Related Research Articles

An axon, or nerve fiber, is a long, slender projection of a nerve cell, or neuron, in vertebrates, that typically conducts electrical impulses known as action potentials away from the nerve cell body. The function of the axon is to transmit information to different neurons, muscles, and glands. In certain sensory neurons, such as those for touch and warmth, the axons are called afferent nerve fibers and the electrical impulse travels along these from the periphery to the cell body and from the cell body to the spinal cord along another branch of the same axon. Axon dysfunction has caused many inherited and acquired neurological disorders which can affect both the peripheral and central neurons. Nerve fibers are classed into three types – group A nerve fibers, group B nerve fibers, and group C nerve fibers. Groups A and B are myelinated, and group C are unmyelinated. These groups include both sensory fibers and motor fibers. Another classification groups only the sensory fibers as Type I, Type II, Type III, and Type IV.
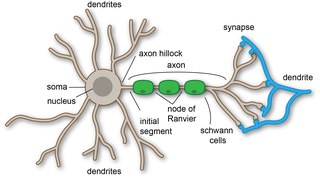
Dendrites, also dendrons, are branched protoplasmic extensions of a nerve cell that propagate the electrochemical stimulation received from other neural cells to the cell body, or soma, of the neuron from which the dendrites project. Electrical stimulation is transmitted onto dendrites by upstream neurons via synapses which are located at various points throughout the dendritic tree.

Exocytosis is a form of active transport and bulk transport in which a cell transports molecules out of the cell. As an active transport mechanism, exocytosis requires the use of energy to transport material. Exocytosis and its counterpart, endocytosis, are used by all cells because most chemical substances important to them are large polar molecules that cannot pass through the hydrophobic portion of the cell membrane by passive means. Exocytosis is the process by which a large amount of molecules are released; thus it is a form of bulk transport. Exocytosis occurs via secretory portals at the cell plasma membrane called porosomes. Porosomes are permanent cup-shaped lipoprotein structure at the cell plasma membrane, where secretory vesicles transiently dock and fuse to release intra-vesicular contents from the cell.
An inhibitory postsynaptic potential (IPSP) is a kind of synaptic potential that makes a postsynaptic neuron less likely to generate an action potential. IPSP were first investigated in motorneurons by David P. C. Lloyd, John Eccles and Rodolfo Llinás in the 1950s and 1960s. The opposite of an inhibitory postsynaptic potential is an excitatory postsynaptic potential (EPSP), which is a synaptic potential that makes a postsynaptic neuron more likely to generate an action potential. IPSPs can take place at all chemical synapses, which use the secretion of neurotransmitters to create cell to cell signalling. Inhibitory presynaptic neurons release neurotransmitters that then bind to the postsynaptic receptors; this induces a change in the permeability of the postsynaptic neuronal membrane to particular ions. An electric current that changes the postsynaptic membrane potential to create a more negative postsynaptic potential is generated, i.e. the postsynaptic membrane potential becomes more negative than the resting membrane potential, and this is called hyperpolarisation. To generate an action potential, the postsynaptic membrane must depolarize—the membrane potential must reach a voltage threshold more positive than the resting membrane potential. Therefore, hyperpolarisation of the postsynaptic membrane makes it less likely for depolarisation to sufficiently occur to generate an action potential in the postsynaptic neurone.
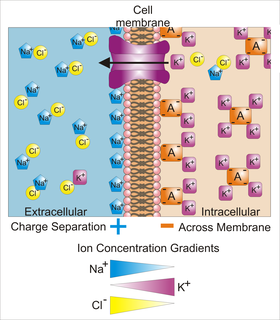
Membrane potential is the difference in electric potential between the interior and the exterior of a biological cell. That is, there is a difference in the energy required for electric charges to move from the internal to exterior cellular environments and vice versa, as long as there is no acquisition of kinetic energy or the production of radiation. The concentration gradients of the charges directly determine this energy requirement. For the exterior of the cell, typical values of membrane potential, normally given in units of millivolts and denoted as mV, range from –80 mV to –40 mV.

An excitatory synapse is a synapse in which an action potential in a presynaptic neuron increases the probability of an action potential occurring in a postsynaptic cell. Neurons form networks through which nerve impulses travel, each neuron often making numerous connections with other cells. These electrical signals may be excitatory or inhibitory, and, if the total of excitatory influences exceeds that of the inhibitory influences, the neuron will generate a new action potential at its axon hillock, thus transmitting the information to yet another cell.
Neural engineering is a discipline within biomedical engineering that uses engineering techniques to understand, repair, replace, or enhance neural systems. Neural engineers are uniquely qualified to solve design problems at the interface of living neural tissue and non-living constructs.
Voltage-gated Sodium channels are integral membrane proteins that form ion channels, conducting sodium ions (Na+) through a cell's plasma membrane.

