| |||
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name 1,2-Dichloroethene | |||
| Other names 1,2-Dichloroethene 1,2-DCE Acetylene dichloride sym-Dichloroethylene | |||
| Identifiers | |||
3D model (JSmol) | |||
| ChEBI | |||
| ChemSpider | |||
| ECHA InfoCard | 100.007.956 | ||
| EC Number |
| ||
| KEGG | |||
PubChem CID | |||
| UNII |
| ||
CompTox Dashboard (EPA) | |||
| |||
| |||
| Properties | |||
| C2H2Cl2 | |||
| Molar mass | 96.95 g/mol | ||
| Appearance | colorless liquid | ||
| Odor | sweet [1] | ||
| Density | Z: 1.28 g/cm3 E: 1.26 g/cm3 | ||
| Melting point | Z: −81.47 °C E: −49.44 °C | ||
| Boiling point | Z: 60.2 °C E: 48.5 °C | ||
| |||
| Z: 1.9 D E: 0 D | |||
| Hazards | |||
| Flash point | 2–4 °C; 36–39 °F; 275–277 K | ||
| Explosive limits | 5.6–12.8% [2] | ||
| Lethal dose or concentration (LD, LC): | |||
LD50 (median dose) | 770 mg/kg (oral, rat) 1275 mg/kg (oral, rat, trans-isomer) [3] | ||
LC50 (median concentration) | 21,273 ppm (mouse, 6 hr, trans-isomer) [3] | ||
LCLo (lowest published) | 16,000 ppm (rat, 6 hr, cis-isomer) [3] | ||
| NIOSH (US health exposure limits): | |||
PEL (Permissible) | TWA 200 ppm (790 mg/m3) [2] | ||
REL (Recommended) | TWA 200 ppm (790 mg/m3) [2] | ||
IDLH (Immediate danger) | 1000 ppm [2] | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |||
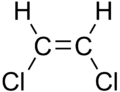
1,2-Dichloroethylene (1,2-DCE) is a pair of organochlorine compounds with the molecular formula C2H2Cl2. The two compounds are isomers, each being colorless liquids with a sweet odor. It can exist as either of two geometric isomers, cis-1,2-dichloroethene or trans-1,2-dichloroethene, but is often used as a mixture of the two. They have modest solubility in water. These compounds have some applications as a degreasing solvent. [1] In contrast to most cis-trans compounds, the Z isomer (cis) is more stable than the E isomer (trans) by 0.4 kcal/mol. [4]