In nuclear physics, a nuclear chain reaction occurs when one single nuclear reaction causes an average of one or more subsequent nuclear reactions, thus leading to the possibility of a self-propagating series or "positive feedback loop" of these reactions. The specific nuclear reaction may be the fission of heavy isotopes. A nuclear chain reaction releases several million times more energy per reaction than any chemical reaction.

A nuclear reactor is a device used to initiate and control a fission nuclear chain reaction or nuclear fusion reaction. Nuclear reactors are used at nuclear power plants for electricity generation and in nuclear marine propulsion. When a fissile nucleus like uranium-235 or plutonium-239 absorbs a neutron, it splits into lighter nuclei, releasing energy, gamma radiation, and free neutrons, which can induce further fission in a self-sustaining chain reaction. The process is carefully controlled using control rods and neutron moderators to regulate the number of neutrons that continue the reaction, ensuring the reactor operates safely.

Radioactive waste is a type of hazardous waste that contains radioactive material. Radioactive waste is a result of many activities, including nuclear medicine, nuclear research, nuclear power generation, nuclear decommissioning, rare-earth mining, and nuclear weapons reprocessing. The storage and disposal of radioactive waste is regulated by government agencies in order to protect human health and the environment.
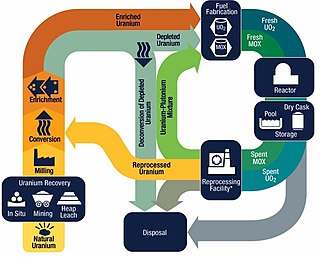
The nuclear fuel cycle, also called nuclear fuel chain, is the progression of nuclear fuel through a series of differing stages. It consists of steps in the front end, which are the preparation of the fuel, steps in the service period in which the fuel is used during reactor operation, and steps in the back end, which are necessary to safely manage, contain, and either reprocess or dispose of spent nuclear fuel. If spent fuel is not reprocessed, the fuel cycle is referred to as an open fuel cycle ; if the spent fuel is reprocessed, it is referred to as a closed fuel cycle.

A radioisotope thermoelectric generator, sometimes referred to as a radioisotope power system (RPS), is a type of nuclear battery that uses an array of thermocouples to convert the heat released by the decay of a suitable radioactive material into electricity by the Seebeck effect. This type of generator has no moving parts and is ideal for deployment in remote and harsh environments for extended periods with no risk of parts wearing out or malfunctioning.

Nuclear fission products are the atomic fragments left after a large atomic nucleus undergoes nuclear fission. Typically, a large nucleus like that of uranium fissions by splitting into two smaller nuclei, along with a few neutrons, the release of heat energy, and gamma rays. The two smaller nuclei are the fission products..

A loss-of-coolant accident (LOCA) is a mode of failure for a nuclear reactor; if not managed effectively, the results of a LOCA could result in reactor core damage. Each nuclear plant's emergency core cooling system (ECCS) exists specifically to deal with a LOCA.
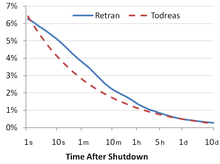
In nuclear engineering, a delayed neutron is a neutron emitted after a nuclear fission event, by one of the fission products, any time from a few milliseconds to a few minutes after the fission event. Neutrons born within 10−14 seconds of the fission are termed "prompt neutrons".

The integral fast reactor (IFR), originally the advancedliquid-metal reactor (ALMR), is a design for a nuclear reactor using fast neutrons and no neutron moderator. IFRs can breed more fuel and are distinguished by a nuclear fuel cycle that uses reprocessing via electrorefining at the reactor site.
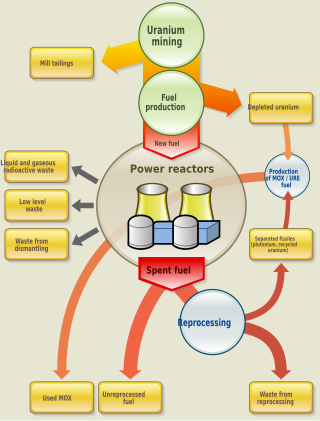
Nuclear fuel is material used in nuclear power stations to produce heat to power turbines. Heat is created when nuclear fuel undergoes nuclear fission. Nuclear fuel has the highest energy density of all practical fuel sources. The processes involved in mining, refining, purifying, using, and disposing of nuclear fuel are collectively known as the nuclear fuel cycle.

