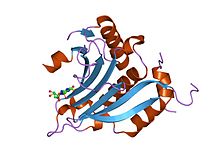
The SRC Homology 3 Domain is a small protein domain of about 60 amino acid residues. Initially, SH3 was described as a conserved sequence in the viral adaptor protein v-Crk. This domain is also present in the molecules of phospholipase and several cytoplasmic tyrosine kinases such as Abl and Src. It has also been identified in several other protein families such as: PI3 Kinase, Ras GTPase-activating protein, CDC24 and cdc25. SH3 domains are found in proteins of signaling pathways regulating the cytoskeleton, the Ras protein, and the Src kinase and many others. The SH3 proteins interact with adaptor proteins and tyrosine kinases. Interacting with tyrosine kinases, SH3 proteins usually bind far away from the active site. Approximately 300 SH3 domains are found in proteins encoded in the human genome. In addition to that, the SH3 domain was responsible for controlling protein-protein interactions in the signal transduction pathways and regulating the interactions of proteins involved in the cytoplasmic signaling.

14-3-3 proteins are a family of conserved regulatory molecules that are expressed in all eukaryotic cells. 14-3-3 proteins have the ability to bind a multitude of functionally diverse signaling proteins, including kinases, phosphatases, and transmembrane receptors. More than 200 signaling proteins have been reported as 14-3-3 ligands.
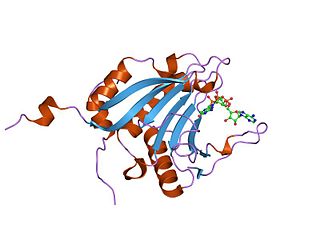
Eukaryotic translation initiation factor 4E-binding protein 1 is a protein that in humans is encoded by the EIF4EBP1 gene.

Polyadenylate-binding protein 1 is a protein that in humans is encoded by the PABPC1 gene. The protein PABP1 binds mRNA and facilitates a variety of functions such as transport out of the nucleus, degradation, translation, and stability. There are two separate PABP1 proteins, one which is located in the nucleus (PABPN1) and the other which is found in the cytoplasm (PABPC1). The location of PABP1 affects the role of that protein and its function with RNA.

Eukaryotic translation initiation factor 4E, also known as eIF4E, is a protein that in humans is encoded by the EIF4E gene.

Eukaryotic translation initiation factor 4 gamma 2 is a protein that in humans is encoded by the EIF4G2 gene.

Eukaryotic translation initiation factor 4 gamma 1 is a protein that in humans is encoded by the EIF4G1 gene.

Eukaryotic translation initiation factor 6 (EIF6), also known as Integrin beta 4 binding protein (ITGB4BP), is a human gene.

Eukaryotic translation initiation factor 2 subunit 2 (eIF2β) is a protein that in humans is encoded by the EIF2S2 gene.

Eukaryotic translation initiation factor 4 gamma 3 is a protein that in humans is encoded by the EIF4G3 gene. The gene encodes a protein that functions in translation by aiding the assembly of the ribosome onto the messenger RNA template. Confusingly, this protein is usually referred to as eIF4GII, as although EIF4G3 is the third gene that is similar to eukaryotic translation initiation factor 4 gamma, the second isoform EIF4G2 is not an active translation initiation factor.

Eukaryotic initiation factor 4A-I is a 46 kDa cytosolic protein that, in humans, is encoded by the EIF4A1 gene, which is located on chromosome 17. It is the most prevalent member of the eIF4A family of ATP-dependant RNA helicases, and plays a critical role in the initiation of cap-dependent eukaryotic protein translation as a component of the eIF4F translation initiation complex. eIF4A1 unwinds the secondary structure of RNA within the 5'-UTR of mRNA, a critical step necessary for the recruitment of the 43S preinitiation complex, and thus the translation of protein in eukaryotes. It was first characterized in 1982 by Grifo, et al., who purified it from rabbit reticulocyte lysate.

Eukaryotic translation initiation factor 4B is a protein that in humans is encoded by the EIF4B gene.

Eukaryotic initiation factor 4A-III is a protein that in humans is encoded by the EIF4A3 gene.

Eukaryotic translation initiation factor 4E type 2 is a protein that in humans is encoded by the EIF4E2 gene. It belongs to the eukaryotic translation initiation factor 4E family.

Eukaryotic translation initiation factor 1 (eIF1) is a protein that in humans is encoded by the EIF1 gene. It is related to yeast SUI1.
Eukaryotic translation initiation factor 4 G (eIF4G) is a protein involved in eukaryotic translation initiation and is a component of the eIF4F cap-binding complex. Orthologs of eIF4G have been studied in multiple species, including humans, yeast, and wheat. However, eIF4G is exclusively found in domain Eukarya, and not in domains Bacteria or Archaea, which do not have capped mRNA. As such, eIF4G structure and function may vary between species, although the human eIF4G 1 has been the focus of extensive studies.
Eukaryotic Initiation Factor 2 (eIF2) is a eukaryotic initiation factor. It is required for most forms of eukaryotic translation initiation. eIF2 mediates the binding of tRNAiMet to the ribosome in a GTP-dependent manner. eIF2 is a heterotrimer consisting of an alpha, a beta, and a gamma subunit.
The eukaryotic initiation factor-4A (eIF4A) family consists of 3 closely related proteins EIF4A1, EIF4A2, and EIF4A3. These factors are required for the binding of mRNA to 40S ribosomal subunits. In addition these proteins are helicases that function to unwind double-stranded RNA.

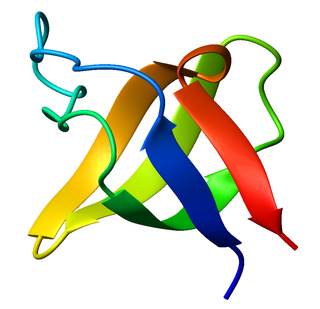
RNA recognition motif, RNP-1 is a putative RNA-binding domain of about 90 amino acids that are known to bind single-stranded RNAs. It was found in many eukaryotic proteins.
WH1 domain is an evolutionary conserved protein domain. Therefore, it has an important function.