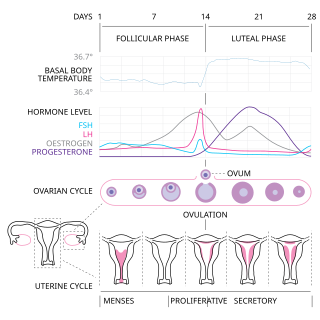
The menstrual cycle is a series of natural changes in hormone production and the structures of the uterus and ovaries of the female reproductive system that makes pregnancy possible. The ovarian cycle controls the production and release of eggs and the cyclic release of estrogen and progesterone. The uterine cycle governs the preparation and maintenance of the lining of the uterus (womb) to receive an embryo. These cycles are concurrent and coordinated, normally last between 21 and 35 days, with a median length of 28 days. Menarche usually occurs around the age of 12 years; menstrual cycles continue for about 30–45 years.

Ovulation is an important part of the menstrual cycle in female vertebrates where the egg cells are released from the ovaries as part of the ovarian cycle. In female humans ovulation typically occurs near the midpoint in the menstrual cycle and after the follicular phase. Ovulation is stimulated by an increase in luteinizing hormone (LH). The ovarian follicles rupture and release the secondary oocyte ovarian cells.
Luteinizing hormone is a hormone produced by gonadotropic cells in the anterior pituitary gland. The production of LH is regulated by gonadotropin-releasing hormone (GnRH) from the hypothalamus. In females, an acute rise of LH known as an LH surge, triggers ovulation and development of the corpus luteum. In males, where LH had also been called interstitial cell–stimulating hormone (ICSH), it stimulates Leydig cell production of testosterone. It acts synergistically with follicle-stimulating hormone (FSH).

Follicle-stimulating hormone (FSH) is a gonadotropin, a glycoprotein polypeptide hormone. FSH is synthesized and secreted by the gonadotropic cells of the anterior pituitary gland and regulates the development, growth, pubertal maturation, and reproductive processes of the body. FSH and luteinizing hormone (LH) work together in the reproductive system.
Gonadotropin-releasing hormone (GnRH) is a releasing hormone responsible for the release of follicle-stimulating hormone (FSH) and luteinizing hormone (LH) from the anterior pituitary. GnRH is a tropic peptide hormone synthesized and released from GnRH neurons within the hypothalamus. GnRH is inhibited by testosterone. The peptide belongs to gonadotropin-releasing hormone family. It constitutes the initial step in the hypothalamic–pituitary–gonadal axis.
Anovulation is when the ovaries do not release an oocyte during a menstrual cycle. Therefore, ovulation does not take place. However, a woman who does not ovulate at each menstrual cycle is not necessarily going through menopause. Chronic anovulation is a common cause of infertility.
Fertility medications, also known as fertility drugs, are medications which enhance reproductive fertility. For women, fertility medication is used to stimulate follicle development of the ovary. There are very few fertility medication options available for men.
In mammalian species, pseudopregnancy is a physical state whereby all the signs and symptoms of pregnancy are exhibited, with the exception of the presence of a fetus, creating a false pregnancy. The corpus luteum is responsible for the development of maternal behavior and lactation, which are mediated by the continued production of progesterone by the corpus luteum through some or all of pregnancy. In most species, the corpus luteum is degraded in the absence of a pregnancy. However, in some species, the corpus luteum may persist in the absence of pregnancy and cause "pseudopregnancy", in which the female will exhibit clinical signs of pregnancy.
The estrous cycle is a set of recurring physiological changes induced by reproductive hormones in females of mammalian subclass Theria. Estrous cycles start after sexual maturity in females and are interrupted by anestrous phases, otherwise known as "rest" phases, or by pregnancies. Typically, estrous cycles repeat until death. These cycles are widely variable in duration and frequency depending on the species. Some animals may display bloody vaginal discharge, often mistaken for menstruation. Many mammals used in commercial agriculture, such as cattle and sheep, may have their estrous cycles artificially controlled with hormonal medications for optimum productivity. The male equivalent, seen primarily in ruminants, is called rut.

In biology, folliculogenesis is the maturation of the ovarian follicle, a densely packed shell of somatic cells that contains an immature oocyte. Folliculogenesis describes the progression of a number of small primordial follicles into large preovulatory follicles that occurs in part during the menstrual cycle.

The hypothalamic–pituitary–gonadal axis refers to the hypothalamus, pituitary gland, and gonadal glands as if these individual endocrine glands were a single entity. Because these glands often act in concert, physiologists and endocrinologists find it convenient and descriptive to speak of them as a single system.

