The Friedel–Crafts reactions are a set of reactions developed by Charles Friedel and James Crafts in 1877 to attach substituents to an aromatic ring. Friedel–Crafts reactions are of two main types: alkylation reactions and acylation reactions. Both proceed by electrophilic aromatic substitution.
In organic chemistry, an acyl chloride (or acid chloride) is an organic compound with the functional group -COCl. Their formula is usually written RCOCl, where R is a side chain. They are reactive derivatives of carboxylic acids. A specific example of an acyl chloride is acetyl chloride, CH3COCl. Acyl chlorides are the most important subset of acyl halides.

An acyl halide is a chemical compound derived from an oxoacid by replacing a hydroxyl group with a halide group.

Caesium fluoride or cesium fluoride is an inorganic compound with the formula CsF and it is a hygroscopic white salt. Caesium fluoride can be used in organic synthesis as a source of the fluoride anion. Caesium also has the highest electropositivity of all non-radioactive elements and fluorine has the highest electronegativity of all elements.

Benzoyl chloride, also known as benzenecarbonyl chloride, is an organochlorine compound with the formula C7H5ClO. It is a colourless, fuming liquid with an irritating odour. It is mainly useful for the production of peroxides but is generally useful in other areas such as in the preparation of dyes, perfumes, pharmaceuticals, and resins.

Bromine trifluoride is an interhalogen compound with the formula BrF3. It is a straw-coloured liquid with a pungent odor. It is soluble in sulfuric acid but reacts violently with water and organic compounds. It is a powerful fluorinating agent and an ionizing inorganic solvent. It is used to produce uranium hexafluoride (UF6) in the processing and reprocessing of nuclear fuel.
Iodine pentafluoride is an interhalogen compound with chemical formula IF5. It is one of the fluorides of iodine. It is a colorless liquid, although impure samples appear yellow. It is used as a fluorination reagent and even a solvent in specialized syntheses.
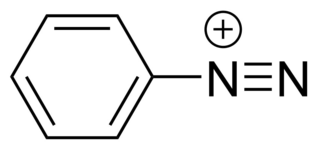
Diazonium compounds or diazonium salts are a group of organic compounds sharing a common functional group R−N+
2X−
where R can be any organic group, such as an alkyl or an aryl, and X is an inorganic or organic anion, such as a halogen.

A formylation reaction in organic chemistry refers to organic reactions in which an organic compound is functionalized with a formyl group (-CH=O). The reaction is a route to aldehydes (C-CH=O), formamides (N-CH=O), and formate esters (O-CH=O). A reagent that delivers the formyl group is called a formylating agent. A particularly important formylation process is hydroformylation which converts alkenes to the homologated aldehyde. The conversion of benzene to benzaldehyde is the basis of the Gattermann–Koch reaction:
Fluorosulfuric acid (IUPAC name: sulfurofluoridic acid) is the inorganic compound with the chemical formula HSO3F. It is one of the strongest acids commercially available. The formula HSO3F emphasizes its relationship to sulfuric acid, H2SO4; HSO3F is a tetrahedral molecule. It is a colourless liquid, although commercial samples are often yellow.
Antimony pentafluoride is the inorganic compound with the formula SbF5. This colourless, viscous liquid is a valuable Lewis acid and a component of the superacid fluoroantimonic acid, formed when mixing liquid HF with liquid SbF5 in a 2:1 ratio. It is notable for its Lewis acidity and its ability to react with almost all known compounds.

Hydrogen fluoride is a chemical compound with the chemical formula ‹See Tfd›HF. This colorless gas or liquid is the principal industrial source of fluorine, often as an aqueous solution called hydrofluoric acid. It is an important feedstock in the preparation of many important compounds including pharmaceuticals and polymers, e.g. polytetrafluoroethylene (PTFE). HF is widely used in the petrochemical industry as a component of superacids. Hydrogen fluoride boils at near room temperature, much higher than other hydrogen halides.

Fluoroantimonic acid is a mixture of hydrogen fluoride and antimony pentafluoride, containing various cations and anions. This substance is a superacid that can be over a billion times stronger than 100% pure sulfuric acid, depending on proportion of its ingredients. It has been shown to protonate even hydrocarbons to afford pentacoordinate carbocations. Extreme caution needs to be in place when handling fluoroantimonic acid. It is exceptionally corrosive, but can be stored in containers lined with PTFE (Teflon).
The Gattermann reaction, (also known as the Gattermann formylation and the Gattermann salicylaldehyde synthesis) is a chemical reaction in which aromatic compounds are formylated by a mixture of hydrogen cyanide (HCN) and hydrogen chloride (HCl) in the presence of a Lewis acid catalyst such as AlCl3. It is named for the German chemist Ludwig Gattermann and is similar to the Friedel–Crafts reaction.
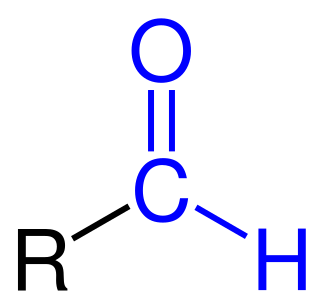
In biochemistry, the addition of a formyl functional group is termed formylation. A formyl functional group consists of a carbonyl bonded to hydrogen. When attached to an R group, a formyl group is called an aldehyde.

Leveling effect or solvent leveling refers to the effect of solvent on the properties of acids and bases. The strength of a strong acid is limited ("leveled") by the basicity of the solvent. Similarly the strength of a strong base is leveled by the acidity of the solvent. When a strong acid is dissolved in water, it reacts with it to form hydronium ion (H3O+). An example of this would be the following reaction, where "HA" is the strong acid:

N-Formylpiperidine is an organic compound with the formula C6H11NO. It is the amide of formic acid and piperidine. It can be used as a polar aprotic solvent, with better hydrocarbon solubility than other amide solvents such as dimethylformamide (DMF). It has also been used to transfer the formyl group to a Grignard reagent:
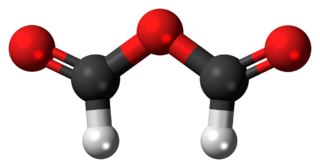
Formic anhydride, also called methanoic anhydride, is an organic compound with the chemical formula C
2H
2O
3 and a structural formula of (H(C=O)−)2O. It can be viewed as the anhydride of formic acid (HCOOH).

Acetic formic anhydride is an organic compound with the chemical formula C
3H
4O
3 and a structural formula of H
3C-(C=O)-O-(C=O)H. It can be viewed as the mixed anhydride of acetic acid and formic acid.
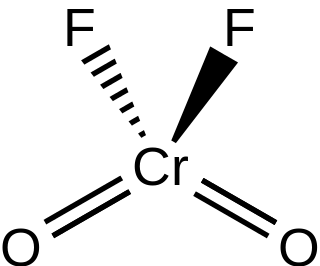
Chromyl fluoride is an inorganic compound with the formula CrO2F2. It is a violet-red colored crystalline solid that melts to an orange-red liquid.















