
Sintering or frittage is the process of compacting and forming a solid mass of material by pressure or heat without melting it to the point of liquefaction. Sintering happens as part of a manufacturing process used with metals, ceramics, plastics, and other materials. The nanoparticles in the sintered material diffuse across the boundaries of the particles, fusing the particles together and creating a solid piece.
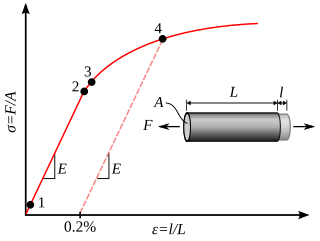
In physics and materials science, plasticity is the ability of a solid material to undergo permanent deformation, a non-reversible change of shape in response to applied forces. For example, a solid piece of metal being bent or pounded into a new shape displays plasticity as permanent changes occur within the material itself. In engineering, the transition from elastic behavior to plastic behavior is known as yielding.
In materials science, superplasticity is a state in which solid crystalline material is deformed well beyond its usual breaking point, usually over about 400% during tensile deformation. Such a state is usually achieved at high homologous temperature. Examples of superplastic materials are some fine-grained metals and ceramics. Other non-crystalline materials (amorphous) such as silica glass and polymers also deform similarly, but are not called superplastic, because they are not crystalline; rather, their deformation is often described as Newtonian fluid. Superplastically deformed material gets thinner in a very uniform manner, rather than forming a "neck" that leads to fracture. Also, the formation of microvoids, which is another cause of early fracture, is inhibited. Superplasticity must not be confused with superelasticity.

A crystallite is a small or even microscopic crystal which forms, for example, during the cooling of many materials. Crystallites are also referred to as grains.

In materials science, creep is the tendency of a solid material to undergo slow deformation while subject to persistent mechanical stresses. It can occur as a result of long-term exposure to high levels of stress that are still below the yield strength of the material. Creep is more severe in materials that are subjected to heat for long periods and generally increases as they near their melting point.

In materials science, a dislocation or Taylor's dislocation is a linear crystallographic defect or irregularity within a crystal structure that contains an abrupt change in the arrangement of atoms. The movement of dislocations allow atoms to slide over each other at low stress levels and is known as glide or slip. The crystalline order is restored on either side of a glide dislocation but the atoms on one side have moved by one position. The crystalline order is not fully restored with a partial dislocation. A dislocation defines the boundary between slipped and unslipped regions of material and as a result, must either form a complete loop, intersect other dislocations or defects, or extend to the edges of the crystal. A dislocation can be characterised by the distance and direction of movement it causes to atoms which is defined by the Burgers vector. Plastic deformation of a material occurs by the creation and movement of many dislocations. The number and arrangement of dislocations influences many of the properties of materials.
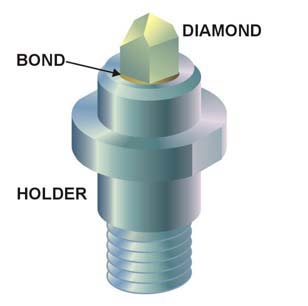
A superhard material is a material with a hardness value exceeding 40 gigapascals (GPa) when measured by the Vickers hardness test. They are virtually incompressible solids with high electron density and high bond covalency. As a result of their unique properties, these materials are of great interest in many industrial areas including, but not limited to, abrasives, polishing and cutting tools, disc brakes, and wear-resistant and protective coatings.

Work hardening, also known as strain hardening, is the strengthening of a metal or polymer by plastic deformation. Work hardening may be desirable, undesirable, or inconsequential, depending on the context.
Precipitation hardening, also called age hardening or particle hardening, is a heat treatment technique used to increase the yield strength of malleable materials, including most structural alloys of aluminium, magnesium, nickel, titanium, and some steels, stainless steels, and duplex stainless steel. In superalloys, it is known to cause yield strength anomaly providing excellent high-temperature strength.

In materials science, a grain boundary is the interface between two grains, or crystallites, in a polycrystalline material. Grain boundaries are two-dimensional defects in the crystal structure, and tend to decrease the electrical and thermal conductivity of the material. Most grain boundaries are preferred sites for the onset of corrosion and for the precipitation of new phases from the solid. They are also important to many of the mechanisms of creep. On the other hand, grain boundaries disrupt the motion of dislocations through a material, so reducing crystallite size is a common way to improve mechanical strength, as described by the Hall–Petch relationship.
In metallurgy, selective leaching, also called dealloying, demetalification, parting and selective corrosion, is a corrosion type in some solid solution alloys, when in suitable conditions a component of the alloys is preferentially leached from the initially homogenous material. The less noble metal is removed from the alloy by a microscopic-scale galvanic corrosion mechanism. The most susceptible alloys are the ones containing metals with high distance between each other in the galvanic series, e.g. copper and zinc in brass. The elements most typically undergoing selective removal are zinc, aluminium, iron, cobalt, chromium, and others.

