Brassicaceae or Cruciferae is a medium-sized and economically important family of flowering plants commonly known as the mustards, the crucifers, or the cabbage family. Most are herbaceous plants, while some are shrubs. The leaves are simple, lack stipules, and appear alternately on stems or in rosettes. The inflorescences are terminal and lack bracts. The flowers have four free sepals, four free alternating petals, two shorter free stamens and four longer free stamens. The fruit has seeds in rows, divided by a thin wall.

In organic chemistry, isothiocyanate is the functional group −N=C=S, formed by substituting the oxygen in the isocyanate group with a sulfur. Many natural isothiocyanates from plants are produced by enzymatic conversion of metabolites called glucosinolates. These natural isothiocyanates, such as allyl isothiocyanate, are also known as mustard oils. An artificial isothiocyanate, phenyl isothiocyanate, is used for amino acid sequencing in the Edman degradation.
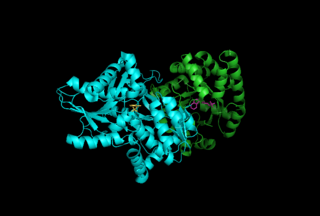
Tryptophan synthase or tryptophan synthetase is an enzyme that catalyses the final two steps in the biosynthesis of tryptophan. It is commonly found in Eubacteria, Archaebacteria, Protista, Fungi, and Plantae. However, it is absent from Animalia. It is typically found as an α2β2 tetramer. The α subunits catalyze the reversible formation of indole and glyceraldehyde-3-phosphate (G3P) from indole-3-glycerol phosphate (IGP). The β subunits catalyze the irreversible condensation of indole and serine to form tryptophan in a pyridoxal phosphate (PLP) dependent reaction. Each α active site is connected to a β active site by a 25 angstrom long hydrophobic channel contained within the enzyme. This facilitates the diffusion of indole formed at α active sites directly to β active sites in a process known as substrate channeling. The active sites of tryptophan synthase are allosterically coupled.

Allyl isothiocyanate (AITC) is a naturally occurring unsaturated isothiocyanate. The colorless oil is responsible for the pungent taste of Cruciferous vegetables such as mustard, radish, horseradish, and wasabi. This pungency and the lachrymatory effect of AITC are mediated through the TRPA1 and TRPV1 ion channels. It is slightly soluble in water, but more soluble in most organic solvents.

Indole-3-acetic acid is the most common naturally occurring plant hormone of the auxin class. It is the best known of the auxins, and has been the subject of extensive studies by plant physiologists. IAA is a derivative of indole, containing a carboxymethyl substituent. It is a colorless solid that is soluble in polar organic solvents.

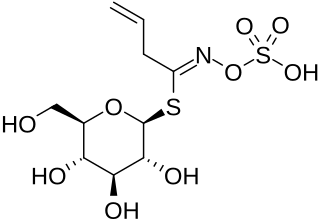
Sulforaphane is a compound within the isothiocyanate group of organosulfur compounds. It is produced when the enzyme myrosinase transforms glucoraphanin, a glucosinolate, into sulforaphane upon damage to the plant, which allows the two compounds to mix and react.

Glucoraphanin is a glucosinolate found in broccoli, mustard and other cruciferous vegetables.

Glucosinolates are natural components of many pungent plants such as mustard, cabbage, and horseradish. The pungency of those plants is due to mustard oils produced from glucosinolates when the plant material is chewed, cut, or otherwise damaged. These natural chemicals most likely contribute to plant defence against pests and diseases, and impart a characteristic bitter flavor property to cruciferous vegetables.
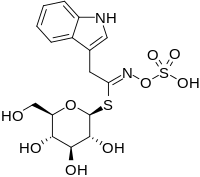
Sinigrin or allyl glucosinolate is a glucosinolate that belongs to the family of glucosides found in some plants of the family Brassicaceae such as Brussels sprouts, broccoli, and the seeds of black mustard. Whenever sinigrin-containing plant tissue is crushed or otherwise damaged, the enzyme myrosinase degrades sinigrin to a mustard oil, which is responsible for the pungent taste of mustard and horseradish. Seeds of white mustard, Sinapis alba, give a less pungent mustard because this species contains a different glucosinolate, sinalbin.

Myrosinase is a family of enzymes involved in plant defense against herbivores, specifically the mustard oil bomb. The three-dimensional structure has been elucidated and is available in the PDB.

Erysimum cheiranthoides, the treacle-mustard,wormseed wallflower, or wormseed mustard is a species of Erysimum native to most of central and northern Europe and northern and central Asia. Like other Erysimum species, E. cheiranthoides accumulates two major classes of defensive chemicals: glucosinolates and cardiac glycosides.
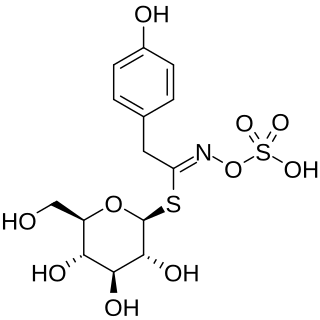
Sinalbin is a glucosinolate found in the seeds of white mustard, Sinapis alba, and in many wild plant species. In contrast to mustard from black mustard seeds which contain sinigrin, mustard from white mustard seeds has only a weakly pungent taste.

Gluconasturtiin or phenethyl glucosinolate is one of the most widely distributed glucosinolates in the cruciferous vegetables, mainly in the roots, and is probably one of the plant compounds responsible for the natural pest-inhibiting properties of growing crucifers, such as cabbage, mustard or rape, in rotation with other crops. This effect of gluconasturtiin is due to its degradation by the plant enzyme myrosinase into phenethyl isothiocyanate, which is toxic to many organisms.
The enzyme indole-3-glycerol-phosphate synthase (IGPS) (EC 4.1.1.48) catalyzes the chemical reaction

Brevicoryne brassicae, commonly known as the cabbage aphid or cabbage aphis, is a destructive aphid native to Europe that is now found in many other areas of the world. The aphids feed on many varieties of produce, including cabbage, broccoli (especially), Brussels sprouts, cauliflower and many other members of the genus Brassica, but do not feed on plants outside of the family Brassicaceae. The insects entirely avoid plants other than those of Brassicaceae; even though thousands may be eating broccoli near strawberries, the strawberries will be left untouched.
Tryptophan N-monooxygenase (EC 1.14.13.125, tryptophan N-hydroxylase, CYP79B1, CYP79B2, CYP79B3) is an enzyme with systematic name L-tryptophan,NADPH:oxygen oxidoreductase (N-hydroxylating). This enzyme catalyses the following chemical reaction
L-tryptophan—pyruvate aminotransferase is an enzyme with systematic name L-tryptophan:pyruvate aminotransferase. This enzyme catalyses the following chemical reaction

Camalexin (3-thiazol-2-yl-indole) is a simple indole alkaloid found in the plant Arabidopsis thaliana and other crucifers. The secondary metabolite functions as a phytoalexin to deter bacterial and fungal pathogens.

Glucotropaeolin or benzyl glucosinolate is a glucosinolate found in cruciferous vegetables, particularly garden cress. Upon enzymatic activity, it is transformed into benzyl isothiocyanate, which contributes to the characteristic flavor of these brassicas.

Georg Jander is an American plant biologist at the Boyce Thompson Institute in Ithaca, New York. He has an adjunct appointment in the Plant Biology Section of the School of Integrative Plant Sciences at Cornell University. Jander is known for his molecular research identifying genes for biochemical compounds of ecological and agricultural importance, particularly those plant traits involved in resistance to insect pests.