Plutonium-239 is an isotope of plutonium. Plutonium-239 is the primary fissile isotope used for the production of nuclear weapons, although uranium-235 is also used for that purpose. Plutonium-239 is also one of the three main isotopes demonstrated usable as fuel in thermal spectrum nuclear reactors, along with uranium-235 and uranium-233. Plutonium-239 has a half-life of 24,110 years.
Caesium (55Cs) has 41 known isotopes, the atomic masses of these isotopes range from 112 to 152. Only one isotope, 133Cs, is stable. The longest-lived radioisotopes are 135Cs with a half-life of 1.33 million years, 137
Cs
with a half-life of 30.1671 years and 134Cs with a half-life of 2.0652 years. All other isotopes have half-lives less than 2 weeks, most under an hour.
Nuclear reactor physics is the field of physics that studies and deals with the applied study and engineering applications of chain reaction to induce a controlled rate of fission in a nuclear reactor for the production of energy. Most nuclear reactors use a chain reaction to induce a controlled rate of nuclear fission in fissile material, releasing both energy and free neutrons. A reactor consists of an assembly of nuclear fuel, usually surrounded by a neutron moderator such as regular water, heavy water, graphite, or zirconium hydride, and fitted with mechanisms such as control rods which control the rate of the reaction.

The thorium fuel cycle is a nuclear fuel cycle that uses an isotope of thorium, 232
Th
, as the fertile material. In the reactor, 232
Th
is transmuted into the fissile artificial uranium isotope 233
U
which is the nuclear fuel. Unlike natural uranium, natural thorium contains only trace amounts of fissile material, which are insufficient to initiate a nuclear chain reaction. Additional fissile material or another neutron source is necessary to initiate the fuel cycle. In a thorium-fuelled reactor, 232
Th
absorbs neutrons to produce 233
U
. This parallels the process in uranium breeder reactors whereby fertile 238
U
absorbs neutrons to form fissile 239
Pu
. Depending on the design of the reactor and fuel cycle, the generated 233
U
either fissions in situ or is chemically separated from the used nuclear fuel and formed into new nuclear fuel.

Spent nuclear fuel, occasionally called used nuclear fuel, is nuclear fuel that has been irradiated in a nuclear reactor. It is no longer useful in sustaining a nuclear reaction in an ordinary thermal reactor and, depending on its point along the nuclear fuel cycle, it will have different isotopic constituents than when it started.

This page discusses each of the main elements in the mixture of fission products produced by nuclear fission of the common nuclear fuels uranium and plutonium. The isotopes are listed by element, in order by atomic number.
Xenon-135 (135Xe) is an unstable isotope of xenon with a half-life of about 9.2 hours. 135Xe is a fission product of uranium and it is the most powerful known neutron-absorbing nuclear poison, with a significant effect on nuclear reactor operation. The ultimate yield of xenon-135 from fission is 6.3%, though most of this is from fission-produced tellurium-135 and iodine-135.
In nuclear power technology, burnup is a measure of how much energy is extracted from a primary nuclear fuel source. It is measured as the fraction of fuel atoms that underwent fission in %FIMA or %FIFA as well as, preferably, the actual energy released per mass of initial fuel in gigawatt-days/metric ton of heavy metal (GWd/tHM), or similar units.
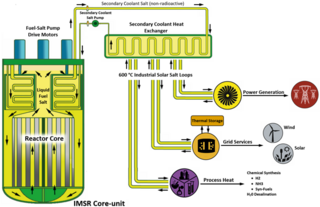
The integral molten salt reactor (IMSR) is a nuclear power plant design targeted at developing a commercial product for the small modular reactor (SMR) market. It employs molten salt reactor technology which is being developed by the Canadian company Terrestrial Energy.

The Stable Salt Reactor (SSR) is a nuclear reactor design under development by Moltex Energy Canada Inc. and its subsidiary Moltex Energy USA LLC, based in Canada, the United States, and the United Kingdom, as well as MoltexFLEX Ltd., based in the United Kingdom.