Amyloid beta A4 precursor protein-binding family B member 1 is a protein that in humans is encoded by the APBB1 gene. [5] [6] [7]
Amyloid beta A4 precursor protein-binding family B member 1 is a protein that in humans is encoded by the APBB1 gene. [5] [6] [7]
The protein encoded by this gene is a member of the Fe65 protein family. It is an adaptor protein localized in the nucleus. It interacts with the Alzheimer's disease amyloid precursor protein (APP), transcription factor CP2/LSF/LBP1 and the low-density lipoprotein receptor-related protein. APP functions as a cytosolic anchoring site that can prevent the gene product's nuclear translocation. This encoded protein could play an important role in the pathogenesis of Alzheimer's disease. It is thought to regulate transcription. Also it is observed to block cell cycle progression by downregulating thymidylate synthase expression. Multiple alternatively spliced transcript variants have been described for this gene but some of their full length sequence is not known. [7]
APBB1 has been shown to interact with APLP2, [8] [9] TFCP2, [10] LRP1 [11] and Amyloid precursor protein. [8] [9] [11] [12] [13]

Amyloid-beta precursor protein (APP) is an integral membrane protein expressed in many tissues and concentrated in the synapses of neurons. It functions as a cell surface receptor and has been implicated as a regulator of synapse formation, neural plasticity, antimicrobial activity, and iron export. It is coded for by the gene APP and regulated by substrate presentation. APP is best known as the precursor molecule whose proteolysis generates amyloid beta (Aβ), a polypeptide containing 37 to 49 amino acid residues, whose amyloid fibrillar form is the primary component of amyloid plaques found in the brains of Alzheimer's disease patients.

The Disabled-1 (Dab1) gene encodes a key regulator of Reelin signaling. Reelin is a large glycoprotein secreted by neurons of the developing brain, particularly Cajal-Retzius cells. DAB1 functions downstream of Reln in a signaling pathway that controls cell positioning in the developing brain and during adult neurogenesis. It docks to the intracellular part of the Reelin very low density lipoprotein receptor (VLDLR) and apoE receptor type 2 (ApoER2) and becomes tyrosine-phosphorylated following binding of Reelin to cortical neurons. In mice, mutations of Dab1 and Reelin generate identical phenotypes. In humans, Reelin mutations are associated with brain malformations and mental retardation. In mice, Dab1 mutation results in the scrambler mouse phenotype.

The low-density lipoprotein receptor gene family codes for a class of structurally related cell surface receptors that fulfill diverse biological functions in different organs, tissues, and cell types. The role that is most commonly associated with this evolutionarily ancient family is cholesterol homeostasis. In humans, excess cholesterol in the blood is captured by low-density lipoprotein (LDL) and removed by the liver via endocytosis of the LDL receptor. Recent evidence indicates that the members of the LDL receptor gene family are active in the cell signalling pathways between specialized cells in many, if not all, multicellular organisms.
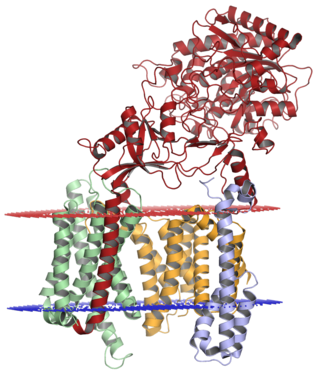
Gamma secretase is a multi-subunit protease complex, an integral membrane protein, that cleaves single-pass transmembrane proteins at residues within the transmembrane domain. Proteases of this type are known as intramembrane proteases. The most well-known substrate of gamma secretase is amyloid precursor protein, a large integral membrane protein that, when cleaved by both gamma and beta secretase, produces a short 37-43 amino acid peptide called amyloid beta whose abnormally folded fibrillar form is the primary component of amyloid plaques found in the brains of Alzheimer's disease patients. Gamma secretase is also critical in the related processing of several other type I integral membrane proteins, such as Notch, ErbB4, E-cadherin, N-cadherin, ephrin-B2, or CD44.

Adapter molecule crk also known as proto-oncogene c-Crk is a protein that in humans is encoded by the CRK gene.

