Spectroscopy is the field of study that measures and interprets the electromagnetic spectra that result from the interaction between electromagnetic radiation and matter as a function of the wavelength or frequency of the radiation. In simpler terms, spectroscopy is the precise study of color as generalized from visible light to all bands of the electromagnetic spectrum.
Fourier-transform spectroscopy is a measurement technique whereby spectra are collected based on measurements of the coherence of a radiative source, using time-domain or space-domain measurements of the radiation, electromagnetic or not. It can be applied to a variety of types of spectroscopy including optical spectroscopy, infrared spectroscopy, nuclear magnetic resonance (NMR) and magnetic resonance spectroscopic imaging (MRSI), mass spectrometry and electron spin resonance spectroscopy.

Raman spectroscopy is a spectroscopic technique typically used to determine vibrational modes of molecules, although rotational and other low-frequency modes of systems may also be observed. Raman spectroscopy is commonly used in chemistry to provide a structural fingerprint by which molecules can be identified.
Interferometry is a technique which uses the interference of superimposed waves to extract information. Interferometry typically uses electromagnetic waves and is an important investigative technique in the fields of astronomy, fiber optics, engineering metrology, optical metrology, oceanography, seismology, spectroscopy, quantum mechanics, nuclear and particle physics, plasma physics, biomolecular interactions, surface profiling, microfluidics, mechanical stress/strain measurement, velocimetry, optometry, and making holograms.
Optics is the branch of physics which involves the behavior and properties of light, including its interactions with matter and the construction of instruments that use or detect it. Optics usually describes the behavior of visible, ultraviolet, and infrared light. Because light is an electromagnetic wave, other forms of electromagnetic radiation such as X-rays, microwaves, and radio waves exhibit similar properties.

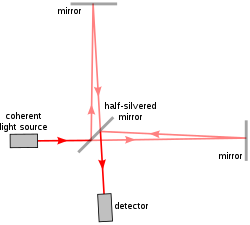
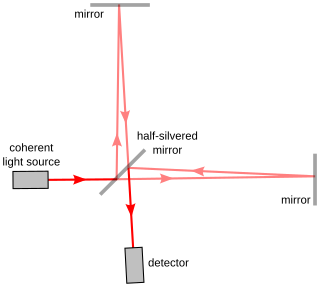
The Michelson interferometer is a common configuration for optical interferometry and was invented by the 19/20th-century American physicist Albert Abraham Michelson. Using a beam splitter, a light source is split into two arms. Each of those light beams is reflected back toward the beamsplitter which then combines their amplitudes using the superposition principle. The resulting interference pattern that is not directed back toward the source is typically directed to some type of photoelectric detector or camera. For different applications of the interferometer, the two light paths can be with different lengths or incorporate optical elements or even materials under test.

Optical coherence tomography (OCT) is an imaging technique that uses interferometry with short-coherence-length light to obtain micrometer-level depth resolution and uses transverse scanning of the light beam to form two- and three-dimensional images from light reflected from within biological tissue or other scattering media. Short-coherence-length light can be obtained using a superluminescent diode (SLD) with a broad spectral bandwidth or a broadly tunable laser with narrow linewidth. The first demonstration of OCT imaging was published by a team from MIT and Harvard Medical School in a 1991 article in the journal Science. The article introduced the term “OCT” to credit its derivation from optical coherence-domain reflectometry, in which the axial resolution is based on temporal coherence. The first demonstrations of in vivo OCT imaging quickly followed.

In physics, backscatter is the reflection of waves, particles, or signals back to the direction from which they came. It is usually a diffuse reflection due to scattering, as opposed to specular reflection as from a mirror, although specular backscattering can occur at normal incidence with a surface. Backscattering has important applications in astronomy, photography, and medical ultrasonography. The opposite effect is forward scatter, e.g. when a translucent material like a cloud diffuses sunlight, giving soft light.

A profilometer is a measuring instrument used to measure a surface's profile, in order to quantify its roughness. Critical dimensions as step, curvature, flatness are computed from the surface topography.

