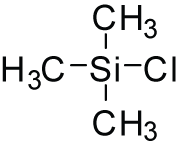Diacetylene (also known as butadiyne) is the organic compound with the formula C4H2. It is the simplest compound containing two triple bonds. It is first in the series of polyynes, which are of theoretical but not of practical interest.

Tetrahedrane is a hypothetical platonic hydrocarbon with chemical formula C4H4 and a tetrahedral structure. The molecule would be subject to considerable angle strain and has not been synthesized as of 2023. However, a number of derivatives have been prepared. In a more general sense, the term tetrahedranes is used to describe a class of molecules and ions with related structure, e.g. white phosphorus.

A trimethylsilyl group (abbreviated TMS) is a functional group in organic chemistry. This group consists of three methyl groups bonded to a silicon atom [−Si(CH3)3], which is in turn bonded to the rest of a molecule. This structural group is characterized by chemical inertness and a large molecular volume, which makes it useful in a number of applications.

Trimethylsilyldiazomethane is the organosilicon compound with the formula (CH3)3SiCHN2. It is classified as a diazo compound. Trimethylsilyldiazomethane, which is a commercially available, reagent used in organic chemistry as a methylating agent of carboxylic acids. Its behavior is akin to the reagent diazomethane, but the trimethylsilyl (TMS) analog is nonexplosive.
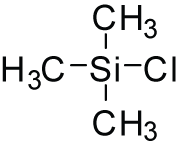
Trimethylsilyl chloride, also known as chlorotrimethylsilane is an organosilicon compound, with the formula (CH3)3SiCl, often abbreviated Me3SiCl or TMSCl. It is a colourless volatile liquid that is stable in the absence of water. It is widely used in organic chemistry.
Bis(trimethylsilyl)acetamide (BSA) is an organosilicon compound with the formula MeC(OSiMe3)NSiMe3 (Me = CH3). It is a colorless liquid that is soluble in diverse organic solvents, but reacts rapidly with moisture and solvents containing OH and NH groups. It is used in analytical chemistry to increase the volatility of analytes, e.g., for gas chromatography. It is also used to introduce the trimethylsilyl protecting group in organic synthesis. A related reagent is N,O-bis(trimethylsilyl)trifluoroacetamide (BSTFA).
Silylation is the introduction of one or more (usually) substituted silyl groups (R3Si) to a molecule. Silylations are core methods for production of organosilicon chemistry. Silanization involves similar methods but usually refers to attachment of silyl groups to solids.

Organoindium chemistry is the chemistry of compounds containing In-C bonds. The main application of organoindium chemistry is in the preparation of semiconducting components for microelectronic applications. The area is also of some interest in organic synthesis. Most organoindium compounds feature the In(III) oxidation state, akin to its lighter congeners Ga(III) and B(III).
Organosodium chemistry is the chemistry of organometallic compounds containing a carbon to sodium chemical bond. The application of organosodium compounds in chemistry is limited in part due to competition from organolithium compounds, which are commercially available and exhibit more convenient reactivity.
Carbene analogs in chemistry are carbenes with the carbon atom replaced by another chemical element. Just as regular carbenes they appear in chemical reactions as reactive intermediates and with special precautions they can be stabilized and isolated as chemical compounds. Carbenes have some practical utility in organic synthesis but carbene analogs are mostly laboratory curiosities only investigated in academia. Carbene analogs are known for elements of group 13, group 14, group 15 and group 16.

tert-Butylphosphaacetylene is an organophosphorus compound. Abbreviated t-BuCP, it was the first example of an isolable phosphaalkyne. Prior to its synthesis, the double bond rule had suggested that elements of Period 3 and higher were unable to form double or triple bonds with lighter main group elements because of weak orbital overlap. The synthesis of t-BuCP discredited much of the double bond rule and opened new studies into the formation of unsaturated phosphorus compounds.

