Okadaic acid, C44H68O13, is a toxin produced by several species of dinoflagellates, and is known to accumulate in both marine sponges and shellfish. One of the primary causes of diarrhetic shellfish poisoning, okadaic acid is a potent inhibitor of specific protein phosphatases and is known to have a variety of negative effects on cells. A polyketide, polyether derivative of a C38 fatty acid, okadaic acid and other members of its family have shined light upon many biological processes both with respect to dinoflagellete polyketide synthesis as well as the role of protein phosphatases in cell growth.

Pederin is a vesicant toxic amide with two tetrahydropyran rings, found in the haemolymph of the beetle genus Paederus, including the Nairobi fly, belonging to the family Staphylinidae. It was first characterized by processing 25 million field-collected P. fuscipes. It makes up approximately 0.025% of an insects weight.

Cryptophycins are a family of macrolide molecules that are potent cytotoxins and have been studied for potential antiproliferative properties useful in developing chemotherapy. They are members of the depsipeptide family.

Indolocarbazoles (ICZs) are a class of compounds that are under current study due to their potential as anti-cancer as well as antimicrobial drugs and the prospective number of derivatives and uses found from the basic backbone alone. First isolated in 1977, a wide range of structures and derivatives have been found or developed throughout the world. Due to the extensive number of structures available, this review will focus on the more important groups here while covering their occurrence, biological activity, biosynthesis, and laboratory synthesis.
Dogger Bank itch is a cutaneous condition characterized by a long-lasting dermatitis caused by exposure to the sea chervil, Alcyonidium diaphanum, a bryozoan. The disease, common in fishermen who work in the North Sea, has been recognized by the Danish Workman's Compensation Act since 1939.

Capnellene is a naturally occurring tricyclic hydrocarbon derived from Capnella imbricata, a species of soft coral found in Indonesia. Since the 1970s, capnellene has been targeted for synthesis by numerous investigators due to its stereochemistry, functionality, and the interesting geometry of the carbon skeleton. Many alcohol derivatives of capnellene have demonstrated potential as a chemotherapeutic agent with antibacterial, anti-inflammatory and anti-tumor properties.

Callystatin A is a polyketide natural product from the leptomycin family of secondary metabolites. It was first isolated in 1997 from the marine sponge Callyspongia truncata which was collected from the Goto Islands in the Nagasaki Prefecture of Japan by the Kobayashi group. Since then its absolute configuration has been elucidated and callystatin A was discovered to have anti-fungal and anti-tumor activities with extreme potency against the human epidermoid carcinoma KB cells (IG50 = 10 pg/ml) and the mouse lymphocytic leukemia Ll210 cells (IG50 = 20 pg/ml).
Sea sponge aquaculture is the process of farming sea sponges under controlled conditions. It has been conducted in the world's oceans for centuries using a number of aquaculture techniques. There are many factors such as light, salinity, pH, dissolved oxygen and the accumulation of waste products that influence the growth rate of sponges. The benefits of sea sponge aquaculture are realised as a result of its ease of establishment, minimum infrastructure requirements and the potential to be used as a source of income for populations living in developing countries. Sea sponges are produced on a commercial scale to be used as bath sponges or to extract biologically active compounds which are found in certain sponge species. Techniques such as the rope and mesh bag method are used to culture sponges independently or within an integrated multi-trophic aquaculture system setting. One of the only true sustainable sea sponges cultivated in the world occur in the region of Micronesia, with a number of growing and production methods used to ensure and maintain the continued sustainability of these farmed species.

Papuamides A and B are depsipeptides which appear to protect T cells from HIV. They were isolated from the sponge Theonella, and are part of a larger group of structurally similar depsipeptides—also isolated from sponges—including neamphamide A, callipeltin A, and mirabamides A-D.

Halicylindramides are a group of antifungal peptides. The first compounds of this type, designated halicylindramides A through E, were isolated from sea sponges of the genus Halichondria. More compounds in the family, designated F, G and H, were found in sponges of the genus Petrosia. Halicylindramide A has been synthesized by chemists.

