Carbon oxohalides are a group of chemical compounds that contain only carbon, oxygen and halogen atoms: fluorine, chlorine, bromine and iodine. They include carbonyl halides, COX2, and oxalyl halides, C2X2O2, where X = F, Cl, Br or I. The halogen atoms X do not have to be identical; they differ in mixed oxohalides. Most combinations of halogens exist but carbonyl iodide, COI2, is unknown. The carbon–oxygen bond length in carbonyl halides (1.13–1.17 Å) [1] is shorter than in other carbonyl compounds [2] such as aldehydes and ketones, carboxylic acids, esters and amides (1.20–1.21 Å). [3] They are reactive reagents for halogenation, [1] acylation and dehydration reactions. [4]
| Name | Formula | Melting point / °C | Boiling point / °C | C–O bond length / Å |
|---|---|---|---|---|
| Carbonyl fluoride | COF2 | −114 | −83.1 | 1.174 |
| Carbonyl chloride fluoride | COFCl | −42 | ||
| Carbonyl bromide fluoride | COFBr | −20.6 | ||
| Phosgene | COCl2 | −127.8 | +7.6 | 1.166 |
| Carbonyl iodide fluoride | COFI | −90 | +23.4 | |
| Carbonyl bromide chloride | COClBr | |||
| Carbonyl bromide | COBr2 | +64.5 | 1.13 | |
| Oxalyl fluoride | C2F2O2 | −3 | +26.6 | |
| Oxalyl chloride | C2Cl2O2 | −16 | +63 | |
| Diphosgene | C2Cl4O2 | −57 | +128 | |
| Triphosgene | C3Cl6O3 | +80 | +206 | |

Bromine is a chemical element with the symbol Br and atomic number 35. It is a volatile red-brown liquid at room temperature that evaporates readily to form a similarly coloured vapour. Its properties are intermediate between those of chlorine and iodine. Isolated independently by two chemists, Carl Jacob Löwig and Antoine Jérôme Balard, its name was derived from the Ancient Greek βρῶμος (bromos) meaning "stench", referring to its sharp and pungent smell.

In organic chemistry, a ketone is a functional group with the structure R−C(=O)−R', where R and R' can be a variety of carbon-containing substituents. Ketones contain a carbonyl group −C(=O)−. The simplest ketone is acetone, with the formula (CH3)2CO. Many ketones are of great importance in biology and in industry. Examples include many sugars (ketoses), many steroids, and the solvent acetone.

A nonmetal is a chemical element that, in the broadest sense of the term, has a relatively low density and high electronegativity; they range from colorless gases to shiny solids. They are usually poor conductors of heat and electricity, and brittle or crumbly when solid due to their electrons having low mobility. In contrast, metals are good conductors and most are easily flattened into sheets and drawn into wires since their electrons are generally free-moving. Nonmetal atoms tend to attract electrons in chemical reactions and to form acidic compounds.

In organic chemistry, a carbonyl group is a functional group with the formula C=O, composed of a carbon atom double-bonded to an oxygen atom, and it is divalent at the C atom. It is common to several classes of organic compounds, as part of many larger functional groups. A compound containing a carbonyl group is often referred to as a carbonyl compound.
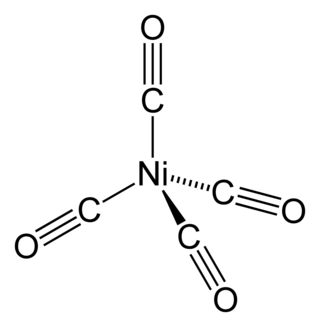
Nickel carbonyl (IUPAC name: tetracarbonylnickel) is a nickel(0) organometallic compound with the formula Ni(CO)4. This colorless liquid is the principal carbonyl of nickel. It is an intermediate in the Mond process for producing very high-purity nickel and a reagent in organometallic chemistry, although the Mond Process has fallen out of common usage due to the health hazards in working with the compound. Nickel carbonyl is one of the most dangerous substances yet encountered in nickel chemistry due to its very high toxicity, compounded with high volatility and rapid skin absorption.

