 | |
| Names | |
|---|---|
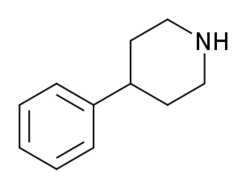
| Preferred IUPAC name 4-Phenylpiperidine | |
| Identifiers | |
3D model (JSmol) | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.011.130 |
PubChem CID | |
| UNII | |
CompTox Dashboard (EPA) | |
| |
| |
| Properties | |
| C11H15N | |
| Molar mass | 161.248 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
4-Phenylpiperidine is a chemical compound. It features a benzene ring bound to a piperidine ring.
Contents
4-Phenylpiperidine is the base structure for a variety of opioids, such as pethidine (meperidine), ketobemidone, alvimopan, loperamide, and diphenoxylate.