
Epoxy is the family of basic components or cured end products of epoxy resins. Epoxy resins, also known as polyepoxides, are a class of reactive prepolymers and polymers which contain epoxide groups. The epoxide functional group is also collectively called epoxy. The IUPAC name for an epoxide group is an oxirane.

In organic chemistry, an epoxide is a cyclic ether, where the ether forms a three-atom ring: two atoms of carbon and one atom of oxygen. This triangular structure has substantial ring strain, making epoxides highly reactive, more so than other ethers. They are produced on a large scale for many applications. In general, low molecular weight epoxides are colourless and nonpolar, and often volatile.

The Johnson–Corey–Chaykovsky reaction is a chemical reaction used in organic chemistry for the synthesis of epoxides, aziridines, and cyclopropanes. It was discovered in 1961 by A. William Johnson and developed significantly by E. J. Corey and Michael Chaykovsky. The reaction involves addition of a sulfur ylide to a ketone, aldehyde, imine, or enone to produce the corresponding 3-membered ring. The reaction is diastereoselective favoring trans substitution in the product regardless of the initial stereochemistry. The synthesis of epoxides via this method serves as an important retrosynthetic alternative to the traditional epoxidation reactions of olefins.
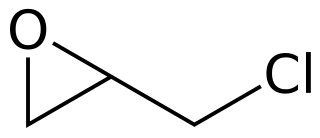
Epichlorohydrin is an organochlorine compound and an epoxide. Despite its name, it is not a halohydrin. It is a colorless liquid with a pungent, garlic-like odor, moderately soluble in water, but miscible with most polar organic solvents. It is a chiral molecule generally existing as a racemic mixture of right-handed and left-handed enantiomers. Epichlorohydrin is a highly reactive electrophilic compound and is used in the production of glycerol, plastics, epoxy glues and resins, epoxy diluents and elastomers.
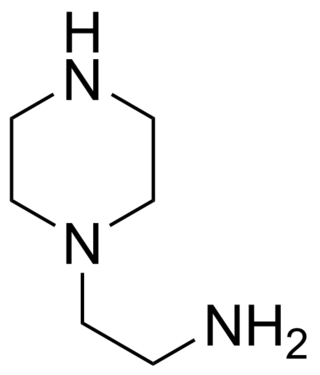
Aminoethylpiperazine (AEP) is a derivative of piperazine. This ethyleneamine contains three nitrogen atoms; one primary, one secondary and one tertiary. It is a corrosive organic liquid and can cause second or third degree burns. Aminoethylpiperazine can also cause pulmonary edema as a result of inhalation. It is REACH and TSCA registered.

Bisphenol A diglycidyl ether is an organic compound and is a liquid epoxy resin. The compound is a colorless viscous liquid. It is a key component of many epoxy resin formulations. Addition of further Bisphenol A and a catalyst and heat can produce Bisphenol A glycidyl ether epoxy resins of higher molecular weight that are solid.

3,4-Epoxycyclohexanecarboxylate methyl ester is a cycloaliphatic epoxide, which is added as monovalent monomer to other monomers for the production of crosslinked epoxy resins. 3,4-Epoxycyclohexanecarboxylate methyl ester itself would give a linear polymer when homopolymerized.

3,4-Epoxycyclohexylmethyl-3',4'-epoxycyclohexane carboxylate (ECC) is a cycloaliphatic epoxy resin which is used in many industrial applications. It reacts by cationic polymerization using thermolatent photoinitiators to form crosslinked insoluble thermosets. Formulations based on cycloaliphatic epoxy resins such as ECC are known to form by curing thermosets with high heat and chemical resistance and good adhesion.

Allyl glycidyl ether is an organic compound used in adhesives and sealants and as a monomer for polymerization reactions. It is formally the condensation product of allyl alcohol and glycidol via an ether linkage. Because it contains both an alkene and an epoxide group, either group can be reacted selectively to yield a product where the other functional group remains intact for future reactions.

ContraPest is a contraceptive pest control tool designed to reduce fertility in brown and black rats. It was developed by the U.S. biotechnology company SenesTech.

