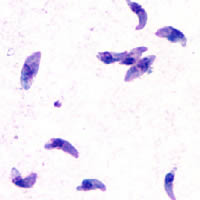
Toxoplasmosis is a parasitic disease caused by Toxoplasma gondii, an apicomplexan. Infections with toxoplasmosis are associated with a variety of neuropsychiatric and behavioral conditions. Occasionally, people may have a few weeks or months of mild, flu-like illness such as muscle aches and tender lymph nodes. In a small number of people, eye problems may develop. In those with a weak immune system, severe symptoms such as seizures and poor coordination may occur. If a woman becomes infected during pregnancy, a condition known as congenital toxoplasmosis may affect the child.

Isosporiasis, also known as cystoisosporiasis, is a human intestinal disease caused by the parasite Cystoisospora belli. It is found worldwide, especially in tropical and subtropical areas. Infection often occurs in immuno-compromised individuals, notably AIDS patients, and outbreaks have been reported in institutionalized groups in the United States. The first documented case was in 1915. It is usually spread indirectly, normally through contaminated food or water (CDC.gov).

Coccidia (Coccidiasina) are a subclass of microscopic, spore-forming, single-celled obligate intracellular parasites belonging to the apicomplexan class Conoidasida. As obligate intracellular parasites, they must live and reproduce within an animal cell. Coccidian parasites infect the intestinal tracts of animals, and are the largest group of apicomplexan protozoa.

A gametocyte is a eukaryotic germ cell that divides by mitosis into other gametocytes or by meiosis into gametids during gametogenesis. Male gametocytes are called spermatocytes, and female gametocytes are called oocytes.

Plasmodium malariae is a parasitic protozoan that causes malaria in humans. It is one of several species of Plasmodium parasites that infect other organisms as pathogens, also including Plasmodium falciparum and Plasmodium vivax, responsible for most malarial infection. Found worldwide, it causes a so-called "benign malaria", not nearly as dangerous as that produced by P. falciparum or P. vivax. The signs include fevers that recur at approximately three-day intervals – a quartan fever or quartan malaria – longer than the two-day (tertian) intervals of the other malarial parasite.

Eimeria is a genus of apicomplexan parasites that includes various species capable of causing the disease coccidiosis in animals such as cattle, poultry and smaller ruminants including sheep and goats. Eimeria species are considered to be monoxenous because the life cycle is completed within a single host, and stenoxenous because they tend to be host specific, although a number of exceptions have been identified. Species of this genus infect a wide variety of hosts. Thirty-one species are known to occur in bats (Chiroptera), two in turtles, and 130 named species infect fish. Two species infect seals. Five species infect llamas and alpacas: E. alpacae, E. ivitaensis, E. lamae, E. macusaniensis, and E. punonensis. A number of species infect rodents, including E. couesii, E. kinsellai, E. palustris, E. ojastii and E. oryzomysi. Others infect poultry, rabbits and cattle. For full species list, see below.
Cytauxzoon felis is a protozoal organism transmitted to domestic cats by tick bites, and whose natural reservoir host is the bobcat. C. felis has been found in other wild felid species such as the cougar, as well as a white tiger in captivity. C. felis infection is limited to the family Felidae which means that C. felis poses no zoonotic risk or agricultural risk. Until recently it was believed that after infection with C. felis, pet cats almost always died. As awareness of C. felis has increased it has been found that treatment is not always futile. More cats have been shown to survive the infection than was previously thought. New treatments offer as much as 60% survival rate.

Sarcocystis is a genus of protozoan parasites, with many species infecting mammals, reptiles and birds. Its name is dervived from Greek sarx = flesh and kystis = bladder.

Equine protozoal myeloencephalitis (EPM) is a disease that affects the central nervous system of horses. It is caused by a protozoal infection that is brought about by the apicomplexan parasites Sarcocystis neurona or Neospora hughesi. Most cases are caused by S. neurona. The lifecycle and transmission of N. hughesi is not well understood. The parasites create lesions in both the brain and spinal cord of the affected horses leading to neurological issues. Most horses infected with S. neurona do not exhibit neurological symptoms consistent with EPM.
Megaloschizonts are large schizonts that produce extremely high numbers of merozoites. They are found in various species of the Phylum Apicomplexa. The Apicomplexa phylum contains several parasitic protozoans. They have a very complex life cycle that includes several stages. Megaloschizonts and the smaller schizonts are the part of the life cycle that takes place inside the infected host organism and operates as an asexually reproductive cell. Megaloschizonts appear as grey-white nodules found in the smooth muscle of major organs, such as the heart, liver, lung or spleen.

