Related Research Articles

The hypothalamus is a small part of the brain that contains a number of nuclei with a variety of functions. One of the most important functions is to link the nervous system to the endocrine system via the pituitary gland. The hypothalamus is located below the thalamus and is part of the limbic system. It forms the ventral part of the diencephalon. All vertebrate brains contain a hypothalamus. In humans, it is the size of an almond.
Luteinizing hormone is a hormone produced by gonadotropic cells in the anterior pituitary gland. The production of LH is regulated by gonadotropin-releasing hormone (GnRH) from the hypothalamus. In females, an acute rise of LH known as an LH surge, triggers ovulation and development of the corpus luteum. In males, where LH had also been called interstitial cell–stimulating hormone (ICSH), it stimulates Leydig cell production of testosterone. It acts synergistically with follicle-stimulating hormone (FSH).

The supraoptic nucleus (SON) is a nucleus of magnocellular neurosecretory cells in the hypothalamus of the mammalian brain. The nucleus is situated at the base of the brain, adjacent to the optic chiasm. In humans, the SON contains about 3,000 neurons.
Gonadotropin-releasing hormone (GnRH) is a releasing hormone responsible for the release of follicle-stimulating hormone (FSH) and luteinizing hormone (LH) from the anterior pituitary. GnRH is a tropic peptide hormone synthesized and released from GnRH neurons within the hypothalamus. The peptide belongs to gonadotropin-releasing hormone family. It constitutes the initial step in the hypothalamic–pituitary–gonadal axis.
Gonadotropins are glycoprotein hormones secreted by gonadotropic cells of the anterior pituitary of vertebrates. This family includes the mammalian hormones follicle-stimulating hormone (FSH) and luteinizing hormone (LH), the placental/chorionic gonadotropins, human chorionic gonadotropin (hCG) and equine chorionic gonadotropin (eCG), as well as at least two forms of fish gonadotropins. These hormones are central to the complex endocrine system that regulates normal growth, sexual development, and reproductive function. LH and FSH are secreted by the anterior pituitary gland, while hCG and eCG are secreted by the placenta in pregnant humans and mares, respectively. The gonadotropins act on the gonads, controlling gamete and sex hormone production.
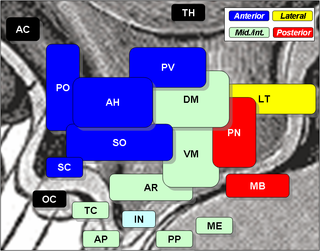
The arcuate nucleus of the hypothalamus is an aggregation of neurons in the mediobasal hypothalamus, adjacent to the third ventricle and the median eminence. The arcuate nucleus includes several important and diverse populations of neurons that help mediate different neuroendocrine and physiological functions, including neuroendocrine neurons, centrally projecting neurons, and astrocytes. The populations of neurons found in the arcuate nucleus are based on the hormones they secrete or interact with and are responsible for hypothalamic function, such as regulating hormones released from the pituitary gland or secreting their own hormones. Neurons in this region are also responsible for integrating information and providing inputs to other nuclei in the hypothalamus or inputs to areas outside this region of the brain. These neurons, generated from the ventral part of the periventricular epithelium during embryonic development, locate dorsally in the hypothalamus, becoming part of the ventromedial hypothalamic region. The function of the arcuate nucleus relies on its diversity of neurons, but its central role is involved in homeostasis. The arcuate nucleus provides many physiological roles involved in feeding, metabolism, fertility, and cardiovascular regulation.
Releasing hormones and inhibiting hormones are hormones whose main purpose is to control the release of other hormones, either by stimulating or inhibiting their release. They are also called liberins and statins (respectively), or releasing factors and inhibiting factors. The principal examples are hypothalamic-pituitary hormones that can be classified from several viewpoints: they are hypothalamic hormones, they are hypophysiotropic hormones, and they are tropic hormones.
Neuroendocrinology is the branch of biology which studies the interaction between the nervous system and the endocrine system; i.e. how the brain regulates the hormonal activity in the body. The nervous and endocrine systems often act together in a process called neuroendocrine integration, to regulate the physiological processes of the human body. Neuroendocrinology arose from the recognition that the brain, especially the hypothalamus, controls secretion of pituitary gland hormones, and has subsequently expanded to investigate numerous interconnections of the endocrine and nervous systems.

The hypothalamic–pituitary–gonadal axis refers to the hypothalamus, pituitary gland, and gonadal glands as if these individual endocrine glands were a single entity. Because these glands often act in concert, physiologists and endocrinologists find it convenient and descriptive to speak of them as a single system.
Growth hormone–releasing hormone (GHRH), also known as somatocrinin or by several other names in its endogenous forms and as somatorelin (INN) in its pharmaceutical form, is a releasing hormone of growth hormone (GH). It is a 44-amino acid peptide hormone produced in the arcuate nucleus of the hypothalamus.
The periventricular nucleus is a thin sheet of small neurons located in the wall of the third ventricle, a composite structure of the hypothalamus. It functions in analgesia.