The Grignard reaction is an organometallic chemical reaction in which carbon alkyl, allyl, vinyl, or aryl magnesium halides are added to the carbonyl groups of either an aldehyde or ketone. This reaction is important for the formation of carbon–carbon bonds.
In chemistry, halogenation is a chemical reaction that entails the introduction of one or more halogens into a compound. Halide-containing compounds are pervasive, making this type of transformation important, e.g. in the production of polymers, drugs. This kind of conversion is in fact so common that a comprehensive overview is challenging. This article mainly deals with halogenation using elemental halogens (F2, Cl2, Br2, I2). Halides are also commonly introduced using salts of the halides and halogen acids. Many specialized reagents exist for and introducing halogens into diverse substrates, e.g. thionyl chloride.
In chemistry, an interhalogen compound is a molecule which contains two or more different halogen atoms and no atoms of elements from any other group.
Phosphorus trifluoride (formula PF3), is a colorless and odorless gas. It is highly toxic and reacts slowly with water. Its main use is as a ligand in metal complexes. As a ligand, it parallels carbon monoxide in metal carbonyls, and indeed its toxicity is due to its binding with the iron in blood hemoglobin in a similar way to carbon monoxide.
Organosulfur chemistry is the study of the properties and synthesis of organosulfur compounds, which are organic compounds that contain sulfur. They are often associated with foul odors, but many of the sweetest compounds known are organosulfur derivatives, e.g., saccharin. Nature abounds with organosulfur compounds—sulfur is vital for life. Of the 20 common amino acids, two are organosulfur compounds, and the antibiotics penicillin and sulfa drugs both contain sulfur. While sulfur-containing antibiotics save many lives, sulfur mustard is a deadly chemical warfare agent. Fossil fuels, coal, petroleum, and natural gas, which are derived from ancient organisms, necessarily contain organosulfur compounds, the removal of which is a major focus of oil refineries.

Metal carbonyls are coordination complexes of transition metals with carbon monoxide ligands. Metal carbonyls are useful in organic synthesis and as catalysts or catalyst precursors in homogeneous catalysis, such as hydroformylation and Reppe chemistry. In the Mond process, nickel tetracarbonyl is used to produce pure nickel. In organometallic chemistry, metal carbonyls serve as precursors for the preparation of other organometallic complexes.

Silver(II) fluoride is a chemical compound with the formula AgF2. It is a rare example of a silver(II) compound. Silver usually exists in its +1 oxidation state. It is used as a fluorinating agent.

A Grignard reagent or Grignard compound is a chemical compound with the general formula R−Mg−X, where X is a halogen and R is an organic group, normally an alkyl or aryl. Two typical examples are methylmagnesium chloride Cl−Mg−CH3 and phenylmagnesium bromide (C6H5)−Mg−Br. They are a subclass of the organomagnesium compounds.
Carbonyl fluoride is a chemical compound with the formula COF2. It is a carbon oxohalide. This gas, like its analog phosgene, is colourless and highly toxic. The molecule is planar with C2v symmetry, bond lengths of 1.174 Å (C=O) and 1.312 Å (C–F), and an F–C–F bond angle of 108.0°.
Silyl enol ethers in organic chemistry are a class of organic compounds that share a common functional group composed of an enolate bonded through its oxygen end to an organosilicon group. They are important intermediates in organic synthesis.
A carbon–oxygen bond is a polar covalent bond between atoms of carbon and oxygen. Carbon–oxygen bonds are found in many inorganic compounds such as carbon oxides and oxohalides, carbonates and metal carbonyls, and in organic compounds such as alcohols, ethers, carbonyl compounds and oxalates. Oxygen has 6 valence electrons of its own and tends to fill its outer shell with 8 electrons by sharing electrons with other atoms to form covalent bonds, accepting electrons to form an anion, or a combination of the two. In neutral compounds, an oxygen atom can form up to two single bonds or one double bond with carbon, while a carbon atom can form up to four single bonds or two double bonds with oxygen.
Carbonyl bromide, also known as bromophosgene, is an organic chemical compound with the chemical formula COBr2. It is a colorless liquid. It is a bromine analogue of phosgene. It is a carbon oxohalide. Carbonyl bromide is a decomposition product of halon compounds used in fire extinguishers.
In chemistry, molecular oxohalides (oxyhalides) are a group of chemical compounds in which both oxygen and halogen atoms are attached to another chemical element A in a single molecule. They have the general formula AOmXn, where X is a halogen. Known oxohalides have fluorine (F), chlorine (Cl), bromine (Br), and/or iodine (I) in their molecules. The element A may be a main group element, a transition element, a rare earth element or an actinide. The term oxohalide, or oxyhalide, may also refer to minerals and other crystalline substances with the same overall chemical formula, but having an ionic structure.

Alpha-substitution reactions occur at the position next to the carbonyl group, the α-position, and involve the substitution of an α hydrogen atom by an electrophile, E, through either an enol or enolate ion intermediate.
Fluorine forms a great variety of chemical compounds, within which it always adopts an oxidation state of −1. With other atoms, fluorine forms either polar covalent bonds or ionic bonds. Most frequently, covalent bonds involving fluorine atoms are single bonds, although at least two examples of a higher order bond exist. Fluoride may act as a bridging ligand between two metals in some complex molecules. Molecules containing fluorine may also exhibit hydrogen bonding. Fluorine's chemistry includes inorganic compounds formed with hydrogen, metals, nonmetals, and even noble gases; as well as a diverse set of organic compounds. For many elements the highest known oxidation state can be achieved in a fluoride. For some elements this is achieved exclusively in a fluoride, for others exclusively in an oxide; and for still others the highest oxidation states of oxides and fluorides are always equal.