
Veterinary medicine is the branch of medicine that deals with the prevention, diagnosis and treatment of disease, disorder and injury in non-human animals. The scope of veterinary medicine is wide, covering all animal species, both domesticated and wild, with a wide range of conditions which can affect different species.

Estriol (E3), also spelled oestriol, is a steroid, a weak estrogen, and a minor female sex hormone. It is one of three major endogenous estrogens, the others being estradiol and estrone. Levels of estriol in women who are not pregnant are almost undetectable. However, during pregnancy, estriol is synthesized in very high quantities by the placenta and is the most produced estrogen in the body by far, although circulating levels of estriol are similar to those of other estrogens due to a relatively high rate of metabolism and excretion. Relative to estradiol, both estriol and estrone have far weaker activity as estrogens.

Gestrinone, sold under the brand names Dimetrose and Nemestran among others, is a medication which is used in the treatment of endometriosis. It has also been used to treat other conditions such as uterine fibroids and heavy menstrual bleeding and has been investigated as a method of birth control. Gestrinone is used alone and is not formulated in combination with other medications. It is taken by mouth or in through the vagina.
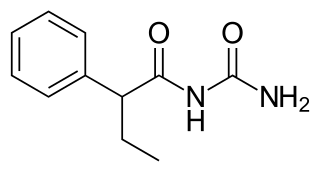
Pheneturide, also known as phenylethylacetylurea, is an anticonvulsant of the ureide class. Conceptually, it can be formed in the body as a metabolic degradation product from [[phenobarbital]]. It is considered to be obsolete, and is now seldom used. It is marketed in Europe, including in Poland, Spain, and the United Kingdom. Pheneturide has a similar profile of anticonvulsant activity and toxicity relative to phenacemide, but is less toxic in comparison, despite still being a toxic drug. As such, it is only used in cases of severe epilepsy when other, less-toxic drugs have failed. Pheneturide inhibits the metabolism and thus increases the levels of other anticonvulsants, such as phenytoin.

English is a West Germanic language that was first spoken in early medieval England and eventually became a global lingua franca. It is named after the Angles, one of the Germanic tribes that migrated to the area of Great Britain that later took their name, as England. Both names derive from Anglia, a peninsula in the Baltic Sea. The language is closely related to Frisian and Low Saxon, and its vocabulary has been significantly influenced by other Germanic languages, particularly Norse, and to a greater extent by Latin and French.
Complement 2 deficiency is a type of complement deficiency caused by any one of several different alterations in the structure of complement component 2.

Promestriene (INN), also known as estradiol 3-propyl 17β-methyl diether, is a synthetic steroidal estrogen which is used topically in a 1% cream formulation. It is the 3-propyl and 17β-methyl diether of estradiol. The drug is described as a tropic agent and antiseborrheic. It has not been found to be effective in the treatment of androgenic alopecia or other conditions of cutaneous androgenization.

Aglepristone (INN) is a synthetic, steroidal antiprogestogen related to mifepristone which is marketed by Virbac in several European countries for use in veterinary medicine. It is specifically used as an abortifacient in pregnant animals. Aglepristone, similarly to mifepristone, also possesses some antiglucocorticoid activity.
Complement 3 deficiency is a genetic condition affecting complement component 3.

The dorsal ulnocarpal ligament is a ligament in the hand. It is a fibrous band passing from the styloid process of the ulna to the dorsal aspect of the carpal bones.
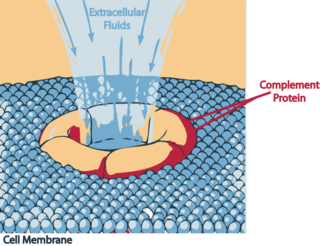
Terminal complement pathway deficiency is a genetic condition affecting the complement membrane attack complex (MAC).

