of the indol-diterepene biosynthesis
Penicillium rugulosum is an anamorph species of fungus in the genus Penicillium which produces inulinase, luteoskyrin and (+) rugulosin.
Penicillium verrucosum is a psychrophilic fungus which was discovered in Belgium and introduced by Dierckx in 1901. Six varieties of this species have been recognized based primarily on differences in colony colour: P. verrucosum var. album, P. verrucosum var. corymbiferum, P. verrucosum var. cyclopium, P. verrucosum var. ochraceum, P. verrucosum var. melanochlorum and P. verrucosum var. verrucosum. This fungus has important implications in food, specifically for grains and other cereal crops on which it grows. Its growth is carefully regulated in order to reduce food spoilage by this fungi and its toxic products. The genome of P. verrucosum has been sequenced and the gene clusters for the biosyntheses of its mycotoxins have been identified.
Xeromyces is a monotypic genus of fungus in the family Monascaceae. Its only species, Xeromyces bisporus, was first described by L.R. Fraser in 1954. No subspecies are listed in the Catalogue of Life.

Penicillin Roquefort toxin is a mycotoxin produced by the fungus Penicillium roqueforti. In 1973, PR toxin was first partially characterized by isolating moldy corn on which the fungi had grown. Although its lethal dose was determined shortly after the isolation of the chemical, details of its toxic effects were not fully clarified until 1982 in a study with mice, rats, anesthetized cats and preparations of isolated rat auricles.

Luteoskyrin is a carcinogenic mycotoxin with the molecular formula C30H22O12 which is produced by the mold Penicillium islandicum. Luteoskyrin has strong cytotoxic effects. Luteoskyrin can cause the yellow rice disease.
John Ingram Pitt was an Australian mycologist, known as a leading expert on the role of fungi in food spoilage. He gained an international reputation as a pioneering researcher on the ecology of spoilage molds in extreme environments.