
Interferon regulatory factors (IRF) are proteins which regulate transcription of interferons. Interferon regulatory factors contain a conserved N-terminal region of about 120 amino acids, which folds into a structure that binds specifically to the IRF-element (IRF-E) motifs, which is located upstream of the interferon genes. Some viruses have evolved defense mechanisms that regulate and interfere with IRF functions to escape the host immune system. For instance, the remaining parts of the interferon regulatory factor sequence vary depending on the precise function of the protein. The Kaposi sarcoma herpesvirus, KSHV, is a cancer virus that encodes four different IRF-like genes; including vIRF1, which is a transforming oncoprotein that inhibits type 1 interferon activity. In addition, the expression of IRF genes is under epigenetic regulation by promoter DNA methylation.

Jan T. Vilček is a biomedical scientist, educator, inventor and philanthropist. He is a professor in the department of microbiology at the New York University School of Medicine, and chairman and CEO of The Vilcek Foundation. Vilček, a native of Bratislava, Slovakia, received his M.D. degree from Comenius University Medical School, Bratislava in 1957; and his Ph.D. in Virology from the Institute of Virology, Czechoslovak Academy of Sciences, Bratislava, Czechoslovakia in 1962. In 1964, Vilček, with his wife Marica, defected from Communist Czechoslovakia during a three-day visit to Vienna. In 1965, the Vilčeks immigrated to the United States, and have since lived in New York City. Vilček devoted his scientific career to studies of soluble mediators that regulate the immune system (cytokines), including interferon and tumor necrosis factor (TNF).

Interferon regulatory factor 3, also known as IRF3, is an interferon regulatory factor.

Interferon regulatory factor 2 is a protein that in humans is encoded by the IRF2 gene.

Interferon regulatory factor 7, also known as IRF7, is a member of the interferon regulatory factor family of transcription factors.
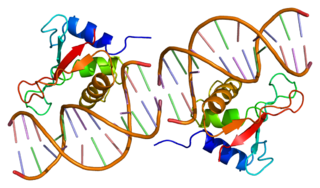
Interferon regulatory factor 1 is a protein that in humans is encoded by the IRF1 gene.

Interferon alpha-1 is a protein that in humans is encoded by the IFNA1 gene.

TBK1 is an enzyme with kinase activity. Specifically, it is a serine / threonine protein kinase. It is encoded by the TBK1 gene in humans. This kinase is mainly known for its role in innate immunity antiviral response. However, TBK1 also regulates cell proliferation, apoptosis, autophagy, and anti-tumor immunity. Insufficient regulation of TBK1 activity leads to autoimmune, neurodegenerative diseases or tumorigenesis.

Interferon regulatory factor 4 (IRF4) also known as MUM1 is a protein that in humans is encoded by the IRF4 gene,. IRF4 functions as a key regulatory transcription factor in the development of human immune cells. The expression of IRF4 is essential for the differentiation of T lymphocytes and B lymphocytes as well as certain myeloid cells. Dysregulation of the IRF4 gene can result in IRF4 functioning either as an oncogene or a tumor-suppressor, depending on the context of the modification.

Interferon regulatory factor 5 is a protein that in humans is encoded by the IRF5 gene. The IRF family is a group of transcription factors that are involved in signaling for virus responses in mammals along with regulation of certain cellular functions.

Interferon regulatory factor 8 (IRF8) also known as interferon consensus sequence-binding protein (ICSBP), is a protein that in humans is encoded by the IRF8 gene. IRF8 is a transcription factor that plays critical roles in the regulation of lineage commitment and in myeloid cell maturation including the decision for a common myeloid progenitor (CMP) to differentiate into a monocyte precursor cell.

Mitochondrial antiviral-signaling protein (MAVS) is a protein that is essential for antiviral innate immunity. MAVS is located in the outer membrane of the mitochondria, peroxisomes, and mitochondrial-associated endoplasmic reticulum membrane (MAM). Upon viral infection, a group of cytosolic proteins will detect the presence of the virus and bind to MAVS, thereby activating MAVS. The activation of MAVS leads the virally infected cell to secrete cytokines. This induces an immune response which kills the host's virally infected cells, resulting in clearance of the virus.

Stimulator of interferon genes (STING), also known as transmembrane protein 173 (TMEM173) and MPYS/MITA/ERIS is a protein that in humans is encoded by the STING1 gene.

Tadatsugu Taniguchi is a Japanese immunologist known for his pioneer research on Interferons and Interferon regulatory factors.
Immunology is the study of the immune system during health and disease. Below is a list of immunology-related articles.

Kathryn C. Zoon is a U.S.-based immunologist, elected to the U.S. Institute of Medicine in 2002 for her research on human interferons. She is the former scientific director of the Division of Intramural Research at the National Institute of Allergy and Infectious Diseases (NIAID), National Institutes of Health (NIH) in Bethesda, Maryland. From 1992 to 2002, Zoon was director of the FDA Center for Biologics Evaluation and Research (CBER).

Akiko Iwasaki is a Sterling Professor of Immunobiology and Molecular, Cellular and Developmental Biology at Yale University. She is also a principal investigator at the Howard Hughes Medical Institute. Her research interests include innate immunity, autophagy, inflammasomes, sexually transmitted infections, herpes simplex virus, human papillomavirus, respiratory virus infections, influenza infection, T cell immunity, commensal bacteria, COVID-19 and Long COVID.
The interleukin-1 receptor (IL-1R) associated kinase (IRAK) family plays a crucial role in the protective response to pathogens introduced into the human body by inducing acute inflammation followed by additional adaptive immune responses. IRAKs are essential components of the Interleukin-1 receptor signaling pathway and some Toll-like receptor signaling pathways. Toll-like receptors (TLRs) detect microorganisms by recognizing specific pathogen-associated molecular patterns (PAMPs) and IL-1R family members respond the interleukin-1 (IL-1) family cytokines. These receptors initiate an intracellular signaling cascade through adaptor proteins, primarily, MyD88. This is followed by the activation of IRAKs. TLRs and IL-1R members have a highly conserved amino acid sequence in their cytoplasmic domain called the Toll/Interleukin-1 (TIR) domain. The elicitation of different TLRs/IL-1Rs results in similar signaling cascades due to their homologous TIR motif leading to the activation of mitogen-activated protein kinases (MAPKs) and the IκB kinase (IKK) complex, which initiates a nuclear factor-κB (NF-κB) and AP-1-dependent transcriptional response of pro-inflammatory genes. Understanding the key players and their roles in the TLR/IL-1R pathway is important because the presence of mutations causing the abnormal regulation of Toll/IL-1R signaling leading to a variety of acute inflammatory and autoimmune diseases.
Carol Shoshkes Reiss, an American viral immunologist, has been professor in New York University's department of biology since 1991. Her research focused on the dynamic contest between the mouse immune system and virus replication during infection of the central nervous system. Reiss was editor-in-chief of the journal Viral Immunology (2000–2006) and is currently editor-in-chief of the journal DNA and Cell Biology (2012–present).
Karen Louise Mossman is a Canadian virologist who is a professor of Pathology and Molecular Medicine at McMaster University. Mossman looks to understand how viruses get around the defence mechanisms of cells. She was part of a team of Canadian researchers who first isolated SARS-CoV-2.
