In materials science, recrystallization is a process by which deformed grains are replaced by a new set of defect-free grains that nucleate and grow until the original grains have been entirely consumed. Recrystallization is usually accompanied by a reduction in the strength and hardness of a material and a simultaneous increase in the ductility. Thus, the process may be introduced as a deliberate step in metals processing or may be an undesirable byproduct of another processing step. The most important industrial uses are softening of metals previously hardened or rendered brittle by cold work, and control of the grain structure in the final product. Recrystallization temperature is typically 0.3–0.4 times the melting point for pure metals and 0.5 times for alloys.
Nickel aluminide refers to either of two widely used intermetallic compounds, Ni3Al or NiAl, but the term is sometimes used to refer to any nickel–aluminium alloy. These alloys are widely used because of their high strength even at high temperature, low density, corrosion resistance, and ease of production. Ni3Al is of specific interest as a precipitate in nickel-based superalloys, where it is called the γ' (gamma prime) phase. It gives these alloys high strength and creep resistance up to 0.7–0.8 of its melting temperature. Meanwhile, NiAl displays excellent properties such as lower density and higher melting temperature than those of Ni3Al, and good thermal conductivity and oxidation resistance. These properties make it attractive for special high-temperature applications like coatings on blades in gas turbines and jet engines. However, both these alloys have the disadvantage of being quite brittle at room temperature, with Ni3Al remaining brittle at high temperatures as well. To address this problem, has been shown that Ni3Al can be made ductile when manufactured in single-crystal form rather than in polycrystalline form.
Damage mechanics is concerned with the representation, or modeling, of damage of materials that is suitable for making engineering predictions about the initiation, propagation, and fracture of materials without resorting to a microscopic description that would be too complex for practical engineering analysis.
Methods have been devised to modify the yield strength, ductility, and toughness of both crystalline and amorphous materials. These strengthening mechanisms give engineers the ability to tailor the mechanical properties of materials to suit a variety of different applications. For example, the favorable properties of steel result from interstitial incorporation of carbon into the iron lattice. Brass, a binary alloy of copper and zinc, has superior mechanical properties compared to its constituent metals due to solution strengthening. Work hardening has also been used for centuries by blacksmiths to introduce dislocations into materials, increasing their yield strengths.

In materials science, grain-boundary strengthening is a method of strengthening materials by changing their average crystallite (grain) size. It is based on the observation that grain boundaries are insurmountable borders for dislocations and that the number of dislocations within a grain has an effect on how stress builds up in the adjacent grain, which will eventually activate dislocation sources and thus enabling deformation in the neighbouring grain as well. By changing grain size, one can influence the number of dislocations piled up at the grain boundary and yield strength. For example, heat treatment after plastic deformation and changing the rate of solidification are ways to alter grain size.
Severe plastic deformation (SPD) is a generic term describing a group of metalworking techniques involving very large strains typically involving a complex stress state or high shear, resulting in a high defect density and equiaxed "ultrafine" grain (UFG) size or nanocrystalline (NC) structure.

Grain boundary sliding (GBS) is a material deformation mechanism where grains slide against each other. This occurs in polycrystalline material under external stress at high homologous temperature and low strain rate and is intertwined with creep. Homologous temperature describes the operating temperature relative to the melting temperature of the material. There are mainly two types of grain boundary sliding: Rachinger sliding, and Lifshitz sliding. Grain boundary sliding usually occurs as a combination of both types of sliding. Boundary shape often determines the rate and extent of grain boundary sliding.

A nanostructured film is a film resulting from engineering of nanoscale features, such as dislocations, grain boundaries, defects, or twinning. In contrast to other nanostructures, such as nanoparticles, the film itself may be up to several microns thick, but possesses a large concentration of nanoscale features homogeneously distributed throughout the film. Like other nanomaterials, nanostructured films have sparked much interest as they possess unique properties not found in bulk, non-nanostructured material of the same composition. In particular, nanostructured films have been the subject of recent research due to their superior mechanical properties, including strength, hardness, and corrosion resistance compared to regular films of the same material. Examples of nanostructured films include those produced by grain boundary engineering, such as nano-twinned ultra-fine grain copper, or dual phase nanostructuring, such as crystalline metal and amorphous metallic glass nanocomposites.
Microstructurally stable nanocrystalline alloys are alloys that are designed to resist microstructural coarsening under various thermo-mechanical loading conditions.




