Kisspeptins are proteins encoded by the KISS1 gene in humans. Kisspeptins are ligands of the G-protein coupled receptor, GPR54. Kiss1 was originally identified as a human metastasis suppressor gene that has the ability to suppress melanoma and breast cancer metastasis. Kisspeptin-GPR54 signaling has an important role in initiating secretion of gonadotropin-releasing hormone (GnRH) at puberty, the extent of which is an area of ongoing research. Gonadotropin-releasing hormone is released from the hypothalamus to act on the anterior pituitary triggering the release of luteinizing hormone (LH), and follicle stimulating hormone (FSH). These gonadotropic hormones lead to sexual maturation and gametogenesis. Disrupting GPR54 signaling can cause hypogonadotrophic hypogonadism in rodents and humans. The Kiss1 gene is located on chromosome 1. It is transcribed in the brain, adrenal gland, and pancreas.

Neurokinin B (NKB) belongs in the family of tachykinin peptides. Neurokinin B is implicated in a variety of human functions and pathways such as the secretion of gonadotropin-releasing hormone. Additionally, NKB is associated with pregnancy in females and maturation in young adults. Reproductive function is highly dependent on levels of both neurokinin B and also the G-protein coupled receptor ligand kisspeptin. The first NKB studies done attempted to resolve why high levels of the peptide may be implicated in pre-eclampsia during pregnancy. NKB, kisspeptin, and dynorphin together are found in the arcuate nucleus (ARC) known as the KNDy subpopulation. This subpopulation is targeted by many steroid hormones and works to form a network that feeds back to GnRH pulse generator.