Ponazuril (INN), sold by Merial, Inc., now part of Boehringer Ingelheim, under the trade name Marquis® , is a drug currently approved for the treatment of equine protozoal myeloencephalitis (EPM) in horses, caused by coccidia Sarcocystis neurona. More recently, veterinarians have been preparing a formulary version of the medication for use in small animals such as cats, dogs, and rabbits against coccidia as an intestinal parasite. Coccidia treatment in small animals is far shorter than treatment for EPM.
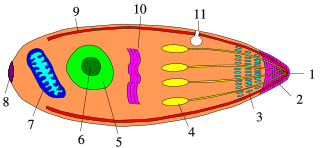
Apicomplexans, a group of intracellular parasites, have life cycle stages that allow them to survive the wide variety of environments they are exposed to during their complex life cycle. Each stage in the life cycle of an apicomplexan organism is typified by a cellular variety with a distinct morphology and biochemistry.
Virus quantification is counting or calculating the number of virus particles (virions) in a sample to determine the virus concentration. It is used in both research and development (R&D) in academic and commercial laboratories as well as in production situations where the quantity of virus at various steps is an important variable that must be monitored. For example, the production of virus-based vaccines, recombinant proteins using viral vectors, and viral antigens all require virus quantification to continually monitor and/or modify the process in order to optimize product quality and production yields and to respond to ever changing demands and applications. Other examples of specific instances where viruses need to be quantified include clone screening, multiplicity of infection (MOI) optimization, and adaptation of methods to cell culture.

Theileria parva is a species of parasites, named in honour of Arnold Theiler, that causes East Coast fever (theileriosis) in cattle, a costly disease in Africa. The main vector for T. parva is the tick Rhipicephalus appendiculatus. Theiler found that East Coast fever was not the same as redwater, but caused by a different protozoan.
The plaque reduction neutralization test is used to quantify the titer of neutralizing antibody for a virus.
Leucocytozoon caulleryi is a species of the genus Leucocytozoon, a genus of parasitic alveolates.
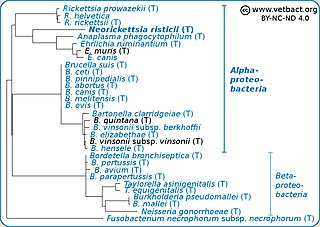
Neorickettsia risticii, formerly Ehrlichia risticii, is an obligate intracellular gram negative bacteria that typically lives as an endosymbiont to parasitic flatworms, specifically flukes. N. risticii is the known causative agent of equine neorickettsiosis, which gets its name from its discovery near the Potomac River in Maryland and Virginia. N. risticii was first recovered from horses in this region in 1984 but was not recognized as the causative agent of PHF until 1979. Potomac horse fever is currently endemic in the United States but has also been reported with lower frequency in other regions, including Canada, Brazil, Uruguay, and Europe. PHF is a condition that is clinically important for horses since it can cause serious signs such as fever, diarrhea, colic, and laminitis. PHF has a fatality rate of approximately 30%, making this condition one of the concerns for horse owners in endemic regions N. risticii is typically acquired in the middle to late summer near freshwater streams or rivers, as well as on irrigated pastures. This is a seasonal infection because it relies on the ingestion of an arthropod vector, which are more commonly found on pasture in the summer months. Although N. risticii is a well known causative agent for PHF in horses, it may act as a potential pathogen in cats and dogs as well. Not only has N. risticii been successfully cultured from monocytes of dogs and cats, but cats have become clinically ill after experimental infection with the bacteria. In addition, N. risticii has been isolated and cultured from human histiocytic lymphoma cells.

Cystoisospora belli, previously known as Isospora belli, is a parasite that causes an intestinal disease known as cystoisosporiasis. This protozoan parasite is opportunistic in immune suppressed human hosts. It primarily exists in the epithelial cells of the small intestine, and develops in the cell cytoplasm. The distribution of this coccidian parasite is cosmopolitan, but is mainly found in tropical and subtropical areas of the world such as the Caribbean, Central and S. America, India, Africa, and S.E. Asia. In the U.S., it is usually associated with HIV infection and institutional living.
Sarcocystis calchasi is an apicomplexan parasite. It has been identified to be the cause of Pigeon protozoal encephalitis (PPE) in the intermediate hosts, domestic pigeons. PPE is a central-nervous disease of domestic pigeons. Initially there have been reports of this parasite in Germany, with an outbreak in 2008 and in 2011 in the United States. Sarcocystis calchasi is transmitted by the definitive host Accipter hawks.

Quartan fever is one of the four types of malaria which can be contracted by humans.















