
Mitragyna speciosa is a tropical evergreen tree of the Rubiaceae family native to Southeast Asia. It is indigenous to Cambodia, Thailand, Indonesia, Malaysia, Myanmar, and Papua New Guinea, where its leaves, known as "kratom" have been used in herbal medicine since at least the 19th century. They have also historically been consumed via chewing, smoking, and as a tea. Kratom has opioid-like properties and some stimulant-like effects. As of 2018, the efficacy and safety of kratom are unclear. In 2019, the United States Food and Drug Administration (FDA) stated that there is no evidence that kratom is safe or effective for treating any condition. Some people take it for managing chronic pain, for treating opioid withdrawal symptoms, or for recreational purposes. The onset of effects typically begins within five to ten minutes and lasts for two to five hours.

Voacangine is an alkaloid found predominantly in the root bark of the Voacanga africana tree, as well as in other plants such as Tabernanthe iboga, Tabernaemontana africana, Trachelospermum jasminoides, Tabernaemontana divaricata and Ervatamia yunnanensis. It is an iboga alkaloid which commonly serves as a precursor for the semi-synthesis of ibogaine. It has been demonstrated in animals to have similar anti-addictive properties to ibogaine itself. It also potentiates the effects of barbiturates. Under UV-A and UV-B light its crystals fluoresce blue-green, and it is soluble in ethanol.

Indole alkaloids are a class of alkaloids containing a structural moiety of indole; many indole alkaloids also include isoprene groups and are thus called terpene indole or secologanin tryptamine alkaloids. Containing more than 4100 known different compounds, it is one of the largest classes of alkaloids. Many of them possess significant physiological activity and some of them are used in medicine. The amino acid tryptophan is the biochemical precursor of indole alkaloids.

Mitraphylline, an oxindole derivative, is an active alkaloid in the leaves of the tree Mitragyna speciosa, commonly known as kratom. As a non-narcotic constituent, it also occurs to a significant amount in the bark of Uncaria tomentosa along with a number of isomeric alkaloids.

The δ-opioid receptor, also known as delta opioid receptor or simply delta receptor, abbreviated DOR or DOP, is an inhibitory 7-transmembrane G-protein coupled receptor coupled to the G protein Gi/G0 and has enkephalins as its endogenous ligands. The regions of the brain where the δ-opioid receptor is largely expressed vary from species model to species model. In humans, the δ-opioid receptor is most heavily expressed in the basal ganglia and neocortical regions of the brain.

Desmetramadol, also known as O-desmethyltramadol (O-DSMT), is an opioid analgesic and the main active metabolite of tramadol. Tramadol is demethylated by the liver enzyme CYP2D6 to desmetramadol in the same way as codeine, and so similarly to the variation in effects seen with codeine, individuals who have a less active form of CYP2D6 will tend to have reduced analgesic effects from tramadol. Because desmetramadol itself does not need to be metabolized to induce an analgesic effect, it can be used in individuals with low CYP2D6 activity unlike tramadol.

7-Hydroxymitragynine (7-OH) is a terpenoid indole alkaloid from the plant Mitragyna speciosa, commonly known as kratom. It was first described in 1994 and is a natural product derived from mitragynine present in the kratom leaf. 7-OH binds to opioid receptors like mitragynine, but research suggests that 7-OH binds with greater efficacy.

Desformylflustrabromine (dFBr) is a NMT derivative indole alkaloid which was first isolated from the marine bryozoan Flustra foliacea.

Akuammine (vincamajoridine) is an indole alkaloid. It is the most abundant alkaloid found in the seeds from the tree Picralima nitida, commonly known as akuamma, comprising 0.56% of the dried powder. It has also been isolated from Vinca major. Akuammine is structurally related to yohimbine, mitragynine and more distantly Voacangine, all of which are alkaloid plant products with pharmacological properties.

Rhynchophylline is an alkaloid found in certain Uncaria species (Rubiaceae), notably Uncaria rhynchophylla and Uncaria tomentosa. It also occurs in the leaves of Mitragyna speciosa (kratom) and Mitragyna tubulosa, a tree native to Thailand. Chemically, it is related to the alkaloid mitragynine.