Bis(trimethylsilyl)acetylene (BTMSA) is an organosilicon compound with the formula Me3SiC≡CSiMe3 (Me = methyl). It is a colorless liquid that is soluble in organic solvents. This compound is used as a surrogate for acetylene.
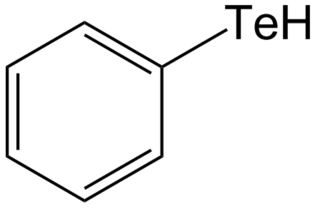
Tellurols are analogues of alcohols and phenols where tellurium replaces oxygen. Tellurols, selenols, and thiols have similar properties, but tellurols are the least stable. Although they are fundamental representatives of organotellurium compounds, tellurols are lightly studied because of their instability. Tellurol derivatives include telluroesters and tellurocyanates (RTeCN).
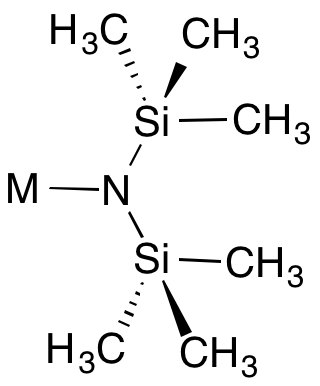
Metal bis(trimethylsilyl)amides are coordination complexes composed of a cationic metal M with anionic bis(trimethylsilyl)amide ligands (the −N 2 monovalent anion, or −N 2 monovalent group, and are part of a broader category of metal amides.
Phosphinoimidates, also known as phophinimides, are the anions derived from phosphine imides with the structure [R3P=N]− (R = alkyl or aryl). Phosphinimide ligands are used to for transition metal complexes that are highly active catalysts in some olefin polymerization reactions.

Tris(trimethylsilyl)amine is the simplest tris(trialkylsilyl)amine which are having the general formula (R3Si)3N, in which all three hydrogen atoms of the ammonia are replaced by trimethylsilyl groups (-Si(CH3)3). Tris(trimethylsilyl)amine has been for years in the center of scientific interest as a stable intermediate in chemical nitrogen fixation (i. e. the conversion of atmospheric nitrogen N2 into organic substrates under normal conditions).
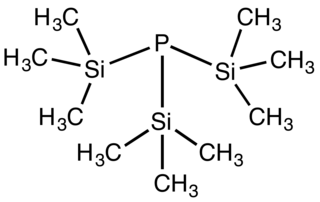
Tris(trimethylsilyl)phosphine is the organophosphorus compound with the formula P(SiMe3)3 (Me = methyl). It is a colorless liquid that ignites in air and hydrolyses readily.
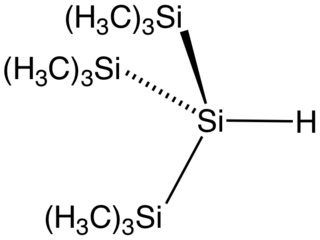
Tris(trimethylsilyl)silane is the organosilicon compound with the formula (Me3Si)3SiH (where Me = CH3). It is a colorless liquid that is classified as a hydrosilane since it contains an Si-H bond. The compound is notable as having a weak Si-H bond, with a bond dissociation energy estimated at 84 kcal/mol. For comparison, the Si-H bond in trimethylsilane is 94 kcal/mol. With such a weak bond, the compound is used as a reagent to deliver hydrogen atoms. The compound has been described as an environmentally benign analogue of tributyltin hydride.

Tetrakis(trimethylsilyl)silane is the organosilicon compound with the formula (Me3Si)4Si (where Me = CH3). It is a colorless sublimable solid with a high melting point. The molecule has tetrahedral symmetry. The compound is notable as having silicon bonded to four other silicon atoms, like in elemental silicon.

(Trimethylsilyl)methyllithium is classified both as an organolithium compound and an organosilicon compound. It has the empirical formula LiCH2Si(CH3)3, often abbreviated LiCH2TMS. It crystallizes as the hexagonal prismatic hexamer [LiCH2TMS]6, akin to some polymorphs of methyllithium. Many adducts have been characterized including the diethyl ether complexed cubane [Li4(μ3-CH2TMS)4(Et2O)2] and [Li2(μ-CH2TMS)2(TMEDA)2].


![Structure of [InC(tms)3]4, an In(I) tetrahedrane (dark gray = In, orange = Si). YUZZOI.svg](http://upload.wikimedia.org/wikipedia/commons/thumb/e/ec/YUZZOI.svg/222px-YUZZOI.svg.png)