
Parathion, also called parathion-ethyl or diethyl parathion and locally known as "Folidol", is an organophosphate insecticide and acaricide. It was originally developed by IG Farben in the 1940s. It is highly toxic to non-target organisms, including humans, so its use has been banned or restricted in most countries. The basic structure is shared by parathion methyl.
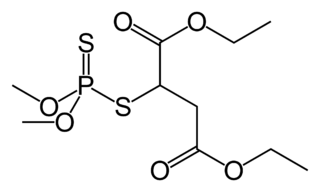
Malathion is an organophosphate insecticide which acts as an acetylcholinesterase inhibitor. In the USSR, it was known as carbophos, in New Zealand and Australia as maldison and in South Africa as mercaptothion.

A defoliant is any herbicidal chemical sprayed or dusted on plants to cause their leaves to fall off. Defoliants are widely used for the selective removal of weeds in managing croplands and lawns. Worldwide use of defoliants, along with the development of other herbicides and pesticides, allowed for the Green Revolution, an increase in agricultural production in mid-20th century. Defoliants have also been used in warfare as a means to deprive an enemy of food crops and/or hiding cover, most notably by the United Kingdom during the Malayan Emergency and the United States in the Vietnam War. Defoliants were also used by Indonesian forces in various internal security operations.
Benomyl is a fungicide introduced in 1968 by DuPont. It is a systemic benzimidazole fungicide that is selectively toxic to microorganisms and invertebrates, but relatively nontoxic toward mammals.

The oligodynamic effect is a biocidal effect of metals, especially heavy metals, that occurs even in low concentrations. This effect is attributed to the antibacterial behavior of metal ions, which are absorbed by bacteria upon contact and damage their cell membranes.

Captan is a general use pesticide (GUP) that belongs to the phthalimide class of fungicides. It is a white solid, although commercial samples appear yellow or brownish.

Chlorothalonil (2,4,5,6-tetrachloroisophthalonitrile) is an organic compound mainly used as a broad spectrum, nonsystemic fungicide, with other uses as a wood protectant, pesticide, acaricide, and to control mold, mildew, bacteria, algae. Chlorothalonil-containing products are sold under the names Bravo, Echo, and Daconil. It was first registered for use in the US in 1966. In 1997, the most recent year for which data are available, it was the third most used fungicide in the US, behind only sulfur and copper, with 12 million pounds used in agriculture that year. Including nonagricultural uses, the United States Environmental Protection Agency (EPA) estimates, on average, almost 15 million lb (6.8 million kg) were used annually from 1990 to 1996.

Calcium arsenate is the inorganic compound with the formula Ca3(AsO4)2. A colourless salt, it was originally used as a pesticide and as a germicide. It is highly soluble in water, in contrast to lead arsenate, which makes it more toxic. Two minerals are hydrates of calcium arsenate: rauenthalite Ca3(AsO4)2·10H2O and phaunouxite Ca3(AsO4)2·11H2O. A related mineral is ferrarisite (Ca5H2(AsO4)4·9H2O.

1,3-Dichloropropene, sold under diverse trade names, is an organochlorine compound with the formula C3H4Cl2. It is a colorless liquid with a sweet smell. It is feebly soluble in water and evaporates easily. It is used mainly in farming as a pesticide, specifically as a preplant fumigant and nematicide. It is widely used in the US and other countries, but is banned in 34 countries.

Azoxystrobin is a broad spectrum systemic fungicide widely used in agriculture to protect crops from fungal diseases. It was first marketed in 1996 using the brand name Amistar and by 1999 it had been registered in 48 countries on more than 50 crops. In the year 2000 it was announced that it had been granted UK Millennium product status.

Pentachloronitrobenzene, typically abbreviated PCNB, is a registered fungicide formally derived from nitrobenzene. It is either an off-white or yellow solid, depending on its purity, with a musty odor.
The British Crop Production Council (BCPC) is an organisation that promotes the use of good science and technology in the understanding and application of effective and sustainable crop production. BCPC is a Registered Charity and a Company limited by Guarantee.
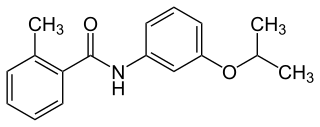
Mepronil is a fungicide used as a seed treatment or foliar spray in agriculture to protect crops from fungal diseases. It was first marketed by Kumiai Chemical Industries in 1981 using their brand name Basitac. The compound is a benzanilide which combines 2-methylbenzoic acid with the O-isopropyl derivative of 3-aminophenol to give an inhibitor of succinate dehydrogenase (SDHI).
Mancozeb is a dithiocarbamate non-systemic agricultural fungicide with multi-site, protective action on contact. It is a combination of two other dithiocarbamates: maneb and zineb. The mixture controls many fungal diseases in a wide range of field crops, fruits, nuts, vegetables, and ornamentals. It is marketed as Penncozeb, Trimanoc, Vondozeb, Dithane, Manzeb, Nemispot, and Manzane. In Canada, a mixture of zoxamide and mancozeb was registered for control of the mildew named Gavel as early as 2008.

Cyproconazole is an agricultural fungicide of the class of azoles, used on cereal crops, coffee, sugar beet, fruit trees and grapes, and peanuts, on sod farms and golf course turf and on wood as a preservative. It has been used against powdery mildew, rust on cereals and apple scab, and applied by air or on the ground or by chemigation.

Oxycarboxin is an organic chemical used in agriculture to protect crops from fungal diseases. It was first marketed by Uniroyal in 1969 using their brand name Plantvax. The compound is an anilide which combines a heterocyclic acid with aniline to give an inhibitor of succinate dehydrogenase (SDHI).
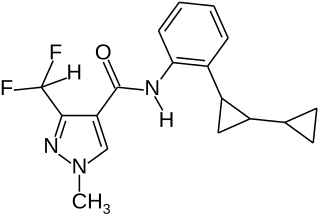
Sedaxane is a broad spectrum fungicide used as a seed treatment in agriculture to protect crops from fungal diseases. It was first marketed by Syngenta in 2011 using their brand name Vibrance. The compound is an amide which combines a pyrazole acid with an aryl amine to give an inhibitor of succinate dehydrogenase.
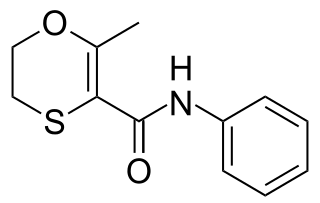
Carboxin is a narrow-spectrum fungicide used as a seed treatment in agriculture to protect crops from fungal diseases. It was first marketed by Uniroyal in 1969 using their brand name Vitavax. The compound is an anilide which combines a heterocyclic acid with aniline to give an inhibitor of succinate dehydrogenase (SDHI).

Prothioconazole is a synthetic chemical produced primarily for its fungicidal properties. It is a member of the class of compounds triazoles, and possesses a unique toxophore in this class of fungicides. Its effective fungicidal properties can be attributed to its ability to inhibit CYP51A1. This enzyme is required to biosynthesize ergosterol, a key component in the cell membrane of fungi.


