The KiSS1-derived peptide receptor is a G protein-coupled receptor which binds the peptide hormone kisspeptin (metastin). Kisspeptin is encoded by the metastasis suppressor gene KISS1, which is expressed in a variety of endocrine and gonadal tissues. Activation of the kisspeptin receptor is linked to the phospholipase C and inositol trisphosphate second messenger cascades inside the cell.
Functional hypothalamic amenorrhea (FHA) is a form of amenorrhea and chronic anovulation and is one of the most common types of secondary amenorrhea. It is classified as hypogonadotropic hypogonadism. It was previously known as "juvenile hypothalamosis syndrome," prior to the discovery that sexually mature females are equally affected. FHA has multiple risk factors, with links to stress-related, weight-related, and exercise-related factors. FHA is caused by stress-induced suppression of the hypothalamic-pituitary-ovarian (HPO) axis, which results in inhibition of gonadotropin-releasing hormone (GnRH) secretion, and gonadotropins, follicle-stimulating hormone (FSH) and luteinizing hormone (LH). Severe and potentially prolonged hypoestrogenism is perhaps the most dangerous hormonal pathology associated with the disease, because consequences of this disturbance can influence bone health, cardiovascular health, mental health, and metabolic functioning in both the short and long-term. Because many of the symptoms overlap with those of organic hypothalamic, pituitary, or gonadal disease and therefore must be ruled out, FHA is a diagnosis of exclusion; "functional" is used to indicate a behavioral cause, in which no anatomical or organic disease is identified, and is reversible with correction of the underlying cause. Diagnostic workup includes a detailed history and physical, laboratory studies, such as a pregnancy test, and serum levels of FSH and LH, prolactin, and thyroid-stimulating hormone (TSH), and imaging. Additional tests may be indicated in order to distinguish FHA from organic hypothalamic or pituitary disorders. Patients present with a broad range of symptoms related to severe hypoestrogenism as well as hypercortisolemia, low serum insulin levels, low serum insulin-like growth factor 1 (IGF-1), and low total triiodothyronine (T3). Treatment is primarily managing the primary cause of the FHA with behavioral modifications. While hormonal-based therapies are potential treatment to restore menses, weight gain and behavioral modifications can have an even more potent impact on reversing neuroendocrine abnormalities, preventing further bone loss, and re-establishing menses, making this the recommended line of treatment. If this fails to work, secondary treatment is aimed at treating the effects of hypoestrogenism, hypercortisolism, and hypothyroidism.
Hypogonadotropic hypogonadism (HH), is due to problems with either the hypothalamus or pituitary gland affecting the hypothalamic-pituitary-gonadal axis. Hypothalamic disorders result from a deficiency in the release of gonadotropic releasing hormone (GnRH), while pituitary gland disorders are due to a deficiency in the release of gonadotropins from the anterior pituitary. GnRH is the central regulator in reproductive function and sexual development via the HPG axis. GnRH is released by GnRH neurons, which are hypothalamic neuroendocrine cells, into the hypophyseal portal system acting on gonadotrophs in the anterior pituitary. The release of gonadotropins, LH and FSH, act on the gonads for the development and maintenance of proper adult reproductive physiology. LH acts on Leydig cells in the male testes and theca cells in the female. FSH acts on Sertoli cells in the male and follicular cells in the female. Combined this causes the secretion of gonadal sex steroids and the initiation of folliculogenesis and spermatogenesis. The production of sex steroids forms a negative feedback loop acting on both the anterior pituitary and hypothalamus causing a pulsatile secretion of GnRH. GnRH neurons lack sex steroid receptors and mediators such as kisspeptin stimulate GnRH neurons for pulsatile secretion of GnRH.
The Organizational-Activational Hypothesis states that steroid hormones permanently organize the nervous system during early development, which is reflected in adult male or female typical behaviors. In adulthood, the same steroid hormones activate, modulate, and inhibit these behaviors. This idea was revolutionary when first published in 1959 because no other previous experiment had demonstrated that adult behaviors could be determined hormonally during early development.
Pulsatile secretion is a biochemical phenomenon observed in a wide variety of cell and tissue types, in which chemical products are secreted in a regular temporal pattern. The most common cellular products observed to be released in this manner are intercellular signaling molecules such as hormones or neurotransmitters. Examples of hormones that are secreted pulsatilely include insulin, thyrotropin, TRH, gonadotropin-releasing hormone (GnRH) and growth hormone (GH). In the nervous system, pulsatility is observed in oscillatory activity from central pattern generators. In the heart, pacemakers are able to work and secrete in a pulsatile manner. A pulsatile secretion pattern is critical to the function of many hormones in order to maintain the delicate homeostatic balance necessary for essential life processes, such as development and reproduction. Variations of the concentration in a certain frequency can be critical to hormone function, as evidenced by the case of GnRH agonists, which cause functional inhibition of the receptor for GnRH due to profound downregulation in response to constant (tonic) stimulation. Pulsatility may function to sensitize target tissues to the hormone of interest and upregulate receptors, leading to improved responses. This heightened response may have served to improve the animal's fitness in its environment and promote its evolutionary retention.
Gonadotropin surge-attenuating factor (GnSAF) is a nonsteroidal ovarian hormone produced by the granulosa cells of small antral ovarian follicles in females. GnSAF is involved in regulating the secretion of luteinizing hormone (LH) from the anterior pituitary and the ovarian cycle. During the early to mid-follicular phase of the ovarian cycle, GnSAF acts on the anterior pituitary to attenuate LH release, limiting the secretion of LH to only basal levels. At the transition between follicular and luteal phase, GnSAF bioactivity declines sufficiently to permit LH secretion above basal levels, resulting in the mid-cycle LH surge that initiates ovulation. In normally ovulating women, the LH surge only occurs when the oocyte is mature and ready for extrusion. GnSAF bioactivity is responsible for the synchronised, biphasic nature of LH secretion.
Gonadotropin-inhibitory hormone (GnIH) is a RFamide-related peptide coded by the NPVF gene in mammals.
References
- 1 2 3 4 5 6 7 8 9 10 11 Moore, Aleisha M; Coolen, Lique M; Porter, Danielle T; Goodman, Robert L; Lehman, Michael N (2018-07-11). "KNDy Cells Revisited". Endocrinology. 159 (9): 3219–3234. doi:10.1210/en.2018-00389. ISSN 1945-7170. PMC 6098225 . PMID 30010844.
- 1 2 Hu, Guangfu; Lin, Chengyuan; He, Mulan; Wong, Anderson O.L. (2014-11-01). "Neurokinin B and reproductive functions: "KNDy neuron" model in mammals and the emerging story in fish". General and Comparative Endocrinology. 208: 94–108. doi:10.1016/j.ygcen.2014.08.009. hdl: 10722/204823 . ISSN 0016-6480. PMID 25172151.
- 1 2 3 Hrabovszky, Erik (2014). "Neuroanatomy of the Human Hypothalamic Kisspeptin System". Neuroendocrinology. 99 (1): 33–48. doi: 10.1159/000356903 . ISSN 1423-0194. PMID 24401651.
- 1 2 3 Tng, Eng Loon (December 2015). "Kisspeptin signalling and its roles in humans". Singapore Medical Journal. 56 (12): 649–656. doi:10.11622/smedj.2015183. ISSN 0037-5675. PMC 4678402 . PMID 26702158.