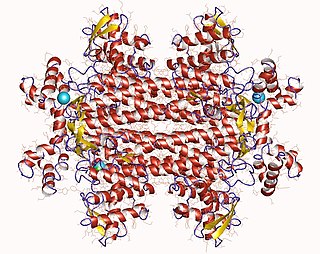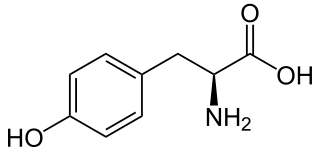
L-Tyrosine or tyrosine or 4-hydroxyphenylalanine is one of the 20 standard amino acids that are used by cells to synthesize proteins. It is a non-essential amino acid with a polar side group. The word "tyrosine" is from the Greek tyrós, meaning cheese, as it was first discovered in 1846 by German chemist Justus von Liebig in the protein casein from cheese. It is called tyrosyl when referred to as a functional group or side chain. While tyrosine is generally classified as a hydrophobic amino acid, it is more hydrophilic than phenylalanine. It is encoded by the codons UAC and UAU in messenger RNA.
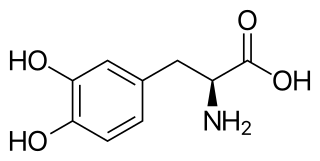
l-DOPA, also known as levodopa and l-3,4-dihydroxyphenylalanine, is an amino acid that is made and used as part of the normal biology of some plants and animals, including humans. Humans, as well as a portion of the other animals that utilize l-DOPA, make it via biosynthesis from the amino acid l-tyrosine. l-DOPA is the precursor to the neurotransmitters dopamine, norepinephrine (noradrenaline), and epinephrine (adrenaline), which are collectively known as catecholamines. Furthermore, l-DOPA itself mediates neurotrophic factor release by the brain and CNS. l-DOPA can be manufactured and in its pure form is sold as a psychoactive drug with the INN levodopa; trade names include Sinemet, Pharmacopa, Atamet, and Stalevo. As a drug, it is used in the clinical treatment of Parkinson's disease and dopamine-responsive dystonia.
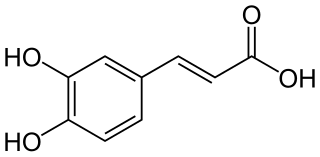
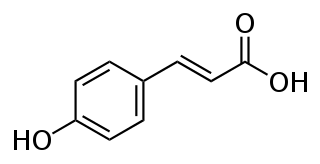
Caffeic acid is an organic compound that is classified as a hydroxycinnamic acid. This yellow solid consists of both phenolic and acrylic functional groups. It is found in all plants because it is an intermediate in the biosynthesis of lignin, one of the principal components of woody plant biomass and its residues.
3,4-dihydroxyphenylalanine oxidative deaminase (EC 1.13.12.15, 3,4-dihydroxy-L-phenylalanine: oxidative deaminase, oxidative deaminase, DOPA oxidative deaminase, DOPAODA) is an enzyme with systematic name 3,4-dihydroxy-L-phenylalanine:oxygen oxidoreductase (deaminating). This enzyme catalyses the following chemical reaction
The enzyme 3,4-dihydroxyphenylalanine reductive deaminase (EC 4.3.1.22, reductive deaminase, DOPA-reductive deaminase, DOPARDA; systematic name 3,4-dihydroxy-L-phenylalanine ammonia-lyase (3,4-dihydroxyphenylpropanoate-forming)) catalyses the following chemical reaction