A growth cone is a large actin-supported extension of a developing or regenerating neurite seeking its synaptic target. It is the growth cone that drives axon growth. Their existence was originally proposed by Spanish histologist Santiago Ramón y Cajal based upon stationary images he observed under the microscope. He first described the growth cone based on fixed cells as "a concentration of protoplasm of conical form, endowed with amoeboid movements". Growth cones are situated on the tips of neurites, either dendrites or axons, of the nerve cell. The sensory, motor, integrative, and adaptive functions of growing axons and dendrites are all contained within this specialized structure.
A neurite or neuronal process refers to any projection from the cell body of a neuron. This projection can be either an axon or a dendrite. The term is frequently used when speaking of immature or developing neurons, especially of cells in culture, because it can be difficult to tell axons from dendrites before differentiation is complete.
Neuroproteomics is the study of the protein complexes and species that make up the nervous system. These proteins interact to make the neurons connect in such a way to create the intricacies that nervous system is known for. Neuroproteomics is a complex field that has a long way to go in terms of profiling the entire neuronal proteome. It is a relatively recent field that has many applications in therapy and science. So far, only small subsets of the neuronal proteome have been mapped, and then only when applied to the proteins involved in the synapse.

Neuronal calcium sensor-1 (NCS-1) also known as frequenin homolog (Drosophila) (freq) is a protein that is encoded by the FREQ gene in humans. NCS-1 is a member of the neuronal calcium sensor family, a class of EF hand containing calcium-myristoyl-switch proteins.
Synaptojanin is a protein involved in vesicle uncoating in neurons. This is an important regulatory lipid phosphatase. It dephosphorylates the D-5 position phosphate from phosphatidylinositol (3,4,5)-trisphosphate (PIP3) and Phosphatidylinositol (4,5)-bisphosphate(PIP2). It belongs to family of 5-phosphatases, which are structurally unrelated to D-3 inositol phosphatases like PTEN. Other members of the family of 5'phosphoinositide phosphatases include OCRL, SHIP1, SHIP2, INPP5J, INPP5E, INPP5B, INPP5A and SKIP.
Biomimetic materials are materials developed using inspiration from nature. This may be useful in the design of composite materials. Natural structures have inspired and innovated human creations. Notable examples of these natural structures include: honeycomb structure of the beehive, strength of spider silks, bird flight mechanics, and shark skin water repellency. The etymological roots of the neologism biomimetic derive from Greek, since bios means "life" and mimetikos means "imitative",
A nerve guidance conduit is an artificial means of guiding axonal regrowth to facilitate nerve regeneration and is one of several clinical treatments for nerve injuries. When direct suturing of the two stumps of a severed nerve cannot be accomplished without tension, the standard clinical treatment for peripheral nerve injuries is autologous nerve grafting. Due to the limited availability of donor tissue and functional recovery in autologous nerve grafting, neural tissue engineering research has focused on the development of bioartificial nerve guidance conduits as an alternative treatment, especially for large defects. Similar techniques are also being explored for nerve repair in the spinal cord but nerve regeneration in the central nervous system poses a greater challenge because its axons do not regenerate appreciably in their native environment.

Glial scar formation (gliosis) is a reactive cellular process involving astrogliosis that occurs after injury to the central nervous system. As with scarring in other organs and tissues, the glial scar is the body's mechanism to protect and begin the healing process in the nervous system.

Receptor-type tyrosine-protein phosphatase mu is an enzyme that in humans is encoded by the PTPRM gene.

Cellular adhesions can be defined as proteins or protein aggregates that form mechanical and chemical linkages between the intracellular and extracellular space. Adhesions serve several critical processes including cell migration, signal transduction, tissue development and repair. Due to this functionality, adhesions and adhesion molecules have been a topic of study within the scientific community. Specifically, it has been found that adhesions are involved in tissue development, plasticity, and memory formation within the central nervous system (CNS), and may prove vital in the generation of CNS-specific therapeutics.
Fasciclin 2 is a 95 kilodalton cell membrane glycoprotein in the immunoglobulin (Ig) – related superfamily of cell adhesion molecules (CAMs). It was first identified in the developing grasshopper embryo, seen dynamically expressed on a subset of fasciculating axons in the central nervous system (CNS), functioning as a neuronal recognition molecule in the regulation of selective axon fasciculation. Subsequently, fasII was cloned and has mainly been studied in the fruit fly. Its extracellular structure consists of two Fibronectin type III domains and five Ig-like C2 domains, having structural homology to the neural cell adhesion molecule (NCAM) found in vertebrates. Alternative splicing of fasII gives rise to its expression in three major isoforms, including a membrane-associated form that is attached to the outer leaflet of the plasma membrane via a glycophosphatidylinositol linkage and two integral transmembrane forms. The larger transmembrane form has an amino acid motif contained in its cytoplasmic domain that is rich in proline, glutamic acid, serine and threonine residues. The fasciclin 1 (Fas1) and fasciclin 3 (Fas3) genes in Drosophila also code for cell adhesion proteins in the nervous system but do not show any structural or functional similarities with NCAM.

Alain Prochiantz is a neurobiology researcher and professor at the Collège de France, of which he became director from 2015 to 2019.
References
- ↑ Palmer, Amy Melisa (2001-01-01). "Neuronal galvanotropism: An investigation of mechanisms and application". Theses and Dissertations Available from ProQuest: 1–97.
- ↑ Euskirchen, Nadine; Nitsche, Michael A.; van Thriel, Christoph (December 2021). "Direct Current Stimulation in Cell Culture Systems and Brain Slices—New Approaches for Mechanistic Evaluation of Neuronal Plasticity and Neuromodulation: State of the Art". Cells. 10 (12): 3583. doi:10.3390/cells10123583. ISSN 2073-4409. PMC 8700319 . PMID 34944091.