Neutron spin echo spectroscopy is an inelastic neutron scattering technique invented by Ferenc Mezei in the 1970s and developed in collaboration with John Hayter. In recognition of his work and in other areas, Mezei was awarded the first Walter Haelg Prize in 1999.
Diffusing-wave spectroscopy (DWS) is an optical technique derived from dynamic light scattering (DLS) that studies the dynamics of scattered light in the limit of strong multiple scattering. It has been widely used in the past to study colloidal suspensions, emulsions, foams, gels, biological media and other forms of soft matter. If carefully calibrated, DWS allows the quantitative measurement of microscopic motion in a soft material, from which the rheological properties of the complex medium can be extracted via the microrheology approach.
Speckle, speckle pattern, or speckle noise is a granular noise texture degrading the quality as a consequence of interference among wavefronts in coherent imaging systems, such as radar, synthetic aperture radar (SAR), medical ultrasound and optical coherence tomography. Speckle is not external noise; rather, it is an inherent fluctuation in diffuse reflections, because the scatterers are not identical for each cell, and the coherent illumination wave is highly sensitive to small variations in phase changes.
The technique of vibrational analysis with scanning probe microscopy allows probing vibrational properties of materials at the submicrometer scale, and even of individual molecules. This is accomplished by integrating scanning probe microscopy (SPM) and vibrational spectroscopy. This combination allows for much higher spatial resolution than can be achieved with conventional Raman/FTIR instrumentation. The technique is also nondestructive, requires non-extensive sample preparation, and provides more contrast such as intensity contrast, polarization contrast and wavelength contrast, as well as providing specific chemical information and topography images simultaneously.
Optical coherence tomography (OCT) is a technique that displays images of the tissue by using the backscattered light.
Multiple scattering low coherence interferometry (ms/LCI) is an imaging technique that relies on analyzing multiply scattered light in order to capture depth-resolved images from optical scattering media. With current applications primarily in medical imaging, has the advantage of a higher range since forward scattered light attenuates less with depth when compared to the specularly reflected light that is assessed in more conventional imaging methods such as optical coherence tomography. This allows ms/LCI to image through up to 90 mean free scattering paths, compared to roughly 27 scattering MFPs in OCT and 1–2 scattering MFPs in confocal microscopy.
Speckle variance optical coherence tomography (SV-OCT) is an imaging algorithm for functional optical imaging. Optical coherence tomography is an imaging modality that uses low-coherence interferometry to obtain high resolution, depth-resolved volumetric images. OCT can be used to capture functional images of blood flow, a technique known as optical coherence tomography angiography (OCT-A). SV-OCT is one method for OCT-A that uses the variance of consecutively acquired images to detect flow at the micron scale. SV-OCT can be used to measure the microvasculature of tissue. In particular, it is useful in ophthalmology for visualizing blood flow in retinal and choroidal regions of the eye, which can provide information on the pathophysiology of diseases.
Spectroscopic optical coherence tomography (SOCT) is an optical imaging and sensing technique, which provides localized spectroscopic information of a sample based on the principles of optical coherence tomography (OCT) and low coherence interferometry. The general principles behind SOCT arise from the large optical bandwidths involved in OCT, where information on the spectral content of backscattered light can be obtained by detection and processing of the interferometric OCT signal. SOCT signal can be used to quantify depth-resolved spectra to retrieve the concentration of tissue chromophores, characterize tissue light scattering, and/or used as a functional contrast enhancement for conventional OCT imaging.
Light scattering spectroscopy (LSS) is a spectroscopic technique typically used to evaluate morphological changes in epithelial cells in order to study mucosal tissue and detect early cancer and precancer.
Dual-axis optical coherence tomography (DA-OCT) is an imaging modality that is based on the principles of optical coherence tomography (OCT). These techniques are largely used for medical imaging. OCT is non-invasive and non-contact. It allows for real-time, in situ imaging and provides high image resolution. OCT is analogous to ultrasound but relies on light waves, which makes it faster than ultrasound. In general, OCT has proven to be compact and portable. It is compatible with arterial catheters and endoscopes, which helps diagnose diseases within long internal cavities, including the esophagus and coronary arteries.