Trifluoroperacetic acid is an organofluorine compound, the peroxy acid analog of trifluoroacetic acid, with the condensed structural formula CF
3COOOH. It is a strong oxidizing agent for organic oxidation reactions, such as in Baeyer–Villiger oxidations of ketones. It is the most reactive of the organic peroxy acids, allowing it to successfully oxidise relatively unreactive alkenes to epoxides where other peroxy acids are ineffective. It can also oxidise the chalcogens in some functional groups, such as by transforming selenoethers to selones. It is a potentially explosive material and is not commercially available, but it can be quickly prepared as needed. Its use as a laboratory reagent was pioneered and developed by William D. Emmons.
SenesTech, Inc. is an agricultural biotechnology life-sciences company, specializing in fertility management as a form of pest control. The company's primary product, ContraPest is designed to make brown and black rats infertile. SenesTech is headquartered in Phoenix, Arizona.
1,4-Butanediol diglycidyl ether (B14DODGE) is an organic chemical in the glycidyl ether family. It is aliphatic and a colorless liquid. It has two epoxide (oxirane) groups per molecule. Its main use is in modifying epoxy resins especially viscosity reduction.
1,6-Hexanediol diglycidyl ether is an organic chemical in the glycidyl ether family. It is an aliphatic compound that is a colorless liquid. It has two epoxide (oxirane) groups per molecule. Its main use is in modifying epoxy resins especially viscosity reduction whilst flexibilizing. It is REACH registered.

Trimethylolpropane triglycidyl ether (TMPTGE) is an organic chemical in the glycidyl ether family. It has the formula C15H26O6 and the IUPAC name is 2-[2,2-bis(oxiran-2-ylmethoxymethyl)butoxymethyl]oxirane, and the CAS number 3454-29-3. It also has another CAS number of 30499-70-8 A key use is as a modifier for epoxy resins as a reactive diluent.

Castor oil glycidyl ether is a liquid organic chemical in the glycidyl ether family. It is sometimes called castor oil triglycidyl ether. It has the theoretical formula C66H116O12 and the CAS number 14228-73-0. The IUPAC name is 2,3-bis[12-(oxiran-2-ylmethoxy)octadec-9-enoyloxy]propyl 12-(oxiran-2-ylmethoxy)octadec-9-enoate. A key use is acting as a modifier for epoxy resins as a reactive diluent that adds flexibility and improved mechanical properties.
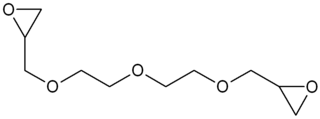
Diethylene glycol diglycidyl ether (DEGDGE) is an organic chemical in the glycidyl ether family with the formula C10H18O5.. The oxirane functionality makes it useful as a reactive diluent for epoxy resin viscosity reduction.

Diglycidyl resorcinol ether, also called Resorcinol diglycidyl ether (RDGE) is a liquid aromatic organic chemical compound and chemically a glycidyl ether.

Phenyl glycidyl ether, is a liquid aromatic organic chemical in the glycidyl ether class of compounds. It has the formula C9H10O2. It has the CAS Registry Number 122-60-1 and the IUPAC name of 2-(phenoxymethyl)oxirane. A key use is in the viscosity reduction of epoxy resin systems. It is REACH registered and on EINECS under the name 2,3-epoxypropyl phenyl ether.
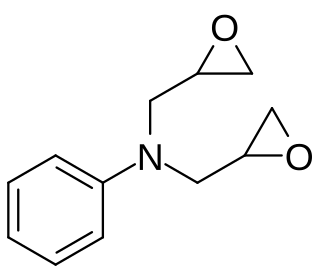
Diglycidyl aniline is an aromatic organic chemical in the glycidyl compound family. It is used to reduce the viscosity of epoxy resin systems. It has the empirical formula C12H15NO2 and the IUPAC name is N,N-bis(oxiran-2-ylmethyl)aniline. The CAS number is 2095-06-9. It is REACH registered in Europe with the EC number 218-259-5. A key use is in the viscosity reduction of epoxy resin systems functioning as a reactive diluent.
