Progynon was an orally active formulation of estrogen that was developed by Adolf Butenandt at Schering and introduced in Germany in 1928. It was reportedly the first sex hormone product and hence also the first estrogen product to be introduced for medical use. Progynon was originally an ovarian or placental extract, but Schering soon switched for economic reasons to using the urine of women who were in late pregnancy. This form of Progynon was essentially the same product as Emmenin, which was developed by James Collip at Ayerst and introduced in Canada in 1930.
Elsevier BIOBASE is a bibliographic database covering all topics pertaining to biological research throughout the world. It was established in the 1950s in print format as Current Awareness in Biological Sciences. Temporal coverage is from 1994 to the present. The database has over 4.1 million records as of December 2008. More than 300,000 records are added annually and 84% contain an abstract. It is updated weekly.

Raseglurant (INN) is a negative allosteric modulator of the mGlu5 receptor and derivative of MPEP which was under development by Addex Therapeutics for the treatment of migraine, gastroesophageal reflux disease, and dental anxiety. It reached phase II clinical trials for all of the aforementioned indications before being discontinued due to the observation of possible predictive signs of hepatotoxicity in patients with long-term use.
Sialodochitis, is inflammation of the duct system of a salivary gland. This is compared to sialadenitis, which is inflammation of the gland parenchyma.
A steroidogenesis inhibitor, also known as a steroid biosynthesis inhibitor, is a type of drug which inhibits one or more of the enzymes that are involved in the process of steroidogenesis, the biosynthesis of endogenous steroids and steroid hormones. They may inhibit the production of cholesterol and other sterols, sex steroids such as androgens, estrogens, and progestogens, corticosteroids such as glucocorticoids and mineralocorticoids, and neurosteroids. They are used in the treatment of a variety of medical conditions that depend on endogenous steroids.

Clogestone, also known as chlormadinol or as 3β,17α-dihydroxy-6-chloropregna-4,6-diene-20-one, is a steroidal progestin that was synthesized in 1964 and was investigated as a progestin-only contraceptive but was never marketed. A diacetate ester, clogestone acetate, also exists and similarly was never marketed.

17α-Epiestriol, or simply 17-epiestriol, also known as 16α-hydroxy-17α-estradiol or estra-1,3,5(10)-triene-3,16α,17α-triol, is a minor and weak endogenous estrogen, and the 17α-epimer of estriol. It is formed from 16α-hydroxyestrone. In contrast to other endogenous estrogens like estradiol, 17α-epiestriol is a selective agonist of the ERβ. It is described as a relatively weak estrogen, which is in accordance with its relatively low affinity for the ERα. 17α-Epiestriol has been found to be approximately 400-fold more potent than estradiol in inhibiting tumor necrosis factor α (TNFα)-induced vascular cell adhesion molecule 1 (VCAM-1) expression in vitro.

16α-Hydroxydehydroepiandrosterone sulfate (16α-OH-DHEA-S), also known as 16α-hydroxy-17-oxoandrost-5-en-3β-yl sulfate, is an endogenous, naturally occurring steroid and a metabolic intermediate in the production of estriol from dehydroepiandrosterone (DHEA) during pregnancy. It is the C3β sulfate ester of 16α-hydroxy-DHEA.

Perlapine, sold under the brand names Hypnodine and Pipnodine, is a hypnotic and sedative of the tricyclic group which is marketed in Japan. It acts primarily as a potent antihistamine, and also has anticholinergic, antiserotonergic, antiadrenergic, and some antidopaminergic activity. The drug has relatively weak affinity for the dopamine D2 receptor (IC50 = 1,803 nM) and, in accordance, is said to be ineffective as an antipsychotic. However, it retains higher affinity for the dopamine D1 receptor (IC50 = 198 nM). Its IC50 values are 19 nM for the α1-adrenergic receptor, 4,945 nM for the α2-adrenergic recpetor, and 70 nM for the serotonin 5-HT2A receptor. Perlapine is closely related to clotiapine, clozapine, fluperlapine, loxapine, and tilozepine.