Low-density lipoprotein receptor-related protein 8 (LRP8), also known as apolipoprotein E receptor 2 (ApoER2), is a protein that in humans is encoded by the LRP8 gene. ApoER2 is a cell surface receptor that is part of the low-density lipoprotein receptor family. These receptors function in signal transduction and endocytosis of specific ligands. Through interactions with one of its ligands, reelin, ApoER2 plays an important role in embryonic neuronal migration and postnatal long-term potentiation. Another LDL family receptor, VLDLR, also interacts with reelin, and together these two receptors influence brain development and function. Decreased expression of ApoER2 is associated with certain neurological diseases.

Presenilin-1(PS-1) is a presenilin protein that in humans is encoded by the PSEN1 gene. Presenilin-1 is one of the four core proteins in the gamma secretase complex, which is considered to play an important role in generation of amyloid beta (Aβ) from amyloid-beta precursor protein (APP). Accumulation of amyloid beta is associated with the onset of Alzheimer's disease.

Cbl is a mammalian gene family. CBL gene, a part of the Cbl family, encodes the protein CBL which is an E3 ubiquitin-protein ligase involved in cell signalling and protein ubiquitination. Mutations to this gene have been implicated in a number of human cancers, particularly acute myeloid leukaemia.

C-jun-amino-terminal kinase-interacting protein 1 is an enzyme that in humans is encoded by the MAPK8IP1 gene.

Presenilin-2 is a protein that is encoded by the PSEN2 gene.

5'-AMP-activated protein kinase subunit beta-1 is an enzyme that in humans is encoded by the PRKAB1 gene.

GIPC PDZ domain containing family, member 1 (GIPC1) is a protein that in humans is encoded by the GIPC1 gene. GIPC was originally identified as it binds specifically to the C terminus of RGS-GAIP, a protein involved in the regulation of G protein signaling. GIPC is an acronym for "GAIP Interacting Protein C-terminus". RGS proteins are "Regulators of G protein Signaling" and RGS-GAIP is a "GTPase Activator protein for Gαi/Gαq", which are two major subtypes of Gα proteins. The human GIPC1 molecule is 333 amino acids or about 36 kDa in molecular size and consists of a central PDZ domain, a compact protein module which mediates specific protein-protein interactions. The RGS-GAIP protein interacts with this domain and many other proteins interact here or at other parts of the GIPC1 molecule. As a result, GIPC1 was independently discovered by several other groups and has a variety of alternate names, including synectin, C19orf3, RGS19IP1 and others. The GIPC1 gene family in mammals consisting of three members, so the first discovered, originally named GIPC, is now generally called GIPC1, with the other two being named GIPC2 and GIPC3. The three human proteins are about 60% identical in protein sequence. GIPC1 has been shown to interact with a variety of other receptor and cytoskeletal proteins including the GLUT1 receptor, ACTN1, KIF1B, MYO6, PLEKHG5, SDC4/syndecan-4, SEMA4C/semaphorin-4 and HTLV-I Tax. The general function of GIPC family proteins therefore appears to be mediating specific interactions between proteins involved in G protein signaling and membrane translocation.
Amyloid beta A4 precursor protein-binding family A member 1 is a protein that in humans is encoded by the APBA1 gene.

Amyloid-like protein 1, also known as APLP1, is a protein that in humans is encoded by the APLP1 gene. APLP1 along with APLP2 are important modulators of glucose and insulin homeostasis.

Alpha-globin transcription factor CP2 is a protein that in humans is encoded by the TFCP2 gene.

Amyloid beta A4 precursor protein-binding family A member 2 is a protein that in humans is encoded by the APBA2 gene.

Amyloid beta A4 precursor protein-binding family B member 2 is a protein that in humans is encoded by the APBB2 gene.
Amyloid beta A4 precursor protein-binding family B member 3 is a protein that in humans is encoded by the APBB3 gene.

Collagen alpha-1(XXV) chain is a protein that in humans is encoded by the COL25A1 gene.

Amyloid beta A4 precursor protein-binding family B member 1-interacting protein (APBB1IP), also known as APBB1-interacting protein 1 or Rap1-GTP-interacting adapter molecule (RIAM) is a protein that in humans is encoded by the APBB1IP gene.