Pericine is one of a number of indole alkaloids found in the tree Picralima nitida, commonly known as akuamma. As with some other alkaloids from this plant such as akuammine, pericine has been shown to bind to mu opioid receptors in vitro, and has an IC50 of 0.6 μmol, within the range of a weak analgesic. It may also have convulsant effects.

Ajmalicine, also known as δ-yohimbine or raubasine, is an antihypertensive drug used in the treatment of high blood pressure. It has been marketed under numerous brand names including Card-Lamuran, Circolene, Cristanyl, Duxil, Duxor, Hydroxysarpon, Iskedyl, Isosarpan, Isquebral, Lamuran, Melanex, Raunatin, Saltucin Co, Salvalion, and Sarpan. It is an alkaloid found naturally in various plants such as Rauvolfia spp., Catharanthus roseus, and Mitragyna speciosa.

Conolidine is an indole alkaloid. Preliminary reports suggest that it could provide analgesic effects with few of the detrimental side-effects associated with opioids such as morphine, though at present it has only been evaluated in mouse models.
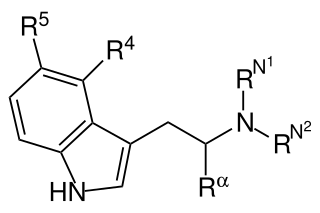
Substituted tryptamines, or serotonin analogues, are organic compounds which may be thought of as being derived from tryptamine itself. The molecular structures of all tryptamines contain an indole ring, joined to an amino (NH2) group via an ethyl (−CH2–CH2−) sidechain. In substituted tryptamines, the indole ring, sidechain, and/or amino group are modified by substituting another group for one of the hydrogen (H) atoms.
Akuammicine is a monoterpene indole alkaloid of the Vinca sub-group. It is found in the Apocynaceae family of plants including Picralima nitida, Vinca minor and the Aspidosperma.

Mitragynine pseudoindoxyl is a rearrangement product of 7-hydroxymitragynine, an active metabolite of mitragynine.
Iboga-type alkaloids are a set of monoterpene indole alkaloids comprising naturally occurring compounds found in Tabernanthe and Tabernaemontana, as well as synthetic structural analogs. Naturally occurring iboga-type alkaloids include ibogamine, ibogaine, tabernanthine, and other substituted ibogamines. Many iboga-type alkaloids display biological activities such as cardiac toxicity and psychoactive effects, and some have been studied as potential treatments for drug addiction.

Apparicine is a monoterpenoid tricyclic indole alkaloid. It is named after Apparicio Duarte, a Brazilian botanist who studied the Aspidosperma species from which apparicine was first isolated. It was the first member of the vallesamine group of indole alkaloids to be isolated and have its structure established, which was first published in 1965. It has also been known by the synonyms gomezine, pericalline, and tabernoschizine.

Mitragynine is an indole-based alkaloid and the most abundant active alkaloid in the Southeast Asian plant Mitragyna speciosa, commonly known as kratom. The total alkaloid concentration in dried leaves ranges from 0.5 to 1.5%. In Thai varieties, mitragynine is the most abundant component, while 7-hydroxymitragynine is a minor constituent. In Malaysian kratom varieties, mitragynine is present at lower concentration. Such preparations are orally consumed and typically involve dried kratom leaves which are brewed into tea or ground and placed into capsules. Mitragynine consumption for medicinal and recreational purposes dates back centuries, although early use was primarily limited to Southeast Asian countries such as Indonesia and Thailand, where the plant grows indigenously. Recently, mitragynine use has spread throughout Europe and the Americas as both a recreational and medicinal drug. While research into the effects of kratom have begun to emerge, investigations on the active compound mitragynine are less common.

Ignavine is a naturally occurring diterpene alkaloid found in Aconiti tuber. It has been reported to act as a μ-opioid receptor (MOR) positive allosteric modulator (PAM). The drug potentiated responses to the selective MOR agonist DAMGO at low concentrations but inhibited DAMGO at high concentrations. Ignavine alone has been found to produce analgesic effects in animals, but with a biphasic dose–response curve. Although described as a MOR PAM, other research suggests that ignavine is a ligand of the orthosteric site of the MOR and does not act as a PAM. Instead, it may be a MOR partial agonist. However, more research is necessary to clarify its MOR actions. Ignavine was first isolated by 1952 and its reported MOR PAM activity was first reported by 2016.




