Aciculitins are antifungal cyclic peptides isolated from a marine sponge. There are 3 Aciculitins that are isolated from the Lithistid sponge Aciculites orientalis that differ by their homologous lipid residues.

Eudistomins are β-carboline derivatives, isolated from ascidians, like Ritterella sigillinoides, Lissoclinum fragile, or Pseudodistoma aureum.

Stevensine is a bromopyrrole alkaloid originally isolated from an unidentified Micronesian marine sponge, as well as the known sponge species, Pseudaxinyssa cantharella and Axinella corrugata. Total synthesis of stevensine has been achieved by Ying-zi Xu et al., and investigations into the biosynthetic origin has been explored by Paul Andrade et al. Understanding methods to synthesize stevensine and other similar compounds is an important step to accomplish, as marine sponges contain numerous biologically active metabolites that have been shown to function as anything from antitumor to antibacterial agents when tested for medicinal applications. Reasons for why marine sponges contain so many bio-active chemicals has been attributed to their sessile nature, and the need to produce chemical defenses to ensure survival. However, since many of these compounds naturally occur in small amounts, harvesting the sponges has in the past led to near-extinction of some species.

Lacking an immune system, protective shell, or mobility, sponges have developed an ability to synthesize a variety of unusual compounds for survival. C-nucleosides isolated from Caribbean Cryptotethya crypta, were the basis for the synthesis of zidovudine (AZT), aciclovir (Cyclovir), cytarabine (Depocyt), and cytarabine derivative gemcitabine (Gemzar).
Dysidea arenaria is a species of marine sponge (poriferan) found in the Pacific Ocean. It is a member of the order Dictyoceratida, one of two sponge orders that make up the keratose or "horny" sponges in which a mineral skeleton is absent and a skeleton of organic fibers is present instead.
Mycale adhaerens, the purple scallop sponge, is a species of marine demosponge in the family Mycalidae. Mycale is a large genus and this species is placed in the subgenus Aegogropila making its full name, Mycale (Aegogropila) adhaerens. It grows symbiotically on the valves of scallop shells and is native to the west coast of North America.

Swinholides are dimeric 42 carbon-ring polyketides that exhibit a 2-fold axis of symmetry. Found mostly in the marine sponge Theonella, swinholides encompass cytotoxic and antifungal activities via disruption of the actin skeleton. Swinholides were first described in 1985 and the structure and stereochemistry were updated in 1989 and 1990, respectively. Thirteen swinholides have been described in the literature, including close structural compounds such as misakinolides/bistheonellides, ankaraholides, and hurgholide A It is suspected that symbiotic microbes that inhabit the sponges rather than the sponges themselves produce swinholides since the highest concentration of swinholides are found in the unicellular bacterial fraction of sponges and not in the sponge fraction or cyanobacteria fraction that also inhabit the sponges.
Mycale hentscheli is a marine sponge, which is known to be a rich source of bioactive small molecules. These natural products originate from the sponge's microbiota. Examples of these molecules include the potent cytotoxic polyketides: pateamine, peloruside, and mycalamide.
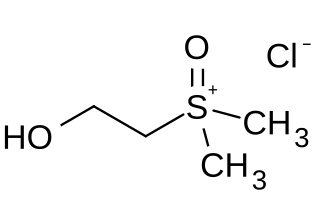
(2-Hydroxyethyl) dimethylsulfoxonium chloride is an organic salt found in sea chervils and sea sponges that causes the Dogger Bank itch.

Pseudoceratina is a genus of sponge within the family Pseudoceratinidae. They are characterized by possession of a dendritic fiber skeleton lacking laminar bark but containing pith. They have been found in a variety of habitats including the Great Barrier reef, the Red Sea, and Jamaica. Sponges of this genus have a microbiome known to produce a variety of chemicals that are used in pharmaceutical and anti-fouling activities. Notably, a species in this genus produces a chemical that is effective in inhibiting the migration of metastatic breast cancer cells.















