
Bromine is a chemical element with the symbol Br and atomic number 35. It is the third-lightest halogen, and is a fuming red-brown liquid at room temperature that evaporates readily to form a similarly coloured gas. Its properties are intermediate between those of chlorine and iodine. Isolated independently by two chemists, Carl Jacob Löwig and Antoine Jérôme Balard, its name was derived from the Ancient Greek βρῶμος ("stench"), referring to its sharp and disagreeable smell.

Heptane or n-heptane is the straight-chain alkane with the chemical formula H3C(CH2)5CH3 or C7H16, and is one of the main components of gasoline (petrol). When used as a test fuel component in anti-knock test engines, a 100% heptane fuel is the zero point of the octane rating scale (the 100 point is 100% iso-octane). Octane number equates to the anti-knock qualities of a comparison mixture of heptane and isooctane which is expressed as the percentage of isooctane in heptane and is listed on pumps for gasoline (petrol) dispensed globally.
A halide is a binary phase, of which one part is a halogen atom and the other part is an element or radical that is less electronegative than the halogen, to make a, e.g., fluoride, chloride, or theoretically tennesside compound. The alkali metals combine directly with halogens under appropriate conditions forming halides of the general formula, MX. Many salts are halides; the hal- syllable in halide and halite reflects this correlation. All Group 1 metals form halides that are white solids at room temperature.

Potassium bromide (KBr) is a salt, widely used as an anticonvulsant and a sedative in the late 19th and early 20th centuries, with over-the-counter use extending to 1975 in the US. Its action is due to the bromide ion. Potassium bromide is used as a veterinary drug, as an antiepileptic medication for dogs.

A chiller is a machine that removes heat from a liquid via a vapor-compression, adsorption refrigeration, or absorption refrigeration cycles. This liquid can then be circulated through a heat exchanger to cool equipment, or another process stream. As a necessary by-product, refrigeration creates waste heat that must be exhausted to ambience, or for greater efficiency, recovered for heating purposes. Vapor compression chillers may use any of a number of different types of compressors. Most common today are the hermetic scroll, semi-hermetic screw, or centrifugal compressors. The condensing side of the chiller can be either air or water cooled. Even when liquid cooled, the chiller is often cooled by an induced or forced draft cooling tower. Absorption and adsorption chillers require a heat source to function.

Hydrogen bromide is the inorganic compound with the formula HBr. It is a hydrogen halide consisting of hydrogen and bromine. A colorless gas, it dissolve in water, forming hydrobromic acid, which is saturated at 68.85% HBr by weight at room temperature. Aqueous solutions that are 47.6% HBr by mass form a constant-boiling azeotrope mixture that boils at 124.3 °C. Boiling less concentrated solutions releases H2O until the constant-boiling mixture composition is reached.

Sodium bromide is an inorganic compound with the formula NaBr. It is a high-melting white, crystalline solid that resembles sodium chloride. It is a widely used source of the bromide ion and has many applications.

Hydrogen halides are diatomic inorganic compounds with the formula HX where X is one of the halogens: fluorine, chlorine, bromine, iodine, or astatine. Hydrogen halides are gases that dissolve in water to give acids which are commonly known as hydrohalic acids.

1-Bromobutane is the organobromine compound with the formula CH3(CH2)3Br. It is a colorless liquid, although impure samples appear yellowish. It is insoluble in water, but soluble in organic solvents. It is a primarily used as a source of the butyl group in organic synthesis. It is one of several isomers of butyl bromide.

Lithium bromide (LiBr) is a chemical compound of lithium and bromine. Its extreme hygroscopic character makes LiBr useful as a desiccant in certain air conditioning systems.
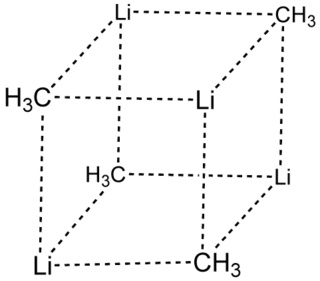
Methyllithium is the simplest organolithium reagent with the empirical formula CH3Li. This s-block organometallic compound adopts an oligomeric structure both in solution and in the solid state. This highly reactive compound, invariably used as a solution in ethers, is a reagent in organic synthesis as well as organometallic chemistry. Operations involving methyllithium require anhydrous conditions, because the compound is highly reactive toward water. Oxygen and carbon dioxide are also incompatible with MeLi. Methyllithium is usually not prepared, but purchased as a solution in various ethers.

Iron(II) bromide is an inorganic compound with the chemical formula FeBr2. The anhydrous compound is a yellow or brownish-colored paramagnetic solid. Several hydrates of FeBr2 are also known, all being pale colored solids. It is a common precursor to other iron compounds in research laboratory, but no applications exist for this compound.

Bis(trimethylsilyl)mercury is a chemical reagent with the formula (CH3)3-Si-Hg-Si-(CH3)3.

Silver hexafluorophosphate, sometimes referred to "silver PF-6," is an inorganic compound with the chemical formula AgPF6.

Barium bromide is the chemical compound with the formula BaBr2. Like barium chloride, it dissolves well in water and is toxic.

Cobalt(II) bromide (CoBr2) is an inorganic compound. In its anhydrous form, it is a green solid that is soluble in water, used primarily as a catalyst in some processes.

Tin(IV) bromide is the chemical compound SnBr4. It is a colourless low melting solid. SnBr4 can be prepared by reaction of the elements at normal temperatures:

Phenacyl bromide is the organic compound with the formula C6H5C(O)CH2Br. This colourless solid is a powerful lachrymator as well as a useful precursor to other organic compounds.

Ethylmagnesium bromide is a Grignard reagent with formula C2H5MgBr. It is widely used in the laboratory synthesis of organic compounds.
The tetrabromonickelate anion contains a doubly-charged nickel atom (Ni2+) surrounded by four bromide ions in a tetrahedral arrangement. The formula is [NiBr4]2−.


