The menstrual cycle is on average 28 days in length. It begins with menses during the follicular phase, followed by ovulation and ending with the luteal phase. while historically, medical experts believed the luteal phase to be relatively fixed at approximately 14 days, recent research suggests that there can be wide variability in luteal phase lengths not just from person to person, but from cycle to cycle within one person. The luteal phase is characterized by changes to hormone levels, such as an increase in progesterone and estrogen levels, decrease in gonadotropins such as follicle-stimulating hormone (FSH) and luteinizing hormone (LH), changes to the endometrial lining to promote implantation of the fertilized egg, and development of the corpus luteum. In the absence of fertilization by sperm, the corpus luteum degenerates leading to a decrease in progesterone and estrogen, an increase in FSH and LH, and shedding of the endometrial lining (menses) to begin the menstrual cycle again.

The follicular phase, also known as the preovulatory phase or proliferative phase, is the phase of the estrous cycle during which follicles in the ovary mature from primary follicle to a fully mature graafian follicle. It ends with ovulation. The main hormones controlling this stage are secretion of gonadotropin-releasing hormones, which are follicle-stimulating hormones and luteinising hormones. They are released by pulsatile secretion. The duration of the follicular phase can differ depending on the length of the menstrual cycle, while the luteal phase is usually stable, does not really change and lasts 14 days.

A gonadotropin-releasing hormone agonist is a type of medication which affects gonadotropins and sex hormones. They are used for a variety of indications including in fertility medicine and to lower sex hormone levels in the treatment of hormone-sensitive cancers such as prostate cancer and breast cancer, certain gynecological disorders like heavy periods and endometriosis, high testosterone levels in women, early puberty in children, as a part of transgender hormone therapy, and to delay puberty in transgender youth among other uses. It is also used in the suppression of spontaneous ovulation as part of controlled ovarian hyperstimulation, an essential component in IVF. GnRH agonists are given by injections into fat, as implants placed into fat, and as nasal sprays.

Gonadotropin-releasing hormone antagonists are a class of medications that antagonize the gonadotropin-releasing hormone receptor and thus the action of gonadotropin-releasing hormone (GnRH). They are used in the treatment of prostate cancer, endometriosis, uterine fibroids, female infertility in assisted reproduction, and for other indications.
Controlled ovarian hyperstimulation is a technique used in assisted reproduction involving the use of fertility medications to induce ovulation by multiple ovarian follicles. These multiple follicles can be taken out by oocyte retrieval for use in in vitro fertilisation (IVF), or be given time to ovulate, resulting in superovulation which is the ovulation of a larger-than-normal number of eggs, generally in the sense of at least two. When ovulated follicles are fertilised in vivo, whether by natural or artificial insemination, there is a very high risk of a multiple pregnancy.
Infertility in polycystic ovary disease (PCOS) is a hormonal imbalance in women that is thought to be one of the leading causes of female infertility. Polycystic ovary syndrome causes more than 75% of cases of anovulatory infertility.
Progesterone devices are broadly used in the control of reproductive management in livestock. They work by increasing circulating plasma progesterone levels with the following consequences:
- Progesterone suppresses the secretion of gonadotropin-releasing hormone (GnRH) from the hypothalamus. This is done via a negative feedback to the hypothalamus neuroendocrine cells, by inhibition of KiSSpeptin KiSS1-derived peptide receptor, a protein needed for the release of GnRH.
- Low levels of GnRH prevent the emergence of a dominant follicle by reducing the release of LH and FSH hormones. Current follicular waves cease and a new wave emerges 3–5 days after implant.
- A dominant follicle develops but there is no ovulation as LH release is prevented by suppression of GnRH.
- Removal of progesterone device produces a surge of GnRH, generating a pulse of LH that induces ovulation.
Induction of final maturation of oocytes is a procedure that is usually performed as part of controlled ovarian hyperstimulation to render the oocytes fully developed and thereby resulting in optimal pregnancy chances. It is basically a replacement for the luteinizing hormone (LH) surge whose effects include final maturation in natural menstrual cycles.
Gonadotropin surge-attenuating factor (GnSAF) is a nonsteroidal ovarian hormone produced by the granulosa cells of small antral ovarian follicles in females. GnSAF is involved in regulating the secretion of luteinizing hormone (LH) from the anterior pituitary and the ovarian cycle. During the early to mid-follicular phase of the ovarian cycle, GnSAF acts on the anterior pituitary to attenuate LH release, limiting the secretion of LH to only basal levels. At the transition between follicular and luteal phase, GnSAF bioactivity declines sufficiently to permit LH secretion above basal levels, resulting in the mid-cycle LH surge that initiates ovulation. In normally ovulating women, the LH surge only occurs when the oocyte is mature and ready for extrusion. GnSAF bioactivity is responsible for the synchronised, biphasic nature of LH secretion.









